- Issue 3 Pages 145-
- Issue 2 Pages 55-
- Issue 1 Pages 1-
- |<
- <
- 1
- >
- >|
-
 Takehiko OGAWA, Takafumi MATSUMURA, Tatsuma YAO, Hiroshi KIMURA, Kiyos ...Article type: Review
Takehiko OGAWA, Takafumi MATSUMURA, Tatsuma YAO, Hiroshi KIMURA, Kiyos ...Article type: Review
2024 Volume 70 Issue 1 Pages 1-9
Published: 2024
Released on J-STAGE: February 19, 2024
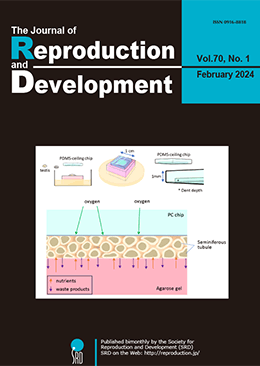
Advance online publication: December 23, 2023JOURNAL OPEN ACCESSIncorporation of bovine serum-derived albumin formulation (AlbuMAX) into a basic culture medium, MEMα, enables the completion of in vitro spermatogenesis through testicular tissue culture in mice. However, this medium was not effective in other animals. Therefore, we sought an alternative approach for in vitro spermatogenesis using a synthetic medium without AlbuMAX and aimed to identify its essential components. In addition to factors known to be important for spermatogenesis, such as retinoic acid and reproductive hormones, we found that antioxidants (vitamin E, vitamin C, and glutathione) and lysophospholipids are vital for in vitro spermatogenesis. Moreover, based on our experience with microfluidic devices (MFD), we developed an alternative approach, the PDMS-ceiling method (PC method), which involves simply covering the tissue with a flat chip made of PDMS, a silicone resin material used in MFD. The PC method, while straightforward, integrates the advantages of MFD, enabling improved and uniform oxygen and nutrient supply via tissue flattening. Furthermore, our studies underscored the significance of lowering the oxygen concentration to 10–15%. Using an integrated cultivation method based on these findings, we successfully achieved in vitro spermatogenesis in rats, which has been a long-standing challenge. Further improvements in culture conditions would pave the way for spermatogenesis completion in diverse animal species.
 View full abstractEditor's pick
View full abstractEditor's pickCover Story:
For the past century, achieving in vitro spermatogenesis has remained a difficult challenge for researchers. In 2011, Ogawa et al. successfully demonstrated in vitro spermatogenesis in mice using an organ culture method. However, extending this method to other species posed challenges for over a decade. In 2023, Ogawa’s team achieved in vitro spermatogenesis in rats by incorporating several critical modifications to enhance their original technique. This review presents a detailed analysis by Ogawa et al. comparing their method with natural in vivo conditions and other synthetic alternatives (Ogawa et al. Improvements in in vitro spermatogenesis: oxygen concentration, antioxidants, tissue-form design, and space control, pp. 1–9). They systematically explore the merits, limitations, and inherent constraints of the organ culture approach, delving into the specifics of medium composition, the principles of the gas-liquid interphase method, use of microfluidic devices, and innovation of the PDMS-ceiling method. Highlighting the challenges faced, including regulating oxygen concentration, managing tissue formation, and regulating culture space-control. The insights and novel concepts shared in this review are particularly valuable for those involved in culture or related disciplines, providing innovative content, and encouraging further exploration in this field.Download PDF (3115K)
-
Chihiro EMORI, Mayo KODANI, Ferheen ABBASI, Masashi MORI, Masahito IKA ...Article type: Original Article
2024 Volume 70 Issue 1 Pages 10-17
Published: 2024
Released on J-STAGE: February 19, 2024
Advance online publication: December 07, 2023JOURNAL OPEN ACCESS
Supplementary materialPoly(A)-binding proteins (PABPs) play roles in mRNA maturation, translational activity, and decay. The functions of PABPs, especially PABPN1 and PABPC1, in somatic cells have been well-studied. However, little is known about the roles of PABPs in oocytes because of the unique mechanisms of mRNA metabolism in oocytes. This study focused on PABPN1L and generated Pabpn1l knockout (KO) mice using the CRISPR/Cas9 system. After mating tests, we found that Pabpn1l KO females were infertile due to the failure of the embryos to develop to the 4-cell stage. RNA-seq analysis revealed aberrant mRNA persistence in Pabpn1l KO-MII oocytes, which indicates impaired mRNA degradation during the germinal vesicle (GV) to MII transition. We also revealed that the exogenous expression of Pabpn1l mRNA in KO-GV oocytes recovered defects of embryonic development. PABPN1L is partly indispensable for female fertility in mice, owing to its necessity for embryonic development, which is supported by mRNA degradation during GV to MII maturation.
