2015 Volume 57 Issue 2 Pages 118-125
2015 Volume 57 Issue 2 Pages 118-125
Objectives: It is known that inhalation of zinc oxide nanoparticles (ZnO NPs) induces acute pulmonary dysfunction, including oxidative stress, inflammation, and injury, but there are no reports on how to prevent these adverse effects. We have previously reported that the pulmonary symptoms caused by ZnO NPs were associated with oxidative stress; in the present study, we therefore investigated the use of ascorbic acid (AA), which is known as vitamin C, to prevent these toxic effects. Methods: A ZnO NP dispersion was introduced into rat lungs by intratracheal injection, and thereafter a 1% aqueous AA solution was given as drinking water. Bronchoalveolar lavage fluid was collected at 1 day and 1 week after injection, and lactate dehydrogenase (LDH) activity, heme oxygenase-1 (HO-1), and interleukin-6 (IL-6) levels were measured. In addition, expression of the chemokine cytokine-induced neutrophil chemoattractants (CINCs), HO-1, and metallothionein-1 (MT-1) genes in the lungs were determined. Results: Acute oxidative stress induced by ZnO NPs was suppressed by supplying AA. Increases in LDH activity and IL-6 concentration were also suppressed by AA, as was the expression of the CINC-1, CINC-3, and HO-1 genes. Conclusions: Oral intake of AA prevents acute pulmonary oxidative stress and inflammation caused by ZnO NPs. Intake of AA after unanticipated exposure to ZnO NPs is possibly the first effective treatment for the acute pulmonary dysfunction they cause.
(J Occup Health 2015; 57: 118–125)
A nanoparticle is defined as a particle in the size range 1–100 nm (ISO/TS 27687, 2008). Currently, many kinds of metal oxide nanoparticles are being produced in increasing amounts. These nanoparticles can cause biological effects, including toxicity, for which some mechanisms have been suggested. One of the major toxic effects is metal ion release1–3). In some cases, nanoparticles release greater quantities of metal ions than microscale particles4, 5).
Zinc oxide nanoparticles (ZnO NPs) are one of the most abundantly produced metal oxide nanoparticles. They have industrial applications and are used in the manufactured of cosmetics and sunscreen. Use of this product is increasing, as is the possibility of production workers being accidentally exposed to it. A complete understanding of ZnO NPs toxicity is therefore necessary. Currently, there are many reports on the toxicity of ZnO NPs6–13), as well as on the level of exposure, and many studies suggest that the most important factor in ZnO NP toxicity is zinc ion (Zn2+) release14–16). The major cellular effect of ZnO NPs is the induction of oxidative stress. When cells are exposed to ZnO NPs, intracellular reactive oxygen species (ROS) are increased by the released Zn2+ 6–13). Moreover, excess ROS generation leads to oxidative stress from which the cells eventually die. A previous study showed that exposure of human respiratory epithelial cells (BEAS-2B) to ZnO NPs decrease cell viability in a time-dependent manner but that intracellular ROS induction and cell death were prevented by N-acetyl cysteine7). We previously reported that Zn2+ released from ZnO NPs was linked to acute oxidative stress in rat lungs after intratracheal instillation1). Hydroxyoctadecadienoic acid, the oxidative product of linoleic acid, was significantly increased in the rat lung by exposure to ZnO NPs. Induction of inflammation is another effect that, in human cells, leads to an inflammation-related response17–20). ZnO NPs induce expression of the interleukin-8 (IL-8) gene18, 20) and IL-8 production17, 19). They also increase intracellular ROS levels and IL-8 production in human renal cells from the proximal tubule epithelium in a dose-dependent manner19). Furthermore, Gojova et al.18) reported that exposure dose-dependently increased Zn uptake and levels of inflammatory marker mRNA in human aortic endothelial cells. It is therefore possible that Zn2+ released from ZnO NPs is associated with inflammation responses. Indeed, their capacity to generate ROS in vitro seems to correlate with their potential to induce inflammation in vivo21). These data suggest that Zn2+ release is possibly the most important factor in the toxic effects of these nanoparticles. The released Zn2+ induces the intracellular ROS generation associated with oxidative stress and inflammation. Prevention of toxic effects induced by ZnO NPs forms an essential component of safe manufacturing and effective utilization of ZnO NPs; however, no studies have discussed approaches to prevent these toxic effects. Prevention of ZnO NP-induced toxicity is influenced by the prevention of oxidative stress, because induction of oxidative stress is a key response in such toxicity.
