
- |<
- <
- 1
- >
- >|
-
Mitsuhiro KISHIMI, Masahito MORITA, Tatsumi HIRANO, Hisao KIUCHI, Kent ...2024 年 92 巻 5 号 p. 057001
発行日: 2024/05/02
公開日: 2024/05/02
[早期公開] 公開日: 2024/04/04ジャーナル オープンアクセス HTML
J-STAGE DataThe effects of the current rate used during the first charging (pre-charging: so-called “formation”) on the cathodic deposition of metallic zinc (Zn) were analyzed for the high capacity (thick) zinc oxide (ZnO) electrode in rechargeable Zn-based batteries. Pre-charging at a lower current rate (1.875 mA cm−2) enabled greater electrode performances for the subsequent charge-discharge cycles. The Zn deposition profiles were investigated by conventional postmortem X-ray diffraction (XRD) and energy-dispersive X-ray spectroscopy using a scanning electron microscope, as well as in situ synchrotron XRD and ex situ synchrotron X-ray computed tomography. The results revealed significant differences in the deposition profiles of the metallic Zn depending on the current rates used during pre-charging. The higher rate (18.75 mA cm−2) resulted in an inhomogeneous deposition of Zn, whereas the lower rate yielded finer Zn particles dispersed homogeneously throughout the thick ZnO electrode. These morphological and spatial variations in the Zn deposition during pre-charging affected the subsequent cycling behavior of the thick ZnO electrode.
抄録全体を表示PDF形式でダウンロード (4436K) HTML形式で全画面表示 -
Peikun ZOU, Chunyu XIANG, Xuyang LI, Nayun ZHOU, Binbin FAN, Limin WAN ...2024 年 92 巻 5 号 p. 057002
発行日: 2024/05/03
公開日: 2024/05/03
[早期公開] 公開日: 2024/04/17ジャーナル オープンアクセス HTML
J-STAGE DataA series of quaternary ammonium salts base on quinacridone aromatic ring were synthesized using 2,9-quinacridone as the parent material, and their leveling performance were evaluated using constant-current chronoamperometry addition curves. Additionally, the adsorption abilities of the leveler molecules on copper surfaces were investigated through quantum chemical calculations and molecular dynamics simulations, and their specific adsorption sites on the copper surface were investigated by XPS, then the effect of concentration on the leveling performance was probed by through-hole plating, and the copper surface was characterized by XRD and SEM. Among four quinacridone aryl quaternary ammonium salts, DCQA-C8-MI exhibits optimal leveling effects, serving as an excellent leveler with outstanding performance. This study expands the application scope of quinacridone aromatic heterocyclic quaternary ammonium salts and offers insights for exploring efficient organic additives for copper electrodeposition.
 抄録全体を表示PDF形式でダウンロード (7688K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (7688K) HTML形式で全画面表示 -
Hiroyasu NISHI, Taro TOJO, Tetsu TATSUMA2024 年 92 巻 5 号 p. 057003
発行日: 2024/05/09
公開日: 2024/05/09
[早期公開] 公開日: 2024/04/11ジャーナル オープンアクセス HTML
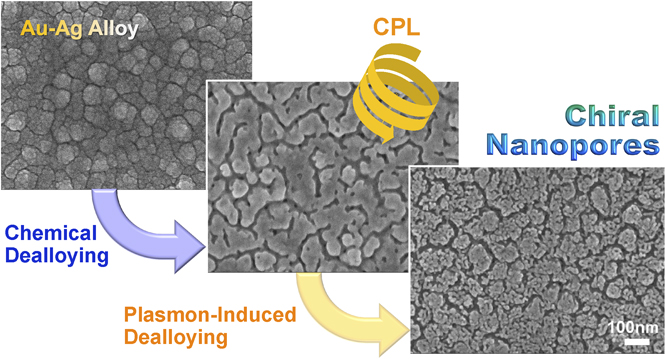
J-STAGE DataChiral plasmonic nanostructures are of significant interest because of their strong chirality compared to typical chiral molecules and their potential for various applications such as enantioselective sensors and metamaterials. Although chemical or photochemical fabrication methods for chiral nanostructures have attracted attention because of their cost-effectiveness and large-area applicability, most of the chemically synthesized chiral nanostructures are two-dimensional ensembles or arrays of individual chiral nanoparticles. In the present study, more three-dimensional, densely interconnected chiral plasmonic nanoporous structures are fabricated via plasmon-induced dealloying of Au-Ag alloy under circularly polarized light (CPL). CPL is used as a sole chiral source and irradiated to a chemically treated Au-Ag alloy film showing absorption due to localized surface plasmon resonance (LSPR). The resulting nanoporous structures exhibit chiroptical responses depending on the handedness CPL illuminated. The mechanism of chirality introduction is discussed on the basis of an electromagnetic simulation.