 View full abstractDownload PDF (2027K)
View full abstractDownload PDF (2027K) -
Ryutaro AIZAWA, Megumi IBAYASHI, Junichiro MITSUI, Satoshi TSUKAMOTOArticle type: Original Article
2024 Volume 70 Issue 1 Pages 18-24
Published: 2024
Released on J-STAGE: February 19, 2024
Advance online publication: December 17, 2023JOURNAL OPEN ACCESSCommunication between oocytes and the surrounding granulosa cells during follicular development is essential for complete oocyte growth. Oocytes contain lipid droplets (LDs), organelles assembled in the endoplasmic reticulum (ER) that store neutral lipids, including triglycerides and cholesterol esters. Although the LD content varies among animals, LDs stored in oocytes have been shown to play an important role in oocyte maturation and preimplantation embryonic development. However, knowledge is lacking regarding how and when LDs are initially produced in developing oocytes within follicles. In the present study, we found that LDs appeared in mouse oocytes in a specific phase during follicular development. The emergence of LDs in intrafollicular oocytes was induced within a similar time window in vitro and in vivo. Fluorescence imaging and electron microscopy revealed that LDs emerging in oocytes during the early stages of follicular growth were in close proximity to the ER. Furthermore, fatty-acid-tracking experiments have revealed that exogenous fatty acids are rapidly incorporated into oocytes, and their uptake is regulated by the interaction between oocytes and granulosa cells, likely in part through transzonal projections. In summary, our results suggest that LD synthesis observed in growing oocytes is spatiotemporally regulated and that oocyte–granulosa cell contact may be involved in LD biosynthesis during follicular development.
 View full abstractDownload PDF (2770K)
View full abstractDownload PDF (2770K) -
Kaede ODA, Maya YOSHIDA, Abdul Razaq IRSHAD, Tomomi KANAZAWA, Toru TAK ...Article type: Original Article
2024 Volume 70 Issue 1 Pages 25-29
Published: 2024
Released on J-STAGE: February 19, 2024
Advance online publication: December 29, 2023JOURNAL OPEN ACCESSThe aim of the present study was to develop a semi-quantitative urine pregnancy test for mares based on the Cuboni reaction and to verify the reliability of this test. The urine specimens were hydrolyzed by heating in the presence of hydrochloric acid. The resulting free estrogens were extracted from the urine matrix using toluene. Sulfuric acid was added to the toluene extract and the mixture was heated again. The lower layer in the test tube containing sulfuric acid was used for fluorescence measurements with excitation at 355 nm and measurement at 535 nm. The fluorometric Cuboni test revealed that the fluorescence counts in urine samples collected after the second trimester of gestation were significantly higher than those obtained from barren mares. The levels of estrogens, including equilin, estrone and estardiol-17β exhibited a dose-dependent increase in fluorescence counts, whereas other steroids, such as progesterone, testosterone, and cortisol, did not affect fluorescence. Heat treatment of urine samples with hydrochloric acid significantly increased the fluorescence counts in those collected after the second trimester of gestation compared to non-pregnant samples, implying the presence of large amounts of conjugated estrogens in pregnant mare urine. Fluorescence counts in urine samples obtained during pregnancy showed a positive relationship with estrone concentrations as measured by enzyme immunoassay. The results of the present study showed that the fluorometric Cuboni test facilitates urine fluorescence counts depending on the urinary estrogen content and is capable of discriminating between pregnancy and non-pregnancy states beyond the second trimester of gestation in mares.
View full abstractDownload PDF (847K) -
Daisuke FUNAKOSHI, Hidetoshi SHIOTANI, Makoto SEKIArticle type: Original Article
2024 Volume 70 Issue 1 Pages 30-34
Published: 2024
Released on J-STAGE: February 19, 2024
Advance online publication: December 30, 2023JOURNAL OPEN ACCESSThis study investigated the effects of timed artificial insemination (TAI) and equine chorionic gonadotropin (eCG) administration on lactating dairy cows under heat-stress conditions (average temperature-humidity index: 80). Timed artificial insemination was performed on the cows with (n = 57) or without (control, n = 41) supplementation with 500 IU of eCG at the day of PGF2α treatment using the CIDR-Ovsynch protocol. GnRH was administered, and a progesterone device (CIDR) was inserted on Day −10 of the treatment protocol. The CIDR was removed on Day −3, and the cows were treated with PGF2α. Two days later, a 2nd GnRH injection was administered. Subsequently, AI was performed on Day 0 (16–20 h after the 2nd GnRH injection), and pregnancy was diagnosed on Days 32 and 60. Plasma progesterone (P4) concentrations were measured after AI. Results showed that the eCG group had a higher pregnancy per AI (P/AI) than the control group (43.9 vs. 12.2%, P = 0.002), which was also accompanied by elevated P4 levels. Four cows in the eCG group had multiple calves, representing 7.0 and 16.0% of the group and pregnant cows, respectively. In conclusion, 500 IU of eCG combined with CIDR-Ovsynch in lactating dairy cows under severe heat stress conditions successfully improved fertility. However, the protocol may have a slight risk of multiple births.
