
- |<
- <
- 1
- >
- >|
-
Minoru MIZUHATA, Mikito UEDA2023 年 91 巻 10 号 p. 100001
発行日: 2023/10/31
公開日: 2023/10/31
[早期公開] 公開日: 2023/10/20ジャーナル オープンアクセス HTMLSix years have passed since 2018, when Electrochemistry became a fully electronic journal after derivation off Denki Kagaku, and after that, becoming a gold open access journal in 2020, being registered in Directory of Open Access Journals (DOAJ) in 2021, and having all articles published in English in 2023. The authors would like to describe the policies and interests for the relaunch of this journal. Additionally, this issue will also include Comprehensive Papers and Review by the winners of the ECSJ awards in 2023 and before. The rationale for these awards will also be explained.
 抄録全体を表示PDF形式でダウンロード (2115K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (2115K) HTML形式で全画面表示
-
Susumu KUWABATA2023 年 91 巻 10 号 p. 101001
発行日: 2023/10/20
公開日: 2023/10/20
[早期公開] 公開日: 2023/09/27ジャーナル オープンアクセス HTMLOn June 25, 2005, the author visited one of his colleagues to discuss collaborative research on the semiconductor nanoparticles. During lunch, they talked about room temperature ionic liquid (RTIL), which was becoming more popular as the third liquid in several chemistry fields at that time. Since the author was particularly curious in the fact that RTIL does not evaporate even in a vacuum, the two discussed whether the RTIL research could be done in combination with precision instruments that require vacuum conditions. While pointing out the problems and talking about how to solve them, they enjoyed a pipe dream discussion of being able to perform wet chemical reactions using precision equipments under the assumption that the concerns could be solved. After returning to the author’s laboratory, he conducted the easiest experiments for solving the above-mentioned problems. Surprisingly, however, the author found that charging of the RTIL, which was the biggest concern, did not occur at all. This finding greatly inspired him and the colleague to start the research on handling RTIL with precision instruments. Soon after joint research was started with some research groups, sobering discoveries were made. Based on these findings, RTIL was introduced to various vacuum precision instruments to develop precision analysis methods including some in-situ analyses and wet nanomaterial production methods under vacuum (see abstract graphic). Among these, this paper introduces several techniques related to electrochemistry, that are electron microscope observations of soft materials and electrochemical reactions, in situ elemental analysis in electrochemical reaction, synthesis of metal nanoparticles used for electrocatalyst, and creation of 3D figures, including how the ideas for their development came to mind.
 抄録全体を表示PDF形式でダウンロード (8425K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (8425K) HTML形式で全画面表示
-
Akitoshi HAYASHI2023 年 91 巻 10 号 p. 101002
発行日: 2023/10/31
公開日: 2023/10/31
[早期公開] 公開日: 2023/10/07ジャーナル オープンアクセス HTMLAll-solid-state sodium batteries are promising rechargeable batteries owing to their safety, cost effectiveness, long cycle life, and high gravimetric energy density. To realize these batteries, a key material is a sodium–ion-conducting solid electrolyte with high Na+ ion conductivity, appropriate mechanical properties, and moderate chemical and electrochemical stability. Sulfide electrolytes are currently the best choice for balancing these requirements. Na3PS4 and Na3BS3 glass-based electrolytes and W-doped Na3SbS4 crystalline electrolytes were developed for application in all-solid-state Na/S batteries with high capacities and long cycle lives.
 抄録全体を表示PDF形式でダウンロード (3279K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (3279K) HTML形式で全画面表示 -
Soichiro YOSHIMOTO2023 年 91 巻 10 号 p. 101003
発行日: 2023/10/05
公開日: 2023/10/05
[早期公開] 公開日: 2023/09/13ジャーナル オープンアクセス HTMLThis study focuses on electrochemical potential control and fabrication methods for various metal complexes and polycyclic aromatic hydrocarbons at solid–liquid interfaces, particularly using electrochemical scanning tunneling microscopy (EC-STM) and atomic force microscopy (AFM). The in situ observation of molecular assemblies and understanding of the phase transition dynamics and ligand exchange reactions at the molecular and/or submolecular levels provide information on functional molecular design and surface engineering. In addition, ionic liquids electrochemistry is summarized from the viewpoint of single-crystal electrochemistry.
