2016 Volume 22 Issue 6 Pages 727-732
2016 Volume 22 Issue 6 Pages 727-732
Mantı, the traditional Turkish food, was subjected to modified atmosphere packaging (MAP) to extend its refrigerated storage time. Beef, which is one of the main raw ingredients in mantı, is a highly perishable product. Therefore, it is essential that the microbiological effects of this product be analysed. The microbiological qualities of each mantı package were assessed by analysing raw mantı samples to determine the number of Lactobacillus spp. they contained, as well as the total anaerobic mesophilic microorganisms, the amounts of Staphylococcus spp., yeast and mould, the coliforms, Escherichia coli, Salmonella spp. and Clostridium perfringens and the total aerobic psychrophilic microorganisms. Salmonella spp. and C. perfringens were not detected in the raw materials or the packed samples, but the Lactobacillus spp. and total anaerobic mesophilic microorganism counts increased during cold storage. Growth of Enterobacteriaceae species in a modified atmosphere during the period in which the mantı is being stored must also be taken into account. The compositions of the MAP samples lasted for the maximum storage time of 126 days as opposed to 20 days in normal atmospheric packaging. In conclusion, more than 40% carbon dioxide (CO2) with nitrogen (N2) should be used in the mantı process.
In Turkey, the traditional food, mantı, is commonly consumed as a nutritious product. Mantı is made using wheat flour dough stuffed with specific combinations of minced beef, onion, paprika, black pepper and salt (Sitti et al., 2009; Öztürk et al., 2009). With its high water activity (>0.87) and the risk of unhygienic production conditions, mantı must be classified as a perishable food (Uzunlu and Var, 2016). Therefore, it certainly has a limited shelf life. However, mantı does have potential for both export and domestic markets.
Modified atmosphere packaging (MAP) is one of the food processing technologies that can extend the shelf lives of many foods. Nitrogen (N2), oxygen (O2) and carbon dioxide (CO2) are used extensively to modify atmospheric gas compositions. CO2 is commonly used for its antimicrobial effect, and CO2-enriched atmospheres have antimicrobial effects on refrigerated foods when temperatures are higher than the ambient conditions (Phillips, 1996; Rao and Sachindra, 2002; Irkin and Kızılırmak, 2010).
Many researchers have studied the effects that MAP gases (CO2 and N2) in cold storage have on the shelf life of raw meat. For example, Ho et al. (2003) reported on the retarding effect of 50% CO2 + 50% N2 packaging on the growth of microbial populations in beef. However, Barrera et al. (2007) have also emphasised the importance of a steady refrigeration period in lamb meat, whether it has been packaged with modified atmospheres or not. Gökoğlu et al. (2011) determined the optimum MAP composition for 14 days of shelf life for beef stored at 4°C to be 70% CO2 + 30% N2.
A number of foods that are similar to mantı, including ravioli, cannelloni, fettuccine and tortellini, have also been documented in the literature. A patent was filed for 4 months stability for fettuccine at 4.4°C. The method included steam pasteurisation at 82°C and packaging with a gas composition of 80% CO2 + 20% N2(McGuire et al., 1989). Liggett et al. (1990) used a similar method for tortellini, which achieved a shelf life of 4 months. Giannuzzi (1998) has also reported the cooking and holding periods before serving to be the critical control points for ravioli according to the hazard analysis and critical control point assessment.
Sanguinetti et al. (2011) reported that combining steam pasteurisation at 91°C with 50% CO2 + 50% N2 packaging resulted in 6 weeks of shelf life at 4°C for fresh pasta.
The current study aimed to determine the optimum MAP composition of mantı during refrigerated storage at 4°C by measuring microbiological parameters.
1.Mantı samples Wheat flour, minced beef meat, onion, black pepper, salt and water were used as the raw materials of mantı. Dough was prepared by mixing wheat flour (65%w/w of dough) and water (35%w/v of dough). The filling was prepared by mixing minced beef meat (23%w/w of mantı), kneaded onion (5%w/w of mantı), salt (2%w/w of mantı) and black pepper (0.5%w/w of mantı). The filling material was added to each square of dough material in an equal amount, averaging four measurements of 1.275 g. The heat treatment was performed in an oven with a product centre temperature of 60°C and treatment time of 4 min. The temperature was monitored by using a manual thermo logger. The product was left to cool at ambient temperature (∼20°C) and immediately transported under refrigeration (4°C) for MAP treatment.
