
- |<
- <
- 1
- >
- >|
-
Takashi KAKIUCHI, Ryunosuke TANIGO, Atsushi TANI, Takeshi YAMAZAKI, Ko ...2023 年 91 巻 1 号 p. 013001
発行日: 2023/01/01
公開日: 2023/01/01
[早期公開] 公開日: 2022/11/25ジャーナル オープンアクセス HTML
J-STAGE DataA reference electrode equipped with ionic liquid salt bridge consisting of tributyl(2-methoxyethyl)phosphonium bis(pentafluoroethanesulfonyl)amide has been employed for potentiometric precipitation titration of chloride with silver ions in water at 25 °C. A model for the titration curve was regressed to experimental curves, taking into account the change in the activity coefficients of relevant ionic species in the course of the titration, to obtain the least square estimates of two adjustable parameters in the model, the solubility product (Ksp) and the analyte concentration. The least-square estimate of Ksp, (1.840 ± 0.060) × 10−10, i.e., pKsp = 9.736 ± 0.014, is in good agreement with literature data, but with higher precision.
抄録全体を表示PDF形式でダウンロード (728K) HTML形式で全画面表示
-
Tomoki HIRONO, Hiroyuki USUI, Yasuhiro DOMI, Wataru IRIE, Takahiro NIS ...2023 年 91 巻 1 号 p. 017001
発行日: 2023/01/01
公開日: 2023/01/01
[早期公開] 公開日: 2022/12/13ジャーナル オープンアクセス HTML
J-STAGE DataOur group has investigated the properties of lithium-ion battery anodes fabricated using Sn/SiOx (SiOx is a mixed phase of Si clusters and amorphous SiO2 matrix). The addition of Sn improves the conductivity of the SiO2 matrix, while the formation of the Li+-conductive Li2Si2O5 phase in the SiO2 matrix improves Si utilization. The charge–discharge cycle life is also extended. In this study, Sn-doped SiOx has been used to fabricate the anode of a sodium-ion battery, and its charge–discharge properties are evaluated. The addition of 3 wt% Sn to SiOx improves the cycle property, as revealed by charge–discharge tests. X-ray diffraction analysis confirmed that the Na+-conductive Na2Si2O5 phase is formed during the charging and discharging processes. These results indicate that adding Sn improves the electronic conductivity of SiO2, and Na2Si2O5 facilitated the movement of Na in the SiO2 matrix. Thus, the utilization of Si is enhanced, and a high discharge capacity is achieved.
抄録全体を表示PDF形式でダウンロード (3672K) HTML形式で全画面表示 -
Alisha YADAV, Hironobu KOBAYASHI, Takayuki YAMAMOTO, Toshiyuki NOHIRA2023 年 91 巻 1 号 p. 017002
発行日: 2023/01/24
公開日: 2023/01/24
[早期公開] 公開日: 2022/12/15ジャーナル オープンアクセス HTMLElectrochemical intercalation behaviors of alkali metals into graphite have been vigorously studied in academic and industrial fields, whereas their mechanisms are still unclarified. In this study, we report a novel rubidium-ion battery utilizing an ionic liquid electrolyte and natural graphite as the negative electrode material operating at room temperature. The electrochemical phase evolution behavior of rubidium–graphite intercalation compounds was elucidated using X-ray diffraction. The graphite negative electrode exhibited an initial discharge capacity of 216 mAh g−1 at 0.05 C rate, and retained its capacities of around 180 mAh g−1 even at 0.5 C rate for 50 cycles.
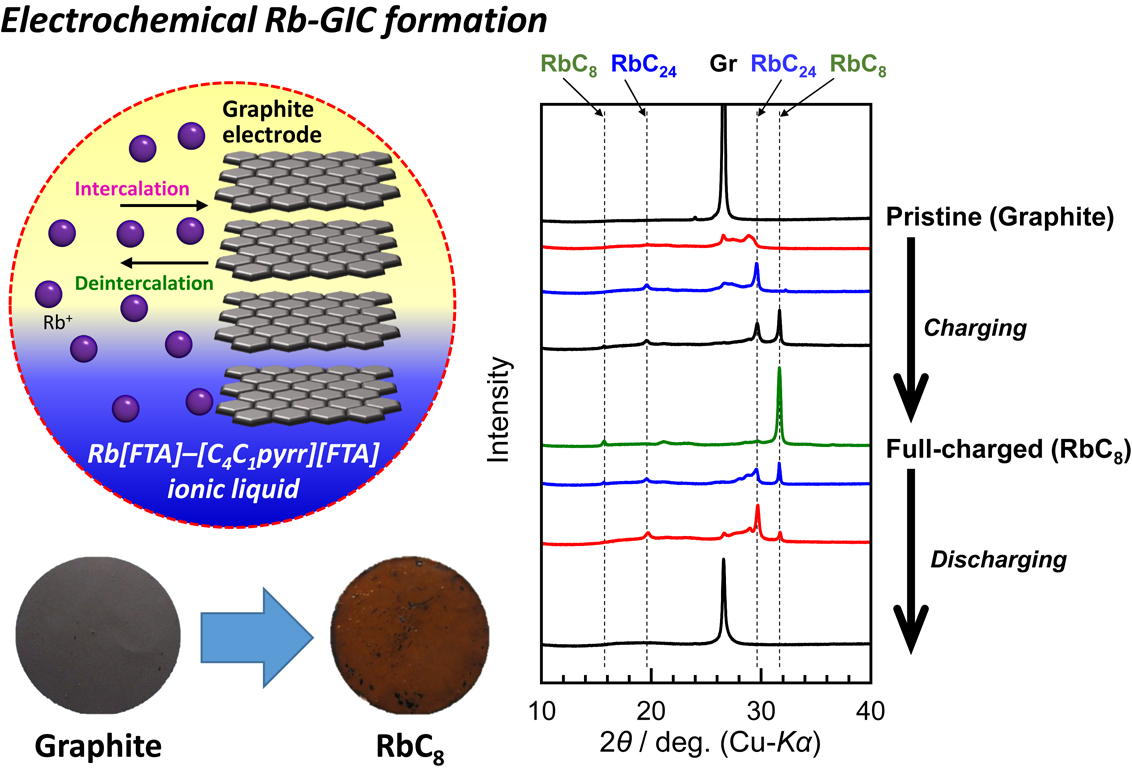 抄録全体を表示PDF形式でダウンロード (1466K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (1466K) HTML形式で全画面表示 -
Shubham KAUSHIK, Kazuhiko MATSUMOTO, Rika HAGIWARA2023 年 91 巻 1 号 p. 017003
発行日: 2023/01/25
公開日: 2023/01/25
[早期公開] 公開日: 2022/12/23ジャーナル オープンアクセス HTMLHard carbon (HC) is receiving widespread attention as a negative electrode material for sodium secondary batteries. In this study, spherical HC with three different diameters (1 µm, 1.5 µm, and 5 µm) are investigated using ionic liquid (IL) and organic electrolytes. The HC with a diameter of 5 µm demonstrated the best performance with IL at 90 °C (capacities of 327 mAh g−1 at 20 mA g−1 and 77 mAh g−1 at 2000 mA g−1 and initial Coulombic efficiency of 78.9 %). The HCs showed stable cycling in ILs as compared to the organic electrolytes in the first few cycles.
抄録全体を表示PDF形式でダウンロード (1409K) HTML形式で全画面表示
- |<
- <
- 1
- >
- >|