 抄録全体を表示PDF形式でダウンロード (9673K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (9673K) HTML形式で全画面表示
-
Yu KATAYAMA2023 年 91 巻 10 号 p. 101004
発行日: 2023/10/05
公開日: 2023/10/05
[早期公開] 公開日: 2023/07/22ジャーナル オープンアクセス HTMLThe quest to design active and stable electrochemical interface hinges on unequivocally identifying the active site(s) and reaction mechanism under realistic operating conditions. This comprehensive paper summarizes the most recent understanding of the electrochemical interface during energy conversion and storage reactions probed by operando surface-enhanced infrared spectroscopy (SEIRAS), operando attenuated total reflection infrared spectroscopy (ATR-IR), and operando synchrotron surface X-ray scattering, coupled with density functional theory (DFT) calculations. The paper also demonstrates that the holistic information about the complex interaction in the electrochemical interface can alter the reaction energetics/kinetics in a way incapable of electrode-centered design strategy. This work shed light on the significance of advanced operando techniques to accelerate the design of electrochemical interfaces by providing additional knobs to tune the reaction energetics/kinetics, leading further improvements in electrocatalytic activity for energy conversion and storage reactions.
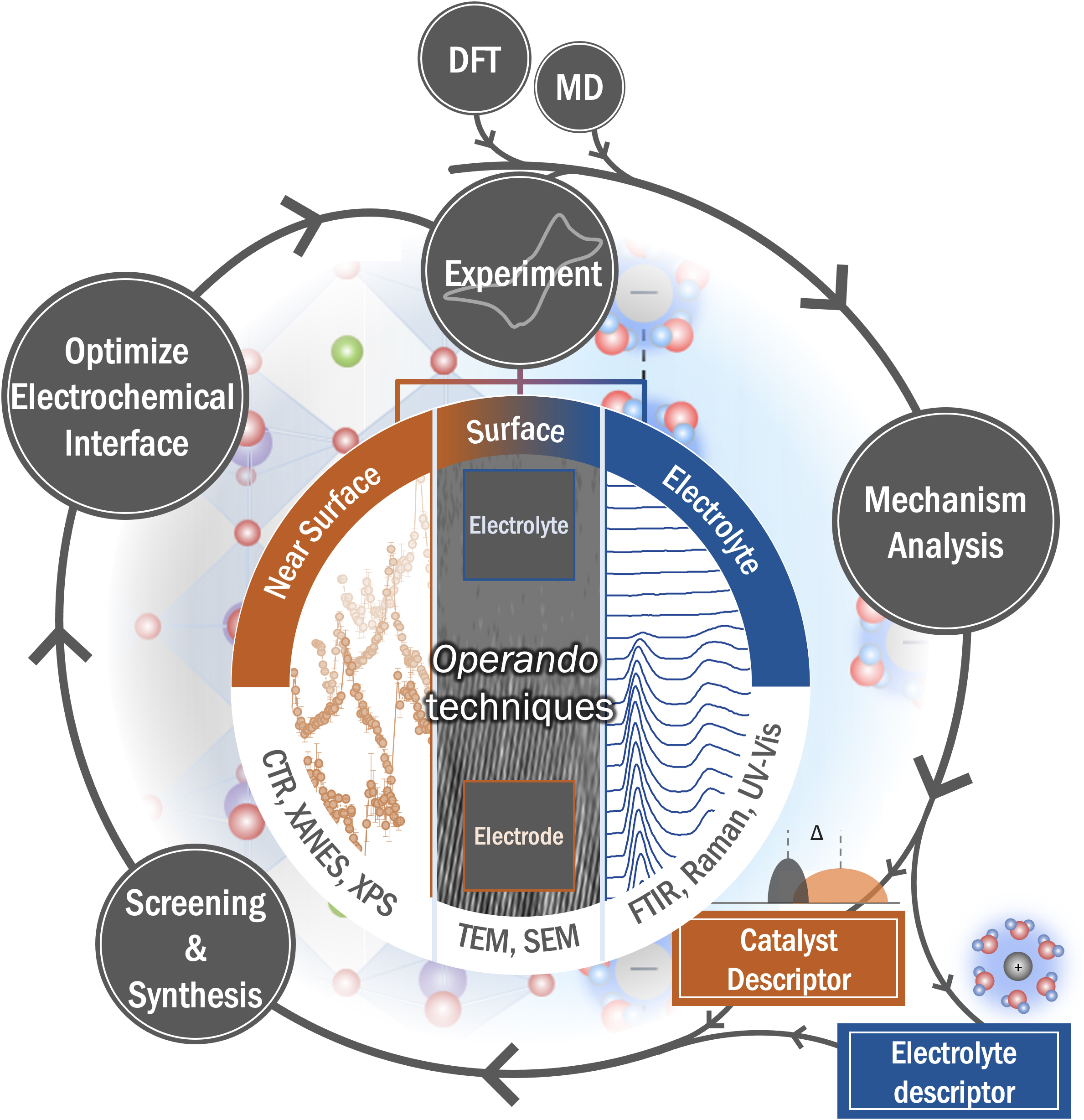 抄録全体を表示PDF形式でダウンロード (7971K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (7971K) HTML形式で全画面表示 -
Yuta KIMURA2023 年 91 巻 10 号 p. 101005
発行日: 2023/10/05
公開日: 2023/10/05
[早期公開] 公開日: 2023/09/05ジャーナル オープンアクセス HTMLThe design of high-performance electrochemical devices requires a profound understanding of the mechanisms governing the electrochemical reactions within the device, ranging from microscopic one involving atoms and molecules to macroscopic one observed in cells/stacks of practical devices. While extensive efforts have been made to understand the microscopic and macroscopic electrochemical phenomena in these devices, understanding the interplay between micro- and macro-scale electrochemical phenomena has not necessarily been sufficient. One reason is that what connects the macroscopic and microscopic electrochemical phenomena is a complex electrochemical phenomenon involving the collective behavior