 View full abstractDownload PDF (889K)
View full abstractDownload PDF (889K) -
Fernando LÓPEZ-GATIUSArticle type: Original Article
2024 Volume 70 Issue 1 Pages 35-41
Published: 2024
Released on J-STAGE: February 19, 2024
Advance online publication: December 29, 2023JOURNAL OPEN ACCESSThis study sought to examine the impact of negative photoperiod on the incidence of multiple ovulations and pregnancies in dairy cattle. The study population consisted of 5,373 pregnant cows in their third or greater lactation that experienced their first post-partum pregnancy after spontaneous estrus. The positive photoperiod (increasing day-length) extends from December 22 to June 21, whereas the negative photoperiod (decreasing day-length) extends from June 22 to December 21. The odds ratios (ORs) for multiple ovulations and pregnancies in cows that became pregnant during the negative photoperiod and the remaining cows that became pregnant during the positive photoperiod were 1.4 and 1.3 (P < 0.0001), respectively. The ORs for cows that became pregnant ≥ 90 days in milk and the remaining cows that became pregnant < 90 days in milk were 4.3 and 4.1 (P < 0.0001), respectively. No significant differences were detected in the monthly rates of multiple ovulations or pregnancies during positive and negative photoperiods. Thus, the present study demonstrates that the ovarian function in cows is related to changes in day-length, with decreasing day-length being associated with greater multiple ovulation and pregnancy rates. The present study also shows that positive and negative photoperiods exhibit different trends. The results of this study are consistent with a growing body of work demonstrating the effects of photoperiod patterns on the reproductive physiology of cows, with clear implications for twin pregnancy prevention.
 View full abstractDownload PDF (1102K)
View full abstractDownload PDF (1102K) -
Norikazu MIYASHITA, Satoshi AKAGI, Tamas SOMFAI, Yuji HIRAOArticle type: Original Article
2024 Volume 70 Issue 1 Pages 42-48
Published: 2024
Released on J-STAGE: February 19, 2024
Advance online publication: January 19, 2024JOURNAL OPEN ACCESS
Supplementary materialEmbryonic transfer of bovine blastocysts produced using in vitro fertilization (IVF) is widely used, although the challenge of compromised conception rates remains. Using bovine oviduct epithelial cells (BOEC) to improve embryo culture conditions has attracted attention, particularly since the recent discovery of extracellular vesicles from BOEC. The selection of embryos for transfer has also been the subject of various studies, and a set of evaluation criteria to predict pregnancy success has been suggested, in which the embryos are judged by their kinetics and morphology at the early stages. In the present study, we established a spontaneously immortalized BOEC line (SI-BOEC) and examined the effects of conditioned medium on IVF embryos, focusing on the results of the recommended criteria. A modified KSOM (mKSOM) was used to prepare conditioned media. Presumptive zygotes were cultured in mKSOM (control), SI-BOEC-conditioned medium, mKSOM supplemented with sediment (pellet) collected after the ultracentrifugation of the conditioned medium (mKSOM/sediment), and the supernatant. A significantly higher percentage of embryos satisfied the recommended criteria when grown in the conditioned medium than in the mKSOM. A higher proportion of embryos developed into blastocysts after achieving the four criteria. A similar tendency was observed when grown in mKSOM/sediment compared to mKSOM; however, this was not observed in the supernatant. Vesicles with a size similar to that of exosomes were observed in the sediment. In conclusion, the culture medium conditioned by SI-BOEC promoted the production of bovine blastocysts that satisfied the four evaluation criteria recommended for embryo selection.
 View full abstractDownload PDF (1529K)
View full abstractDownload PDF (1529K)
-
Riho OZAKI, Seiji INOUE, Yuki YOROZUI, Rei ICHIKAWA, Naoki YAMADA, Sei ...Article type: Technology Report
2024 Volume 70 Issue 1 Pages 49-54
Published: 2024
Released on J-STAGE: February 19, 2024
Advance online publication: November 23, 2023JOURNAL OPEN ACCESS
Supplementary materialPre-ovulatory follicles are cooler than the neighboring reproductive organs in cows. Thus, measuring the temperature of reproductive organs could be a useful method for predicting estrus and ovulation in cows, and the establishment of a non-invasive technique is required. In this study, we used infrared thermography (IRT) to measure ocular surface temperature as a potential surrogate for reproductive organ temperature. Five Japanese Black cows with synchronized estrus were subjected to temperature measurements in five regions of the ocular surface, including the nasal conjunctiva, nasal limbus, center cornea, temporal limbus, and temporal conjunctiva, twice a day (0800 h and 1600 h) during the experimental period. The temperatures in the five regions significantly declined in cows from estrus to ovulation. To the best of our knowledge, this study is the first to use IRT to show a temperature decrease in the ocular surface along with estrus to ovulation in Japanese Black cows.
 View full abstractDownload PDF (3299K)
View full abstractDownload PDF (3299K)
- |<
- <
- 1
- >
- >|