In this study, we employed l-ascorbic acid (AA), a well-known radical scavenger, as an antioxidant. By trapping radicals, AA prevents cell membrane oxidation and injury caused by ROS. Moreover, treatment with AA has been shown to reduce intracellular lipid peroxidation induced by talc nanoparticles in A549 cells22), and Ahmad et al.23) reported that AA prevented ROS generation and activation of apoptosis signaling caused by silica nanoparticles. Additionally, AA potentially prevents not only oxidative stress but also inflammation. Tebbe et al.24) reported that AA decreased secretion of the pro-inflammatory cytokines, IL-1α and IL-6, caused by UVA irradiation of human keratinocytes.
The aim of the present study was to use AA to prevent the acute pulmonary symptoms caused by ZnO NPs. Because ZnO NPs are known to induce oxidative stress and inflammation in the lung, we monitored biomarkers of oxidative stress and pro-inflammation responses.
ZnO NPs (product name FZO-50) were purchased from Ishihara Sangyo Kaisha, Ltd. (Osaka, Japan). According to the manufacturer's material data sheets, the primary particle size was 21 nm, the purity was 97.4%, and the specific surface area was 49.6 m2/g. Ascorbic acid was purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan).
Preparation of ZnO NP dispersion for intratracheal instillationThe ZnO NP dispersion for intratracheal instillation in this study was the same as that we previously reported1). To prepare the dispersion, 0.5 g of ZnO NP powder was mixed into 50 ml of endotoxin-free distilled water containing 5% (v/v) fetal bovine serum (FBS; CELLect GOLD; MP Biomedicals Inc., Solon, OH, USA). The particles were dispersed by ultrasonication for 3 hours in an ultrasonic bath (Branson 5510J-MT, 135 W, 42 kHz), with the temperature maintained below 30°C. Secondary particle size was adjusted by centrifugation at 16,000 × g for 5 minutes at room temperature. Thirty to 40 ml of the supernatant was collected, and the ZnO concentration was determined by UV absorbance at 380–700 nm. There was no endotoxin activity in the dispersion, as determined by the Limulus Amebocyte Lysate Assay (Pyrotell; Associates of Cape Cod, Inc., East Falmouth, MA, USA). The size distribution of the ZnO particles in the dispersion was measured by dynamic light scattering (DLS) using a Zetasizer Nano (Malvern Instruments Limited, Malvern, United Kingdom). The soluble zinc concentration in the dispersion was detected by colorimetry using 2-(5-bromo-2- pyridylazo)-5-[N-n-propyl-N-(3-sulfopropyl) amino] phenol, disodium salt, and dihydrate (5-Br-PAPS) (Dojindo Laboratories, Kumamoto, Japan), in accordance with the procedure outlined by Makino et al.25) The ZnO dispersions were centrifuged at 16,000 × g for 20 minutes, and the supernatant was carefully collected. After suitable dilution of the supernatant, 400 µl of supernatant, 100 µl of 5 mM APS, 100 µl of 1 mM DTCS and 50 µl of 1 mM 5-Br-PAPS were added to 2.0 ml of a buffer (6 g of NaOH dissolved in 500 ml of 0.5 M HEPES, pH 7.8). The above solution was mixed well and incubated for 10 minutes at room temperature. Its absorbance at 555 nm was measured using a DU 530-spectrophotometer (Beckman Coulter Inc., Miami, FL, USA). The soluble zinc concentration was estimated from the standard curve for the zinc standard solution (Wako Pure Chemical Industries).
Animals and intratracheal instillationAll animal experiments were approved by the Institutional Animal Care and Use Committee of the National Institute of Advanced Industrial Science and Technology (AIST). Eight-week-old male Wistar rats were obtained from CLEA Japan Inc. (Tokyo, Japan). Rats were fed a standard diet and allowed to acclimatize to their environment for 1 week before experimentation.