2.MAP of the samples Samples of mantı were packaged under different gas combinations at the plant Antalya Altın Et Entegre Tesisleri, Turkey. Samples were placed in polypropylene (PP) trays (155x197x55 mm) with a water vapour transmission rate of 0.629 g/(m2 · day) and trays were sealed with a 25 µm thick polyolefin based film, an oxygen transmission rate of 24 cm3/m2 · day · bar and with an 18 g/(m2 · day) (38°C, % 100 RH) water vapour transmission rate. MAP was carried out using G. Mondini (Italy) packaging machine. Sealing temperature was 150°C and a 2/1 gas volume to mantı weight ratio in each pack was applied. Gas mixtures, volume based, were designed for MAP 1 (80% CO2 + 20% N2), MAP 2 (40% CO2 + 60% N2), MAP 3 (60% CO2 + 40% N2) and Control (atmospheric gas composition) treatments.
Samples from MAP 1, MAP 2 and MAP 3 were stored at 4°C, 50 – 63% RH for 126 days. Control samples were stored at 4°C, 50 – 63% RH until they reached the spoilage period, then mould mycelia inside the tray was observed. Analyses were performed on days 0, 1, 3, 5 and 7 and then at seven day intervals up to the day 126. Maximum storage day was determined by spoilage record, both visually and growth data of microorganisms.
3. Microbiological analysis
3.a) Analysis performed before MAP treatment Before and after heat treatment, the dough and filling of mantı samples were analysed separately for total aerobic mesophilic microorganisms (TAMB), Staphylococcus aureus, C. perfringens, Salmonella spp., Lactobacillus spp., yeast and mould, Coliform and Escherichia coli. Petri dishes were incubated at 30°C for 24 – 48 h for all aerobic mesophilic microorganisms; other analyses were performed as detailed below. Microbiological data were expressed in logarithm basis for the number of colonies (CFU/g).
3.b) Analyses performed after MAP treatment Lactobacillus spp., total anaerobic mesophilic microorganisms, S. aureus, yeast and mould, Coliform, E. coli, Salmonella spp., C. perfringens and total aerobic psychrophilic microorganisms were analysed during the storage period.
Maximum Recovery Diluent (Merck, Darmstadt-Germany) was used for homogenization and dilution, except in the Salmonella spp. analyses where buffered peptone water (Merck) was used (Anonymous, 2007).
Total aerobic psychrophilic microorganisms and total anaerobic mesophilic microorganisms were enumerated at Plate Count Agar (Merck). Anaerobic mesophilic counts were incubated at 25°C for 24 – 48 h by using Anaerocult A (Merck) in jars, and total aerobic psychrophilic microorganisms were incubated aerobically at 4°C for seven days (Anonymous, 2007).
MRS agar (de MAN, ROGOSA and SHARPE (Merck) was used for Lactobacillus spp. and were incubated anaerobically by using Anaerocult A (Merck) in jars at 25°C for two days (Anonymous, 2007). Morphologic and biochemical analyses were performed for identification. Baird Parker Agar (Merck) was used to estimate Staphylococcus spp. counts. After incubation at 35°C for 45 – 48 h, typical colonies were tested for detection of coagulase production. For this purpose, Staphylase test kits (Oxoid, Hampshire-UK) were used. Coagulase positive colonies were counted as S. aureus on the plates containing 20 – 200 colonies, coagulase (-) clear colonies were evaluated as coagulase negative Staphylococcus spp. (Anonymous, 2007).
Yeast and mould were counted using Potato Dextrose Agar (Merck) and incubated at 25°C for seven days (Anonymous, 2007). For identification of the mould species, media characteristics and cell morphology were investigated (Samson et al., 1981).
Enumerations of Coliform and E. coli were performed separately within a solid and broth medium. For this purpose, double layer Fluorocult Violet Red Bile (VRB) Agar (Merck) and Fluorocult Lauryl sulphate broth (LST) Broth (Merck) were used and incubated at 35°C for 16 – 18 h and 35°C for 24 – 48 h, respectively. After the incubation period, samples were exposed to a long wave (366 nm) UV light (Merck) in a darkened area and colonies or tubes that reflect fluorescence were counted as E. coli on plates and tubes by using the most probable number tables with biochemical tests. Gram stain, indole production, Voges-Proskauer reactive compounds, Methly red reactive compounds and citrate utility tests were evaluated to differentiate Coliform group bacteria (Anonymous, 2007).