of a large population of particles within an electrode under the influence of external fields such as stress. This comprehensive paper presents the research conducted by the authors to develop novel techniques for understanding the intricate electrochemical phenomena that bridge the micro and macro scales. The first part of this paper presents our work on the development of a technique to perform three-dimensional operando observation of heterogeneous electrochemical reactions occurring in a particle ensemble within solid state battery electrodes using computed-tomography with X-ray absorption near edge structure spectroscopy (CT-XANES). The latter part focuses on the development of a method to quantitatively evaluate stress-induced modulation of material properties of battery electrode materials (chemo-mechanical coupling phenomena).
 抄録全体を表示PDF形式でダウンロード (3343K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (3343K) HTML形式で全画面表示 -
Shoichi MATSUDA2023 年 91 巻 10 号 p. 101006
発行日: 2023/10/05
公開日: 2023/10/05
[早期公開] 公開日: 2023/07/20ジャーナル オープンアクセス HTMLThere is growing demand for the energy storage devices with superior energy density than that of conventional lithium-ion batteries. Lithium-air batteries (LABs) are promising candidates for next-generation rechargeable batteries due to their extremely high theoretical energy density. In recent several years, there are many research progress for LABs mainly in the field of academia. However, in most of the studies, the performance evaluation of LABs is performed under inappropriate technological parameters from the viewpoint of the high energy density cell design. As results, the cell-level energy density of LABs is lower than conventional lithium-ion batteries. For realizing the cell-level high energy density LABs, such as 500 Wh/kg class LABs, the cell should be operated under lean electrolyte and high areal capacity condition and the suitable electrode materials and electrolyte should be developed. This article over-views the recent research progress of LABs, from the viewpoint of practical cell design and material development. In addition, the perspective for future research direction for realizing LABs with practically high energy density and long cycle life is also described.
