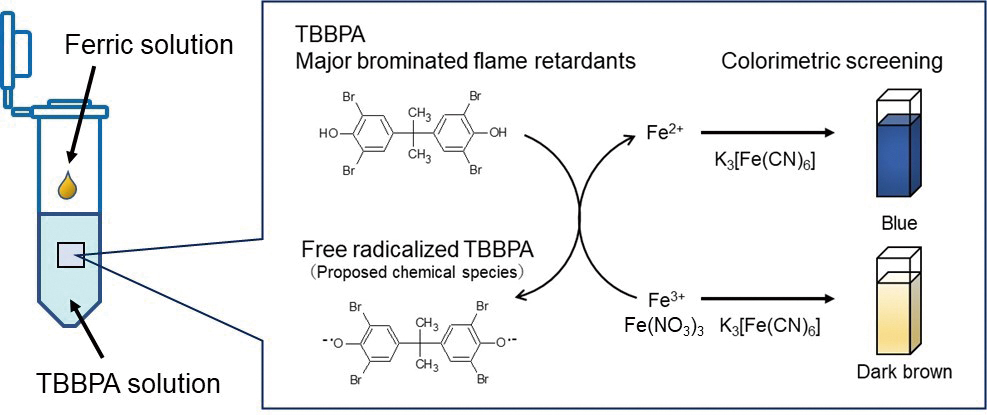
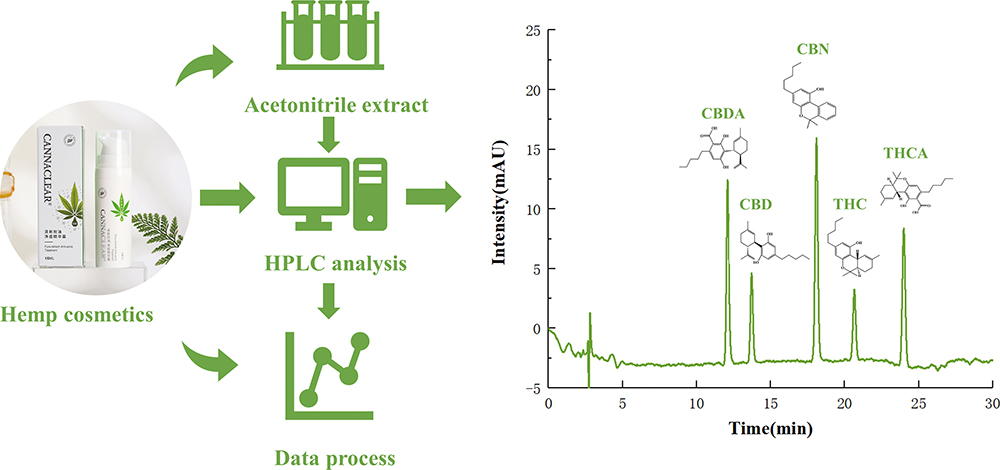
最新号
選択された号の論文の32件中1~32を表示しています
- |<
- <
- 1
- >
- >|
Highlights
-
原稿種別: Highlights
2021 年 37 巻 12 号 p. 1653-1654
発行日: 2021/12/10
公開日: 2021/12/10
PDF形式でダウンロード (163K)
Reviews
-
原稿種別: Reviews
2021 年 37 巻 12 号 p. 1655-1664
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/05/21PDF形式でダウンロード (320K) -
原稿種別: Reviews
2021 年 37 巻 12 号 p. 1665-1673
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/23PDF形式でダウンロード (888K)
Original Papers
-
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1675-1680
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2020/11/06PDF形式でダウンロード (625K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1681-1685
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/04/23PDF形式でダウンロード (320K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1687-1693
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/05/21PDF形式でダウンロード (1177K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1695-1700
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/05/21PDF形式でダウンロード (2740K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1701-1706
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/05/28PDF形式でダウンロード (656K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1707-1712
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/06/04PDF形式でダウンロード (385K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1713-1718
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/06/11PDF形式でダウンロード (441K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1719-1725
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/06/18PDF形式でダウンロード (437K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1727-1733
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/06/18PDF形式でダウンロード (622K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1735-1740
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/06/18PDF形式でダウンロード (165K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1741-1748
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/06/25PDF形式でダウンロード (1017K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1749-1755
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/02PDF形式でダウンロード (1580K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1757-1763
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/02PDF形式でダウンロード (248K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1765-1769
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/09PDF形式でダウンロード (771K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1771-1774
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/09PDF形式でダウンロード (122K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1775-1781
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/16PDF形式でダウンロード (257K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1783-1787
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/16PDF形式でダウンロード (427K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1789-1794
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/23PDF形式でダウンロード (535K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1795-1802
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/08/06PDF形式でダウンロード (704K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1803-1810
発行日: 2021/12/10
公開日: 2021/12/10
PDF形式でダウンロード (838K) -
原稿種別: Original Papers
2021 年 37 巻 12 号 p. 1811-1814
発行日: 2021/12/10
公開日: 2021/12/10
PDF形式でダウンロード (235K)
Notes
-
原稿種別: Notes
2021 年 37 巻 12 号 p. 1815-1819
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/06/18PDF形式でダウンロード (305K) -
原稿種別: Notes
2021 年 37 巻 12 号 p. 1821-1824
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/06/18PDF形式でダウンロード (234K) -
原稿種別: Notes
2021 年 37 巻 12 号 p. 1825-1828
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/02PDF形式でダウンロード (434K) -
原稿種別: Notes
2021 年 37 巻 12 号 p. 1829-1833
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/16PDF形式でダウンロード (490K) -
原稿種別: Notes
2021 年 37 巻 12 号 p. 1835-1837
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/16PDF形式でダウンロード (922K) -
原稿種別: Notes
2021 年 37 巻 12 号 p. 1839-1841
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/16PDF形式でダウンロード (221K) -
原稿種別: Notes
2021 年 37 巻 12 号 p. 1843-1846
発行日: 2021/12/10
公開日: 2021/12/10
[早期公開] 公開日: 2021/07/23PDF形式でダウンロード (236K)
Announcements
-
原稿種別: Announcements
2021 年 37 巻 12 号 p. 1847
発行日: 2021/12/10
公開日: 2021/12/10
PDF形式でダウンロード (194K)
- |<
- <
- 1
- >
- >|