- |<
- <
- 1
- >
- >|
-
2025Volume 42 Pages Cover42_1
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Download PDF (1583K) Full view HTML -
2025Volume 42 Pages EdBrd42_1
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Download PDF (1113K) Full view HTML
-
2025Volume 42 Pages i-v
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Download PDF (6905K) Full view HTML
-
2025Volume 42 Pages vi
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Download PDF (802K) Full view HTML -
2025Volume 42 Pages 1-2
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Download PDF (997K) Full view HTML
-
2025Volume 42 Pages 3-14
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: June 29, 2024Download PDF (5764K) Full view HTML -
2025Volume 42 Pages 15-36
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: March 23, 2024Download PDF (4450K) Full view HTML -
2025Volume 42 Pages 37-56
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: August 31, 2024Download PDF (18709K) Full view HTML -
2025Volume 42 Pages 57-78
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: August 10, 2024Download PDF (14617K) Full view HTML -
 2025Volume 42 Pages 79-99
2025Volume 42 Pages 79-99
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
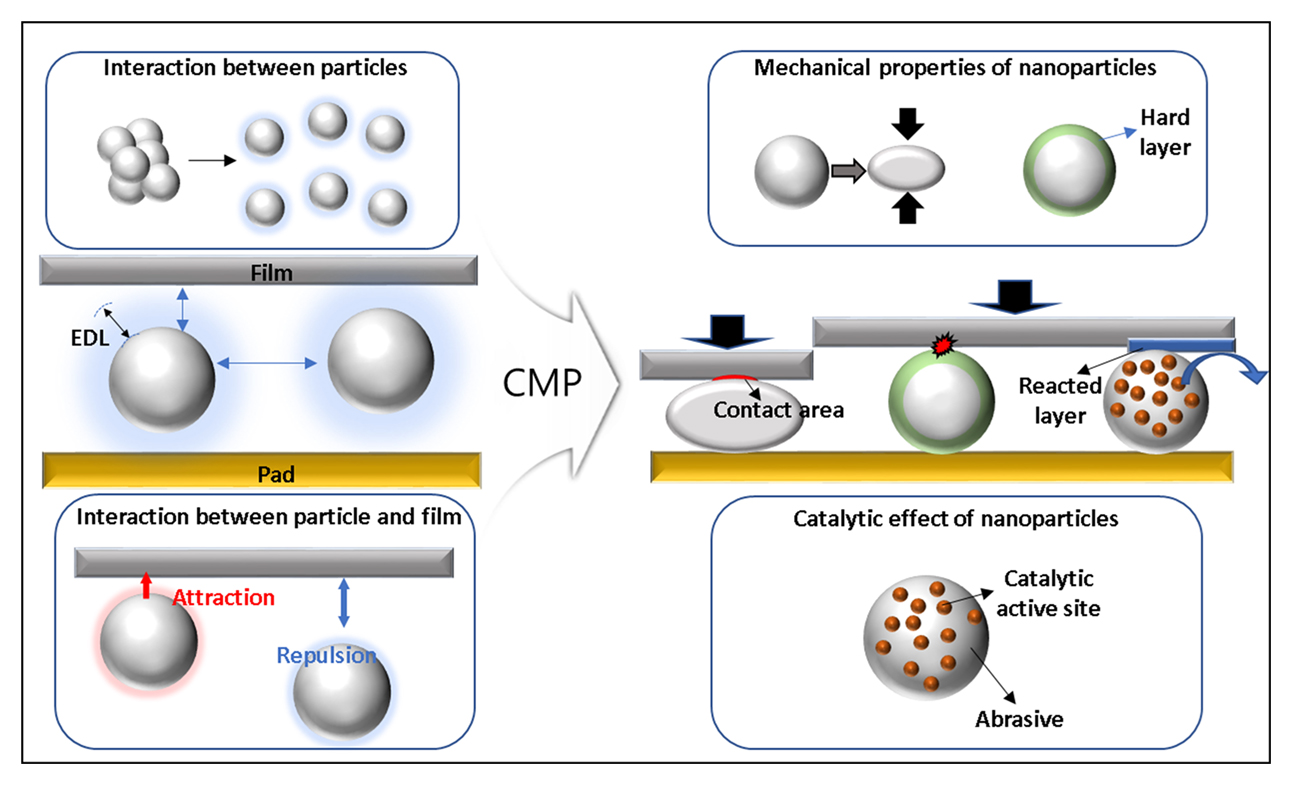
Advance online publication: December 28, 2023Editor's pickSiO₂ nanoparticles are the most widely used abrasives in chemical mechanical planarization (CMP) slurries, where their structure and surface properties govern dispersion stability, abrasive-abrasive interactions, and abrasive-film interactions. Controlling these factors is essential for achieving desired material removal rates, defect suppression, and polishing uniformity in CMP. This review explores SiO₂ nanoparticle engineering, focusing on synthesis approaches and surface modifications such as functionalization, coating, and doping to regulate dispersion stability, interfacial interactions, and chemical reactivity. It provides insights into how these engineered nanoparticles influence the CMP performance and their role in semiconductor manufacturing.
Download PDF (6303K) Full view HTML -
2025Volume 42 Pages 100-120
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: March 17, 2024Download PDF (4746K) Full view HTML -
2025Volume 42 Pages 121-133
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: April 06, 2024Download PDF (1801K) Full view HTML -
2025Volume 42 Pages 134-155
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: June 29, 2024Download PDF (18843K) Full view HTML -
2025Volume 42 Pages 156-169
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: February 24, 2024Download PDF (16823K) Full view HTML -
2025Volume 42 Pages 170-187
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: February 24, 2024Download PDF (25310K) Full view HTML -
2025Volume 42 Pages 188-199
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: August 10, 2024Download PDF (8069K) Full view HTML -
2025Volume 42 Pages 200-212
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: August 24, 2024Download PDF (2194K) Full view HTML -
2025Volume 42 Pages 213-226
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: March 17, 2024Download PDF (1968K) Full view HTML
-
2025Volume 42 Pages 227-240
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: January 13, 2024Download PDF (2132K) Full view HTML -
2025Volume 42 Pages 241-250
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: April 13, 2024Download PDF (2306K) Full view HTML -
2025Volume 42 Pages 251-260
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: June 29, 2024Download PDF (3228K) Full view HTML -
2025Volume 42 Pages 261-267
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: August 10, 2024Download PDF (1744K) Full view HTML -
2025Volume 42 Pages 268-284
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Advance online publication: October 19, 2024Download PDF (15086K) Full view HTML
-
2025Volume 42 Pages 285-287
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Download PDF (6975K) Full view HTML -
2025Volume 42 Pages 288
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Download PDF (1170K) Full view HTML -
2025Volume 42 Pages 289
Published: January 10, 2025
Released on J-STAGE: February 28, 2025
Download PDF (679K) Full view HTML
- |<
- <
- 1
- >
- >|