抄録全体を表示PDF形式でダウンロード (3832K) HTML形式で全画面表示
-
Lina YOSHIDA2023 年 91 巻 10 号 p. 101007
発行日: 2023/10/05
公開日: 2023/10/05
[早期公開] 公開日: 2023/09/15ジャーナル オープンアクセス HTMLSolution processing of thin films is a key energy-saving technology for sustainable development. Not only for its cost advantages, but it can achieve materials with unique structures and new functionalities through the combination of earth-abundant elements, especially by their hybridization with organic molecules. As the process relies on mild activation, such as low-temperature heating and irradiation with ultraviolet light having high photon energy, it is compatible with plastic film substrates. This compatibility makes it suitable for roll-to-roll mass production of flexible optoelectronic devices with large areas at a low cost. Herein, we present a review of our recent progress in the low-temperature solution processing of inorganic/organic hybrid thin films with various functionalities, including thin film encapsulation (TFE) to protect organic light-emitting diodes (OLEDs) from moisture for long-lasting operation, color-tunable photoluminescent nanocrystalline ZnO core-organic shell hybrids, and an energy-selective electron injection layer (EIL) for highly efficient inverted OLEDs. While general concept, methodology and challenges of solution processing are summarized, the future of the related technology is scoped for our sustainable development.
 抄録全体を表示PDF形式でダウンロード (5391K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (5391K) HTML形式で全画面表示 -
Mariko MATSUNAGA2023 年 91 巻 10 号 p. 101008
発行日: 2023/10/05
公開日: 2023/10/05
[早期公開] 公開日: 2023/08/10ジャーナル オープンアクセス HTMLThe influence of nano- and micro-structures on various physicochemical phenomena is a subject of interest to many researchers. Physicochemical reactions at solid/electrolyte interfaces, such as crystallization, metal complex formation, adsorption, and electrochemical reactions are influenced by surface modifications with organic thin films. In this study, we review and generalize the findings of our research on the application of organic thin films with or without three-dimensional structures to chiral sensors, interactive motions of nano/micro materials, and electrodes for electrochemical energy devices.
抄録全体を表示PDF形式でダウンロード (3716K) HTML形式で全画面表示
-
Ryoji KANNO2023 年 91 巻 10 号 p. 102001
発行日: 2023/10/05
公開日: 2023/10/05
[早期公開] 公開日: 2023/08/18ジャーナル オープンアクセス HTMLThe development of new materials leads to the invention of new devices. The exploitation of high ionic conductivity materials has facilitated the emergence of a new category of energy storage devices, including the all-solid-state battery. This paper reviews the history of the development of lithium solid electrolytes and their application in all-solid-state batteries. Particular focus is given to the development process of Li10GeP2S12, which surpasses the conductivity characteristics of liquid-electrolyte systems targeted by lithium-ion conductors, and its application to solid-state batteries is described. Furthermore, this review describes new science that will be born when batteries become solid-state.
抄録全体を表示PDF形式でダウンロード (3616K) HTML形式で全画面表示
-
Yoshitaka AOKI2023 年 91 巻 10 号 p. 102002
発行日: 2023/10/05
公開日: 2023/10/05
[早期公開] 公開日: 2023/03/25ジャーナル オープンアクセス HTMLIn this paper, we describe new strategies to reduce the resistances related to cathode reactions and interfacial proton transfer in protonic solid oxide fuel cells (H+-SOFCs) based on proton-conducting BaZrxCe0.8−xM0.2O3−δ (M = Y, Yb, Sc etc.) by means of material and cell-structure design changes. First, an extension of the effective cathode reaction areas by employing the H+/O2−/e− triple-conducting cathode is described. Cubic La0.7Sr0.3Mn1−xNixO3−δ (x = 0–0.3) can be hydrated under fuel cell conditions due to its large hydration enthalpy (∼100 kJ mol−1), whereas rhombohedral La0.7Sr0.3Mn1−xNixO3−δ does not exhibit hydration capabilities; hence, the porous anode cermet support fuel cells (PAFCs), which use the former as a cathode, possess significantly smaller cathode polarization resistances than the PAFCs that use the latter. Second, we describe a new thermodynamic mechanism for reducing the electrolyte and cathode reaction resistances in a hydrogen-permeable metal-support fuel cell (HMFC), which involves the blocking of the oxide ion minor conduction in the BaZrxCe0.8−xM0.2O3−δ electrolyte at metal/oxide heterointerfaces. The BaZrxCe0.8−xM0.2O3−δ membrane of HMFCs is forced to gain extra protons to compensate for the charge from the oxide ions accumulating near the heterointerfaces via blocking, resulting in extremely high proton conductivity. This promotes significant interfacial proton diffusion for cathode reactions.