In our previous study, the induction of acute lung toxicity, which associated with oxidative stress and injury, was observed with a dose of 0.4 ml containing 0.2 mg ZnO NPs1). Therefore, the ZnO dispersion (dose of 0.2 mg/0.4 ml) was administered to rats in a single intratracheal instillation in this study. Control rats were given 0.4 ml of distilled water containing 5% FBS. After intratracheal instillation, the control and experimental groups were further subdivided into groups given water and groups receiving AA until autopsy. The AA groups were supplied with 1% AA water beginning immediately after ZnO NPs instillation. Each rat could drink water freely. There were at least four rats in each group (Table 1).
| Group | Intratracheal instillation | Supply of drinking after intratracheal instillation |
|---|---|---|
| Non-treated | — | — |
| Group 1 | Vehiclea | Water |
| Group 2 | AAc | |
| Group 3 | ZnO NPsb | Water |
| Group 4 | AAc |
At 24 hours or 1 week after exposure, rats were anesthetized by intraperitoneal injection of 50 mg/ml of 5-ethyl-5-(1-methylbutyl)-2,4,6-trioxohexahydropyrimidine (pentobarbital sodium salt; Sigma-Aldrich Corp., St. Louis, MO, USA). At autopsy, blood was taken from the abdominal aorta, and a lung was then perfused with saline. Bronchoalveolar lavage fluid (BALF) was recovered by administering 5 ml of saline into the right lung while clamping the left main bronchus. The injection and recovery of saline were repeated three times, and 15 ml of BALF was collected. It was then centrifuged at 1,500 rpm for 10 minutes to remove cell debris, and the supernatant was collected and used for measurement of markers. For real-time polymerase chain reaction (PCR), total RNA was prepared from the left lung tissue using the RNeasy Protect Mini Kit (Qiagen GmbH, Hilden, Germany).
Measurement of LDH in BALFLactate dehydrogenase (LDH) activity in BALF was measured using the Cytotoxicity Detection KitPLUS (LDH) (Roche Diagnostics GmbH, Mannheim, Germany) according to the manufacturer's instructions. The amount of formazan salt formed was measured at 492 nm using a Multiskan Ascent plate reader (Thermo Labsystems, Helsinki, Finland). LDH activity was estimated using a standard curve obtained from known concentrations of LDH from the porcine heart (Serva Electrophoresis GmbH, Heidelberg, Germany).
Measurement of the HO-1 concentration in BALFHeme oxygenase-1 (HO-1) concentrations in BALF were measured using an enzyme-linked immunosorbent assay (ELISA) with the Rat Heme Oxygenase-1 (HO-1) EIA Kit (Takara Bio Inc., Otsu, Japan) according to the manufacturer's instructions.
Measurement of the IL-6 concentration in BALFInterleukin-6 (IL-6) concentrations in BALF were measured using the Quantikine(R) IL-6 ELISA Kit (R&D Systems Inc., Minneapolis, MN, USA) according to the manufacturer's instructions.
Real-time PCRThe gene expression levels of HO-1, cytokine-induced neutrophil chemoattractant (CINC)-1, CINC-2, CINC-3 and MT-1 were determined by real-time PCR. Total RNA was isolated from lung tissue using the RNeasy Protect mini kit (Qiagen GmbH). Real-time PCR was conducted using a StepOne Real-Time PCR System (Life Technologies Corp., Carlsbad, CA, USA). Gene expression levels were analyzed by the ΔΔCt method. PCR amplification of lung tissue was analyzed using TaqMan® Gene Expression Assays (Life Technologies Corp.), with the rat β-actin gene as an endogenous control. The gene expression assays for HO-1, CINC-1, CINC-2, CINC-3, and MT-1 were designated as Rn00561387_m1, Rn00578225_m1, Rn00593435_m1, Rn00586403_m1, and Rn00821759_g1, respectively.
Statistical analysisData represent means ± SE of at least three separate experiments. Statistical analyses were performed via analysis of variance (ANOVA) using Tukey's test for multiple comparisons. The calculation methods are described in each figure legend.
We reported the characterization data for the primary particle in the ZnO NP and ZnO dispersion in our previous study1). The same dispersion was also used for this study. The primary particle size was approximately 20–50 nm based on TEM observation, and the average secondary particle size of the ZnO NPs in the ZnO water for intratracheal instillation was 101 nm based on the light intensity measured by DLS. The concentration of Zn2+ released from the ZnO NPs in the dispersion was 70 µg/ml.
