
- Issue 12 Pages 695-
- Issue 11 Pages 611-
- Issue 10 Pages 551-
- Issue 9 Pages 485-
- Issue 8 Pages 405-
- Issue 7 Pages 345-
- Issue 6 Pages 281-
- Issue 5 Pages 239-
- Issue 4 Pages 191-
- Issue 3 Pages 137-
- Issue 2 Pages 83-
- Issue 1 Pages 1-
- Issue Special_Issue P・・・
- Issue Supplement2 Pag・・・
- Issue Supplement1 Pag・・・
- |<
- <
- 1
- >
- >|
-
Saori Yabe, Takahito Nakagawa, Kazuyoshi Okumura, Tatsushi Shimokuni, ...Article type: ORIGINAL ARTICLE
2020Volume 53Issue 5 Pages 399-408
Published: May 01, 2020
Released on J-STAGE: May 30, 2020
JOURNAL FREE ACCESS FULL-TEXT HTMLPurpose: The aim of this study was to assess the diagnostic accuracy of preoperative images in detecting accessory hepatic ducts and frequency of bile duct injuries. Materials and Methods: A total of 482 patients were treated consecutively with cholecystectomy for gallbladder disease during a recent 10-year period. In this case, 36 patients (7.5%) were given a diagnosis of accessory hepatic ducts, and we reviewed their preoperative evaluation of hepatic ducts, operative procedure and the rate of hepatic duct injuries and compared them with normal anatomy groups. Results: Type I, II, III, IV, and V accessory hepatic ducts were detected in 2, 6, 26, 0, and 2 patients, respectively, based on the Hisatsugu classification, draining posterior segments in up to 80% of cases. The accuracy for detecting accessory hepatic ducts was 89.3% (25 out of 28 patients) for magnetic resonance cholangiograph (MRC) and 100% (15 patients) for drip infusion cholangiographic-CT (DIC-CT), respectively. In accessory hepatic duct groups, laparotomy was performed in 7 patients (19.4%), laparoscopy in 29 patients (80.6%), with conversion to laparotomy in 3 patients (10.3%). Laparotomy was performed more in accessory hepatic duct groups than normal anatomy groups, but there was no significant difference in the conversion rate to laparotomy. None of the patients with accessory hepatic ducts suffered bile duct injuries, postoperative obstructions of hepatic ducts or remnant gallbladder stones. Conclusion: A detailed evaluation of hepatic ducts on preoperative precise imaging studies was necessary to perform safe laparoscopic cholecystectomy, and surgical techniques such as intraoperative cholangiography could be considered in operations for accessory hepatic duct groups.
View full abstractDownload PDF (1434K) Full view HTML
-
Takeshi Suda, Sumito Hoshino, Yu Takagi, Kenichi Iwasaki, Yosuke Makuu ...Article type: CASE REPORT
2020Volume 53Issue 5 Pages 409-417
Published: May 01, 2020
Released on J-STAGE: May 30, 2020
JOURNAL FREE ACCESS FULL-TEXT HTMLCongenital factor VII deficiency is a rare disease. We performed gastrectomy safely in a gastric cancer case with congenital factor VII deficiency after administering recombinant activated factor VII (rFVIIa) preoperatively. The patient, who was a 58-year-old man, underwent examination to treat hepatitis B and was given a diagnosis of gastric cancer with congenital factor VII deficiency. Prothrombin time was prolonged to 18.0 seconds (control 12.7 seconds), and factor VII activity was reduced to 10%. Upper gastrointestinal endoscopy showed gastric cancer on the anterior wall of the gastric angle. We performed distal gastrectomy after intravenous injection of rFVIIa 1.2 mg (18 μg/kg) preoperatively. After distal gastrectomy, 1.2 mg of rFVIIa was injected intravenously for a total of 13 times by postoperative day 4, after measuring factor VII activity. Few cases of congenital factor VII deficiency have been reported in surgery, and distal gastrectomy was performed safely by measuring factor VII activity and administering rFVIIa.