 抄録全体を表示PDF形式でダウンロード (5366K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (5366K) HTML形式で全画面表示
-
Atsushi SAKUDA2023 年 91 巻 10 号 p. 102003
発行日: 2023/10/05
公開日: 2023/10/05
[早期公開] 公開日: 2023/05/10ジャーナル オープンアクセス HTMLLithium/transition-metal polysulfide batteries are a promissing candidate for next-generation batteries with high energy densities. Transition metal polysulfides and lithium- or sodium-containing transition metal sulfides exhibit large reversible capacities based on multi-electron processes, owing to the redox reactions of S in addition to the transition metal. This comprehensive paper aims to address the idea, research, and development of transition metal polysulfide electrode active materials and summarizes the author’s views on the concept of transition metal polysulfide electrodes. Furthermore, the diversity of coordination structures and unique structural changes during charging and discharging will be discussed.
抄録全体を表示PDF形式でダウンロード (5587K) HTML形式で全画面表示
-
Kentaro YAMAMOTO2023 年 91 巻 10 号 p. 102004
発行日: 2023/10/05
公開日: 2023/10/05
[早期公開] 公開日: 2023/04/26ジャーナル オープンアクセス HTMLElectrode reactions in electrochemical devices often consist of charge transfer at electrode/electrolyte interface and charge compensation in electrode active material. Therefore, to design electrochemical devices with high electrochemical performance, it is important to understand electronic structures of the electrode/electrolyte interface and electrode bulk during electrochemical reactions, and to design electrode materials to control them. In this paper, certain phenomena at the electrode/electrolyte interface and electrode bulk in lithium-ion batteries are clarified by using synchrotron radiation X-ray analyses. The information obtained from those X-ray analyses are applied to control the structure of the electrode/electrolyte interface and electrode bulk. Moreover, new cathode materials for all-solid-state fluoride-ion with high power and cyclic performances have been developed, compared to simple metal/metal fluoride materials, by using the findings obtained from the synchrotron radiation X-ray analyses.
抄録全体を表示PDF形式でダウンロード (4262K) HTML形式で全画面表示
-
Navapat KROBKRONG, Taro UEMATSU, Tsukasa TORIMOTO, Susumu KUWABATA2023 年 91 巻 10 号 p. 107001
発行日: 2023/10/31
公開日: 2023/10/31
[早期公開] 公開日: 2023/09/27ジャーナル オープンアクセス HTML
J-STAGE DataSilver indium sulfide (AIS)/gallium sulfide (Ga–S) core/shell QDs exhibit a narrow band-edge photoluminescence (PL) in the yellow color region, and shifting the PL wavelength is crucial for optical applications. In this study, we attempt to redshift the band-edge PL by incorporating indium sulfide (In–S) shells, which have a smaller bandgap than Ga–S and are expected to broaden the exciton wavefunction. When coated with In–S shells instead of Ga–S shells, a redshift of the band-edge PL was attained. However, an increase in defective PL and a reduction in PL quantum yield occurred due to carrier trapping associated with the extended wavefunction. To address these issues, we coated the AIS/In–S cores/shell QDs with Ga–S shells using recently developed procedures, resulting in spectrally narrow PL in the red region. Interestingly, compositional and structural analyses revealed a decrease in the In ratio, which typically leads to blue shift. The observed redshift, reaching up to 40 nm, is discussed in relation to the formation of shells with graded composition, which provide a broader wavefunction in the excited state compared to discrete shells.