AA treatment eliminates the rise in LDH activity caused by ZnO NPsResults for each animal group (1–4) are shown in Table 1. Lung injury was determined based on the LDH activity in BALF (Fig. 1). One day after intratracheal instillation of ZnO NPs, there was a significant increase in LDH activity in group 3 compared with the non-treated group. In contrast, despite the instillation of ZnO NPs, the group supplied with AA did not show any increase in LDH activity in BALF. After 1 week, no significant changes were observed in LDH activities in any of the groups compared with the non-treated group.

LDH activity in BALF after intratracheal instillation of ZnO NPs.
BALF was collected 1 day or 1 week after instillation of 0.4 ml of a dispersion containing 200 µg of ZnO NPs. After removing cells by centrifugation, LDH activity was measured. Significant differences versus the non-treated group are indicated in the figure (ANOVA, Tukey). *p<0.05.
The concentration of HO-1, one of the major oxidative stress response enzymes, increased significantly in BALF 1 day after intratracheal instillation of ZnO NPs (Fig. 2A). This was partially suppressed by supplying AA. Similarly, HO-1 gene expression in lung tissue was greatly induced 1 day after ZnO NPs instillation (Fig. 2B). In contrast, there was no induction of HO-1 gene expression in group 4. After 1 week, no significant differences were observed between the groups.
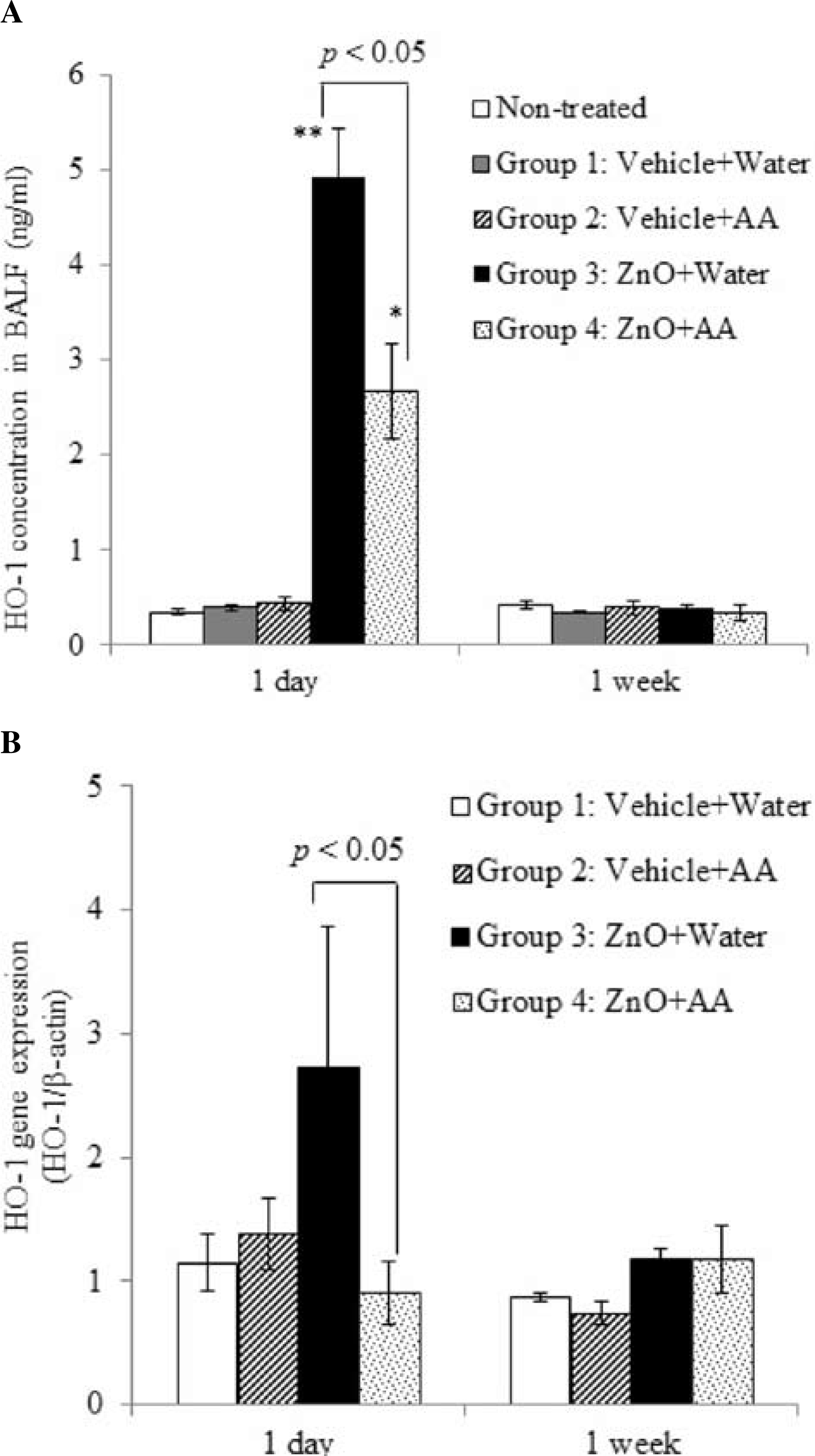
HO-1 concentration and gene expression in the lungs after intratracheal instillation of ZnO NPs.