View full abstractDownload PDF (1361K) Full view HTML -
Takahiro Yokose, Minoru Kitago, Yoshinobu Akiyama, Hiroki Hoshino, Yut ...Article type: CASE REPORT
2020Volume 53Issue 5 Pages 418-424
Published: May 01, 2020
Released on J-STAGE: May 30, 2020
JOURNAL FREE ACCESS FULL-TEXT HTMLOne potential risk of percutaneous transhepatic biliary drainage (PTBD) is tumor seeding along the catheter tract. A 72-year-old woman was referred to our hospital because of obstructive jaundice. Five weeks after inserting PTBD from the 7th right intercostal space, a pylorus-preserving pancreaticoduodenectomy was performed for the diagnosis of distal bile duct carcinoma. The patient was given a pathological diagnosis of stage I (T1 N0 M0) distal bile duct carcinoma. A tumor with rash on PTBD fistula was found and was removed 3 years after the first operation. Histological findings were compatible with bile duct carcinoma metastasis. An isolated lymph nodal mass of the right axilla was found by CT 4 years and 6 months after the first operation, and an excision biopsy was performed. Histological findings were compatible with bile duct carcinoma metastasis again. The patient underwent chemotherapy, but multiple lung metastases appeared 4 months later and she died 6 years after the first operation. We report on a case of PTBD fistula recurrence and right axillary lymph node recurrence after radical surgery for early distal bile duct carcinoma.
View full abstractDownload PDF (2016K) Full view HTML -
Chikanori Tsutsumi, Yoshihiro Miyasaka, Yasuhisa Mori, Kohei Nakata, T ...Article type: CASE REPORT
2020Volume 53Issue 5 Pages 425-434
Published: May 01, 2020
Released on J-STAGE: May 30, 2020
JOURNAL FREE ACCESS FULL-TEXT HTMLA 75-year-old man presented with upper abdominal pain. CT and MRI showed cystic lesions 27 mm in size with no malignant findings in the head of the pancreas. Under the diagnosis of intraductal papillary mucinous neoplasm (IPMN), the patient was followed-up with imaging studies. Six years after the initial presentation, a contrast CT scan showed an enhanced mural nodule 10 mm in size in the cyst, and the patient was referred to our hospital for further examination. CT and MRI revealed the cysts had shrunk compared to previous imaging findings. Pancreatic juice cytology showed no malignant cells. Based on detailed examination, a diagnosis of IPMN with high risk stigmata was made and laparoscopy-assisted pancreatoduodenectomy was performed. A histopathological examination revealed a cystic lesion consisting of IPMN with low-grade dysplasia, inflammatory infiltrates with lymphoplasmacytes corresponded to nodules in cysts, obliterative phlebitides, and storiform fibrosis. Immunohistochemical staining demonstrated that the infiltrating lymphoplasmacytes were positive for IgG4 and that IgG4+/IgG+ plasm cell ratio was over 40%. Finally, we diagnosed branch duct type IPMN associated with autoimmune pancreatitis type-1, mimicking an enhanced mural nodule. We encountered a rare case of IPMN associated with autoimmune pancreatitis. It can be suggested that serological and histological examinations, including blood IgG4 testing and ultrasound-guided fine-needle aspiration, should be considered in patients with IPMN that show rapid appearance of enhanced nodules and shrinkage of cysts.
View full abstractDownload PDF (2109K) Full view HTML -
Yuki Nakagawa, Koji Takahashi, Shigeki Miyahara, Yoshihisa Tamura, Koj ...Article type: CASE REPORT
2020Volume 53Issue 5 Pages 435-441
Published: May 01, 2020
Released on J-STAGE: May 30, 2020
JOURNAL FREE ACCESS FULL-TEXT HTMLA 66-year-old woman was referred to our hospital with a splenic tumor, pointed out by CT, for close examination of the chest nodule shadow during a health check. No subjective symptoms were observed. The abdomen was soft and flat and the mass was non-palpable. Blood biochemistry showed elevated levels of soluble IL2 receptor 2,537 U/ml and IgG4 139 mg/dl. Contrast-enhanced CT showed a splenic mass 7.5 cm in size with inflated growth and heterogeneous inside. Laparoscopic splenectomy was performed because a malignant tumor could not be ruled out. Histopathologically, a 7.7×6.0 cm yellow-whitish tumor with fibrous stroma and infiltration of lymphocytes and plasma cells was observed. Immunostaining revealed a number of IgG4-positive plasma cells, which were diagnosed as IgG4-related splenic inflammatory pseudotumor. We report on a rare case of IgG4 positive plasma cell infiltration in a splenic inflammatory pseudotumor.
