抄録
In order to provide a prediction tool for sulfide/oxide/oxysulfide inclusion evolution in Mn–Al steel with a Ca addition/CaO-based flux, a comprehensive thermodynamic database for the inclusion system composed of CaO–MnO–Al
2O
3–CaS–MnS–Al
2S
3 was developed in the present study. Activity of MnS in a CaS–MnS sulfide solid solution was experimentally determined by employing a chemical equilibrium technique at 1400°C and 1500°C. The measured activity exhibits a positive deviation from an ideal behavior, which is in consistent with the known two-phase separation of the sulfide solid solution at lower temperature (T
cr = ~ 1200°C). Based on the activity and the phase diagram data available in literature, a thermodynamic modeling of the CaS–MnS system was carried out. The following excess Gibbs free energy of the CaS–MnS sulfide solid solution was obtained:
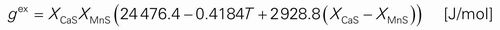
Furthermore, using available thermodynamic modeling results for other constituent sub-systems, a larger thermodynamic database of the CaO–MnO–Al
2O
3–CaS–MnS–Al
2S
3 system was developed. A Modified Quasichemical Model in the quadruplet approximation was used to model the Gibbs free energy of the oxysulfide liquid solution. Comparisons between the model calculation and available experimental data show good agreement. The developed thermodynamic model and the database were used to predict unexplored phase diagrams with various
nMn/(
nCa +
nMn) ratio, and sulfide capacity of the CaO–MnO–Al
2O
3 oxide liquid phase. The database can be used along with software for Gibbs free energy minimization in order to calculate any phase diagram section or thermodynamic property.