 抄録全体を表示PDF形式でダウンロード (3700K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (3700K) HTML形式で全画面表示 -
Liyuan YAO, Xihua WANG, Dongze LI, Xingai WANG, Haichang ZHANG, Ning W ...2024 年 92 巻 5 号 p. 057004
発行日: 2024/05/24
公開日: 2024/05/24
[早期公開] 公開日: 2024/04/25ジャーナル オープンアクセス HTML
J-STAGE DataDue to the poor thermal stability of the lithium hexafluorophosphate (LiPF6) electrolyte system, commercial lithium-ion batteries (LIBs) are difficult for normal operation at high temperatures above 55 °C. The limitation of the LiPF6 electrolyte severely limits the practical application of LIBs under extremely high temperatures conditions. Here, a high-concentration electrolyte based on lithium bis(fluorosufonyl)imide (LiFSI) as electrolyte salt and ethyl methyl carbonate (EMC) as solvent is proposed, which possesses superior electrochemical stability and thermal stability. The LiCoO2/graphite (Gr) pouch battery with the LiFSI high-concentration electrolyte (5.0 mol L−1 (M)) has been shown excellent cycling performance even at 100 °C, an impressive capacity retention of 87.7 % can be still maintained after 100 cycles at 1.0 C-rate. The superior high temperatures performance is mainly attributed to the unique solvated structure, along with the robust solid electrolyte interphase (SEI) rich in anions. This work presents an effective strategy for promoting the development of high-temperature lithium-ion batteries.
抄録全体を表示PDF形式でダウンロード (5208K) HTML形式で全画面表示 -
Hiroaki NISHIJIMA, Asato INOUE, Takashi HARADA, Kazuhide KAMIYA, Shuji ...2024 年 92 巻 5 号 p. 057005
発行日: 2024/05/31
公開日: 2024/05/31
[早期公開] 公開日: 2024/05/08ジャーナル オープンアクセス HTML
J-STAGE DataThe electrochemical reduction of carbon dioxide (CO2 electrolysis) is attracting much attention as a promising approach to address environmental challenges. Among the various products of CO2 electrolysis, formaldehyde (HCHO) stands out for its potential as a precursor for longer-chain compounds. However, reports on HCHO production are scarce, and comprehensive discussions are still lacking. A major obstacle to deeper analysis is the inherent instability of HCHO, which tends to undergo disproportionation reactions, transforming it into different substances. In this study, we propose a quantitative indicator to evaluate the progression level of the disproportionation reaction of HCHO. Utilizing this indicator, we explore how the progression of the disproportionation reaction is influenced by changes in the electrolyte’s pH and catalyst layer thickness in the electrochemical reduction of carbon monoxide (CO) – a key intermediate in CO2 electrolysis. Our results showed that the impact of the disproportionation reaction increases with higher pH values and greater thickness of the catalyst layer. Moreover, the insights obtained from CO electrolysis are effective in managing the generation of HCHO during CO2 electrolysis.
 抄録全体を表示PDF形式でダウンロード (1553K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (1553K) HTML形式で全画面表示
- |<
- <
- 1
- >
- >|