(A) Effects of AA on HO-1 protein concentrations in BALF after intratracheal instillation of ZnO NPs. BALF was collected 1 day or 1 week after instillation of 0.4 ml of a dispersion containing 200 µg of ZnO NPs. An ELISA was then used to measure the concentration of HO-1. (B) Ratio of HO-1 gene expression relative to the non-treated group in the lung after instillation of 0.4 ml of a dispersion containing 200 µg of ZnO NPs. Gene expression of HO-1 was determined by real-time PCR. The values relative to the non-treated group are plotted. Significant differences versus the non-treated group are indicated in the figure (ANOVA, Tukey). *p<0.05; **p<0.01.
The concentration of IL-6, a pro-inflammatory cytokine, was measured in BALF. One day after ZnO NP instillation, the IL-6 concentration increased significantly in group 3 (Fig. 3A), but that in group 4 remained the same as that in the non-treated group. At this time point, we also observed the induction of CINC-1 and CINC-3 gene expression in group 3, but not in group 4, where it was suppressed (Fig. 3B and 3D). No large increases in CINC-2 expression were observed (Fig. 3C). After 1 week, there were no significant differences in IL-6 and CINCs between the groups.

IL-6 concentration and gene expression of CINCs in the lungs after intratracheal instillation of ZnO NPs.
(A) Concentration of IL-6 in BALF after intratracheal instillation of ZnO NPs. BALF was collected 1 day or 1 week after instillation of 0.4 ml of a dispersion containing 200 µg of ZnO NPs. An ELISA was then used to measure the concentration of IL-6. Ratios of (B) CINC-1, (C) CINC-2, and (D) CINC-3 gene expression relative to the non-treated group in the lungs after instillation of 0.4 ml of a dispersion containing 200 µg of ZnO NPs. The gene expression levels of CINC-1, CINC-2 and CINC-3 were measured by real-time PCR. The values relative to the non-treated group are plotted. No significant changes were observed. Significant differences versus the non-treated group are indicated in the figure (ANOVA, Tukey). *p<0.05.
MT-1 gene expression was induced 1 day after intratracheal instillation of ZnO NPs, and it was little suppressed by supplying AA. (Fig. 4). After 1 week, no significant differences were observed between the groups.

Gene expression of MT-1 in the lungs after intratracheal instillation.
Ratio of MT-1 gene expression relative to the non-treated group in the lungs after instillation of 0.4 ml of a dispersion containing 200 µg of ZnO NPs. Gene expression of MT-1 was measured by real-time PCR. The values relative to the non-treated group are plotted. Significant differences versus the non-treated group are indicated in the figure (ANOVA, Tukey). **p<0.01.
The plasma AA concentrations were increased in groups 2 and 4 after 1 day, but remained elevated only in group 4 after 1 week (Fig. 5).

Concentration of AA in plasma after intratracheal instillation of ZnO NPs.
Plasma was collected 1 day or 1 week after instillation of 0.4 ml of a dispersion containing 200 µg of ZnO NPs, with or without 1% AA. The concentration of AA in plasma was measured by HPLC. Significant differences versus the non-treated group are indicated in the figure (ANOVA, Tukey). *p<0.05.