 View full abstractDownload PDF (1762K) Full view HTML
View full abstractDownload PDF (1762K) Full view HTML -
Yoshitaka Ishikawa, Keisuke Goto, Takuya Nojiri, Katsumaro Suzuki, Kat ...Article type: CASE REPORT
2020Volume 53Issue 5 Pages 442-448
Published: May 01, 2020
Released on J-STAGE: May 30, 2020
JOURNAL FREE ACCESS FULL-TEXT HTMLAn 83-year-old man underwent a mesh repair for an abdominal incisional hernia 13 years previously in another hospital. He had occasionally experienced abdominal pain for several years. He visited our department because abdominal pain lasted for one week and gradually worsened. Redness, swelling, and tenderness of the surgical wound were noted and abdominal CT scan showed a localized abscess formation above the mesh with penetration of the transverse colon. An emergency incision and drainage was performed and pus discharge from the surgical wound was continued. Since the penetration into the transverse colon was suspected to be the cause of mesh infection and pus discharge, mesh removal and hernia repair were performed. After the surgery, partial necrosis of the abdominal skin and wound infection were observed and treated with continuous negative pressure therapy. When repairing an abdominal incisional hernia with a mesh, it is important to understand that there is a possibility of infection a long time after mesh repair, therefore it is important to fix the mesh properly so that it does not become damaged.
 View full abstractDownload PDF (2340K) Full view HTML
View full abstractDownload PDF (2340K) Full view HTML -
Daisuke Iitaka, Yusuke Takashima, Fumiaki Ochi, Susumu Nakashima, Juns ...Article type: CASE REPORT
2020Volume 53Issue 5 Pages 449-455
Published: May 01, 2020
Released on J-STAGE: May 30, 2020
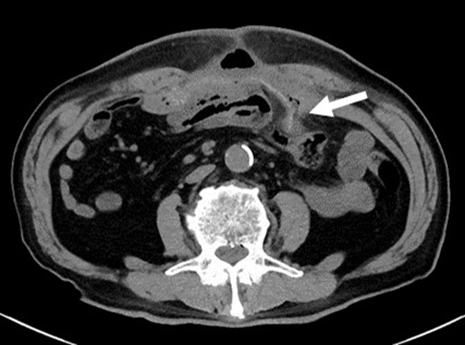
JOURNAL FREE ACCESS FULL-TEXT HTMLA 76-year-old man was emergently brought to our hospital because of dizziness. He was given a diagnosis of severe anemia, ascending colon cancer, and abscesses on the dorsal side, on contrast-enhanced abdominal CT. Percutaneous abscess drainage was performed, followed by laparoscopic right hemicolectomy. During the surgery, disseminated nodes were noted in the mesentery of the small intestine. Therefore, other regions of the small intestine were examined, revealing Meckel’s diverticulum, which was simultaneously resected. A tumor was found in Meckel’s diverticulum and we diagnosed Meckel’s diverticular adenocarcinoma based on postoperative pathological examination. Ectopic pancreatic tissue continuously surrounding the tumor and immunostaining findings led to a diagnosis of adenocarcinoma originating from ectopic pancreatic tissue. After surgery, chemotherapy