Analyses of Salmonella spp. were carried out in four steps. Briefly, 25 grams of samples were weighed in Buffered Peptone Water (Merck) for non-selective pre enrichment step for 16 – 20 h at 35°C. Following the incubation, for selective enrichment step 0.1 mL and 10 mL were transferred to 10 mL of Rappaport Vassiliadis Broth (Merck) and 100 mL of Selenite Cystein Broth (Merck) and incubated at 42°C and 35°C for 24 h, respectively. After incubation a loopful were streaked on both Salmonella Shigella Agar (Merck) and XLT4 Agar (Merck), and incubated at 35°C for 24 – 48 h. Colonies isolated from each media were further analysed by biochemical reactions (lactose, gas from glucose, H2 S, urea, indole, citrate, lysine decarboxylase), where necessary API 10 E (Biomérieux, Marcy I'Etoile-France) was used to identify the bacteria (Anonymous, 2007).
4.Statistical analysis Each trial was repeated twice and triplicate samples were tested at each sampling time. Data were subjected to variance analysis in order to determine the effect of gas composition and storage time on each variable. The analysis was performed using analysis of variance (ANOVA) one-way analysis, and statistical package (SPSS 15.0, USA) was used to identify the different groups. The Duncan's post hoc test was applied; significance level was p < 0.05. Control group samples were not included in statistical analysis owing to the earlier spoilage record on day 21st.
The microbiological analyses demonstrated the efficacy of the heat treatment. Maximum log reductions immediately after the heat treatment were documented: 5 log CFU/g for TAMB and 4 log CFU/g for Lactobacillus spp. C.perfringens, Salmonella spp. and E.coli were not found before and after heat treatment, while moulds and Coliform grew slightly before heat treatment, however they were absent after the treatment. S.aureus was not found in dough; however they were counted in filling as 2.84 log CFU/g.
After heat treatment microbial growth were prevented by the effect of heat treatment at 60°C for 4 min. It should be noted that the filling showed a higher microbial load than the dough. Similar results have been reported (Sanguinetti et al., 2011). Earlier researches demonstrated that microorganisms' resistance against heat treatment might be variable at various food stuffs. In general, yeast and moulds resistance are lower than bacteria, which pertains to an inhibition at 60 – 65°C for 5 – 10 min.
In the case of effect of modified atmosphere packaging; total anaerobic mesophilic bacteria growth were recorded slightly (<1 log CFU/g) until day 5, while control group reached to 3.62 log CFU/g on day 5 (Fig. 1). From day 7 till to the end of storage period under all MAP compositions growth increased, however counts were displayed any significance (p < 0.05) until day 91 (data not shown). Control group samples reached to 6.73 log CFU/g on day 21, in which were the product spoiled and detected visually. Thereafter, control samples were not furthermore analysed. Under some conditions, microbial growth may stop and then restart after the microorganisms adapt to a new set of environmental conditions (Rao and Sachindra, 2002).
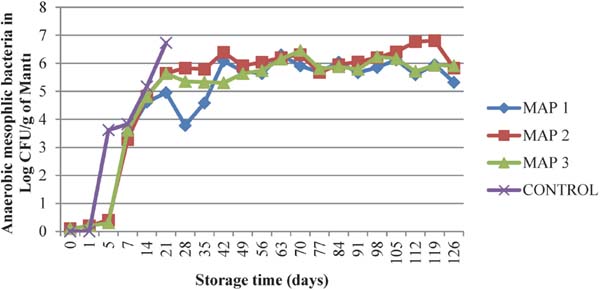
Evolution of total anaerobic mesophilic microorganisms during storage at 4°C (log10 CFU/g)
This is in agreement with our findings that microorganisms were adapted and grew after heat and MAP treatment (Fig. 1 and Fig. 2). Anaerobic microorganisms growth rate were faster than lactobacilli initially, while they reached to similar rates after 49 days (data not shown).

Evolution of Lactobacilli during storage at 4°C (log10 CFU/g)
MAP 1 (80% CO2 + 20% N2), MAP 2 (40% CO2 + 60% N2), MAP 3 (60% CO2 + 40% N2) and Control (atmospheric gas composition) treatments.
Meanwhile, modified atmosphere packaging affected the growth of Lactobacilli either. Growth of bacteria were recorded slightly (<1 log CFU/g) until day 5 in all package compositions, which reached to the countable values (approx. 1 log CFU/g) on day 7. Microbial growth increased during the storage period (Fig. 2), and it was significantly different among the 3 compositions on day 84th (data not shown). Control group samples reached to 4.91 log CFU/g on day 21, which were the last record, because of the spoilage (Fig. 2).