It is known that ZnO NPs induce acute pulmonary dysfunction that is associated with oxidative stress1). They are also severely cytotoxic to cultured cells6–13). It was recently reported that an important factor in metal oxide nanoparticle toxicity is metal ion release2, 3). ZnO NPs release Zn2+ into culture media, inducing intracellular ROS. In addition, cellular studies revealed that intracellular Zn2+ release and induction of oxidative stress are key factors in the biological effects of ZnO NPs14–16), eventually leading to cell death. In vivo and in vitro studies have also shown that intratracheal instillation of ZnO NPs induces oxidative stress and transient injury in the rat lung1), as well as an increase in neutrophil cells in BALF26). Exposure of human cells to ZnO NPs also induces pro-inflammatory responses, such as increases in pro-inflammatory cytokines and chemokines17–20), among which IL-6 and CINCs are known to induce the accumulation of macrophages. These, along with other phagocytic cells, such as neutrophils, generate ROS27, 28). Thus, ZnO NPs cause two types of oxidative stress in the lung: primary (direct) stress caused by the released Zn2+, and secondary (indirect) stress caused by infiltration of neutrophils and macrophages.
In this study, we showed that AA, a well-known radical-scavenging antioxidant, inhibits the induction of pulmonary oxidative stress, injury, and inflammation observed in the rat lung 1 day after intratracheal instillation of ZnO NPs. HO-1 gene and protein expression increased in response to ZnO NP exposure, but not in rats given AA. These results suggest that AA suppresses oxidative stress caused by ZnO NPs. Analyses of BALF showed that AA also suppressed increases in IL-6 concentration and expression of the CINC gene family caused by ZnO NPs, as was increased LDH activity. These results suggest that AA suppresses both the induction of inflammation and injury. In contrast, MT-1 gene expression was induced following ZnO NP instillation, and remained elevated independently of AA, as our previous study with ZnO NPs and ZnCl2 had indicated1). These results suggest that the Zn2+ released from ZnO NPs induces MT-1 gene expression. We therefore conclude that AA has a suppressive effect on responses to oxidative stress, inflammation, and injury, but no effect on Zn2+ released from ZnO NPs in the lung. The relationship between inhibition of oxidative stress and inhibition of inflammation by AA is unclear. There is a possibility, however, that the preventive effect of AA against inflammation is due to inhibition of the production of ROS caused by activation of phagocytes, such as neutrophils and macrophages. Our data show that AA suppressed expression of CINC genes. CINCs are known as neutrophil chemoattractants; therefore, inhibition of their expression is associated with inhibition of neutrophil activation and the resultant ROS generation.
AA possibly suppresses inflammation by an indirect mechanism: enhancement of the anti-oxidative system. Oxidative stress is strongly related to inflammation, and it is known that ROS have a role as inflammation mediators29). Generally, orally ingested AA is absorbed in the small intestine and transported to tissues in blood. In this study, we observed a significant increase in AA concentrations in blood samples from the AA groups. On the other hand, it is known that the intracellular Zn2+ concentration is increased after ZnO NPs exposure1). Therefore, it is considered that induction of oxidative stress and inflammation caused by ZnO NPs in the lung was the result of uptake of ZnO NPs into cells. AA is also incorporated into cells through the membrane transporter SVCT30). In particular, it has been reported that both SVCT1 and SVCT2 are present in the human lung31). Consequently, there is the possibility that the AA was incorporated into the cells, and then showed the protective effect against the cytotoxicity of ZnO NPs in the cells. Moreover, there are also reports indicating that AA reduces intracellular lipid peroxidation induced by talc nanoparticles, and there are other reports indicating that AA inhibits intracellular ROS generation induced by silica nanoparticles22, 23). We therefore suggest that the supplied AA was transported to via the blood to lung tissue where it scavenged ROS caused by ZnO NPs. In this way, induction of pro-inflammatory responses mediated by ROS might be suppressed by AA acting as a radical scavenger. Nonetheless, the mechanism that induces ROS generation by ZnO NPs is unclear. There are two possibilities for the ROS generation mechanism. Namely, it is thought that the direct mechanism is Fenton-like reaction. In addition, there is also possibility that indirect mechanism is induced by dysfunction of mitochondrial electron transport system by Zn2+ released from incorporated ZnO NPs. The clarifications of the ROS generation mechanism caused by ZnO NPs, and prevention mechanism by AA is the future investigation task. Moreover, we evaluated only single dose of both ZnO NPs and AA in this study, and so it is also necessary to evaluate multiple doses in a future investigation.
In conclusion, the present study shows that supply of AA with drinking water for 1 day after intratracheal instillation of ZnO NPs has a suppressive effect on acute pulmonary oxidative stress, inflammation, and injury caused by ZnO NPs, although the mechanism is still unclear.