was performed for peritoneal dissemination and hepatic metastasis. However, at 2 years and 3 months after surgery, the patient started to receive BSC. Adenocarcinoma is uncommon among patients with Meckel’s diverticulum, with only 21 cases, including ours, reported in Japan. We herein report a case of Meckel’s diverticular adenocarcinoma originating from ectopic pancreatic tissue, histopathologically detected by immunostaining.
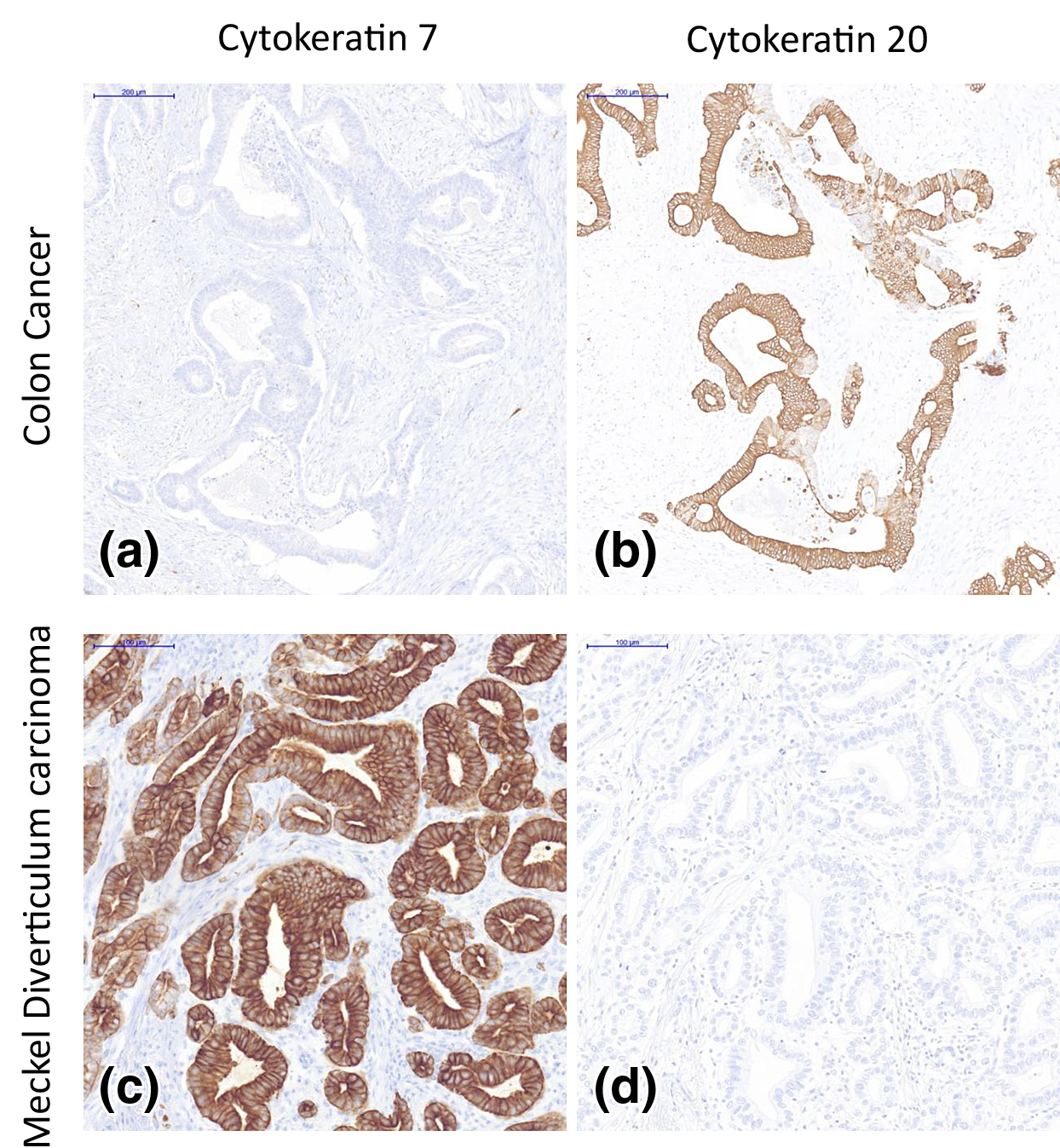 View full abstractDownload PDF (1433K) Full view HTML
View full abstractDownload PDF (1433K) Full view HTML -
Aya Noguchi, Hiroshi Yoshida, Shinya Kawaguchi, Akihiko Hashimoto, Sat ...Article type: CASE REPORT
2020Volume 53Issue 5 Pages 456-462
Published: May 01, 2020
Released on J-STAGE: May 30, 2020
JOURNAL FREE ACCESS FULL-TEXT HTMLA 65-year-old woman underwent laparoscopic anterior resection for rectal cancer in December 2013 and received adjuvant chemotherapy because it was pStage IIIb. She underwent thoracoscopic partial lung resection for lung metastasis in September 2015. One year later, mediastinal lymph node metastasis was noted and chemotherapy was started. In November 2018, she complained of dyspnea and exertional shortness of breath due to cardiac tamponade and underwent pericardial drainage. The cytology showed adenocarcinoma, therefore it was diagnosed as pericardial metastasis of primary rectal cancer. After being discharged, chemotherapy was restarted. In March 2019, she had loss of appetite and re-accumulation of pericardial effusion. Pericardial drainage was performed due to recurrence of cardiac tamponade. Her symptoms disappeared with drainage but she passed away on the 21st day after being admitted. Cardiac metastasis of gastrointestinal cancer is uncommon and pericardial metastasis of rectal cancer is very rare. It is presumed that lymphogenous metastasis is a major mechanism of pericardial metastasis. In cases like ours, where a patient has mediastinal lymph node metastasis, the possibility of cardiac tamponade should be considered.
View full abstractDownload PDF (1472K) Full view HTML
-
Naoya Sasaki, Ryuta NishitaiArticle type: SPECIAL REPORT
2020Volume 53Issue 5 Pages 463-472
Published: May 01, 2020
Released on J-STAGE: May 30, 2020
JOURNAL FREE ACCESS FULL-TEXT HTML -
Ryo Morimura, Eigo OtsujiArticle type: SPECIAL REPORT
2020Volume 53Issue 5 Pages 473-479
Published: May 01, 2020
Released on J-STAGE: May 30, 2020
JOURNAL FREE ACCESS FULL-TEXT HTML
-
Itaru EndoArticle type: EDITOR'S NOTE
2020Volume 53Issue 5 Pages en5-
Published: May 01, 2020
Released on J-STAGE: May 30, 2020
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (666K) Full view HTML
- |<
- <
- 1
- >
- >|