Researches highlighted that, lactobacilli dominate the microflora of food stuffs that stored in anaerobic conditions (Rodriguez et al., 1999; Rao and Sachindra, 2002). Lactobacilli are able to grow even at low temperature degrees, resistant to CO2 and produces higher activation energy than G (-) bacteria under temperature abuse (Lambert et al., 1991; Rodriguez et al.,1999; Kennedy et al., 2004; Berruga et al., 2005; Estürk and Ayhan, 2009; Limbo et al., 2010; Kızılırmak et al., 2011). It was also stated that modified atmosphere packaging for beef meat using CO2/N2 inhibits lactobacilli growth more than vacuum packaging (Rao and Sachindra, 2002). Lactobacilli produce CO2 that causes swelling in packaging, therefore recent researches have been focusing on preventing Lactobacilli growth in modified atmosphere packed foods (Rodriguez et al., 1999; Degirmencioglu et al., 2011). It is in accordance with our results that Lactobacilli grew in cold and CO2 atmospheres. It can be concluded that CO2 rates supressed the growth, and protected the food when compared to its control.
S. aureus displayed values of 101 CFU/g under MAP 2 conditions after 5 days of storage. However, it did not survive longer, while coagulase negative Staphylococcus spp. was rarely present during the storage period (data not shown). S.aureus is able to grow at aerobic conditions, sensitive to heat treatment and abused incubation temperatures (Singh et al., 2011). A research documented that S. aureus inactivated by using 80% CO2 at 2 – 7°C temperature range in 3 days (Rao and Sachindra, 2002). Our findings stated that heat treatment, modified atmosphere packaging and cold storage inactivated surviving cells. It can be concluded that high CO2 compositions (MAP 1 and MAP 3) did not allow any growth of coagulase negative S.aureus.
Yeast and mould grew in control group samples. Yeast reached to 2.38 log CFU/g on day 14th and 1 week later they increased to 4.94 log CFU/g. Moulds were slightly observed, however they reached to 5.75 log CFU/g on day 21st at air packed (control) samples. As a result, they could be visually detected and discarded for further analysis. Moulds were belonged to Penicillium spp.
Salmonella was absent in 25 g of the products. However, Coliform grew under all MAP compositions and control samples with a slightly (<1 log CFU/g) record until day 63, while E. coli was found under MAP 2 composition on day 63 of storage. Hafnia alvei and Enterobacter aerogenes survived until days 42 and 70, respectively. However, Citrobacter freundii were found in both dough and filling and were able to grow until day 126 in all MAP conditions. In dough C.freundii, and in filling C.freundii and Proteus spp. were found, while in MAP 1 C.freundii, Enterobacter aerogenes and E.coli, in MAP 2 samples C.freundii, E.aerogenes, Hafnia alvei/Serratia marcescens, in MAP 3 C.freundii, E.aerogenes, H.alvei/S.marcescens and E.coli, in control group C.freundii and H.alvei/S.marcescens were found. When taking account the facultative anaerobe growth characteristic of Enterobacteriaceae, several researches documented that they might be able to grow at various modified atmospheric conditions and frequently isolated from beef meat, such as our isolates. These finds are in accordance with our results (Skandamis and Nychas, 2002; Berruga et al., 2005; Ercolini et al., 2006; Argyri et al., 2011; Doulgeraki et al., 2011). Aerobic psychrophilic microorganisms showed a slight increase but remained below the spoilage tolerances of 7 log CFU/g (data not shown).
There is a concern with the modified atmosphere packed products to create an anaerobic condition that might allow growth of Clostridium species. However, cold storage and preventing temperature fluctuations during transporting and/or storage might prevent their growth. It is not a solution to increase CO2 and O2 rates in headspace to prevent the growth (Phillips 1996; Rao and Sachindra, 2002). In addition, vegetative cells of C. perfringens would be inactivated at 60°C degrees (Danler et al., 2003). It might be the reason that our samples were absence from Clostridium species owing to the heat treatment at pre-packaging.
In conclusion, the optimum refrigerated storage for mantı was provided using the MAP 1 (80% CO2 + 20% N2) composition and was for 4 months, while this was only 3 weeks for the control (atmospheric gas composition) samples.
Acknowledgments This research was funded by Çukurova University (Project Nr: ZF2010D6). The assistance provided by Muzaffer Yılmaz and Cemil Karakoç, Barıç Koçak, Nadide Koçak, Lütfi Aydın and Süleyman Yılmaz is also appreciated.