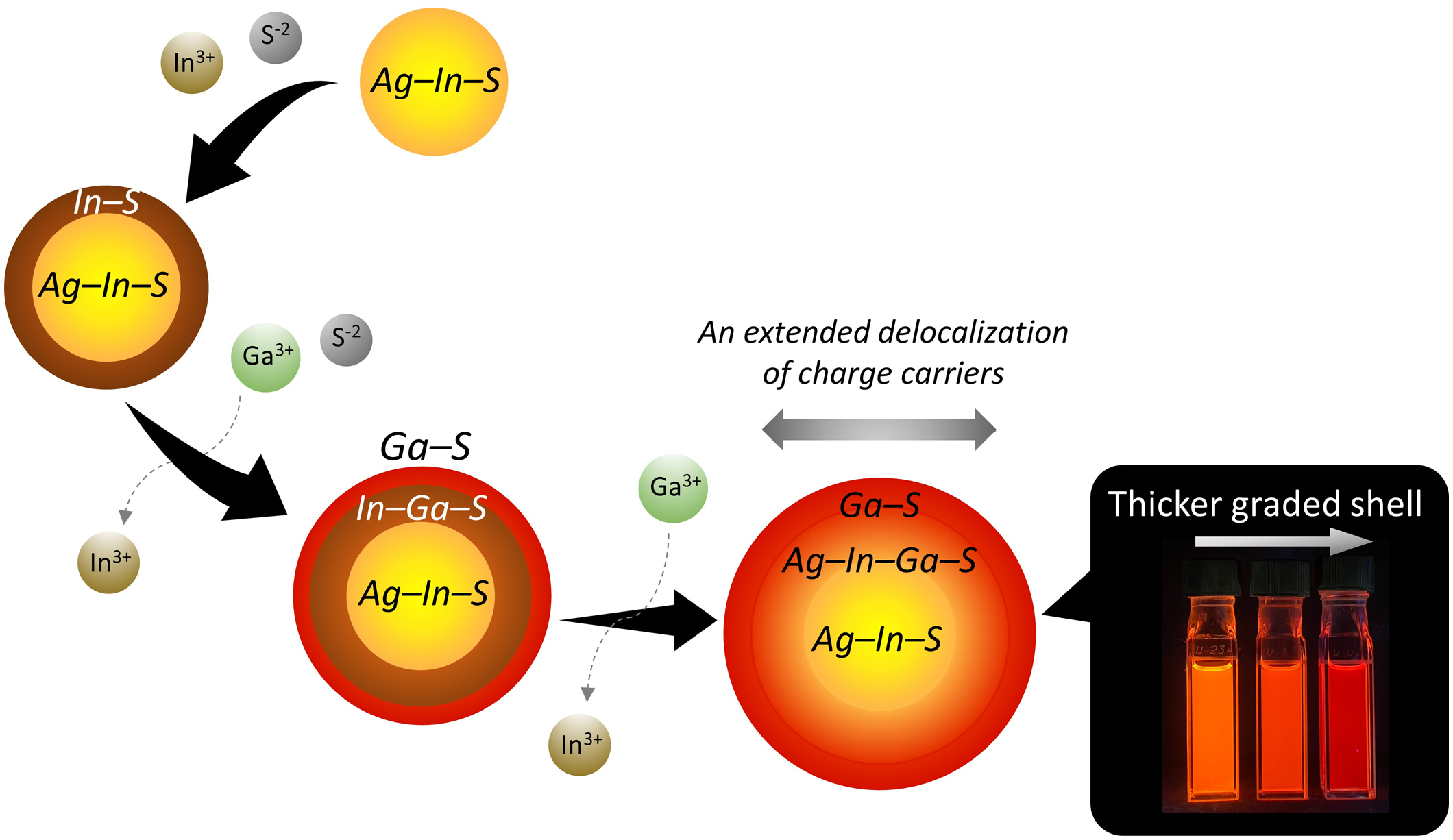 抄録全体を表示PDF形式でダウンロード (4366K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (4366K) HTML形式で全画面表示
- |<
- <
- 1
- >
- >|