- |<
- <
- 1
- >
- >|
-
Hiroyuki NOJI2023Volume 63Issue 1 Pages 1
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (134K) Full view HTML
-
Kingo TAKIGUCHI, Hiroki SAKUTA, Masahito HAYASHI, Tatsuyuki WAIZUMI, K ...2023Volume 63Issue 1 Pages 5-11
Published: 2023
Released on J-STAGE: March 25, 2023
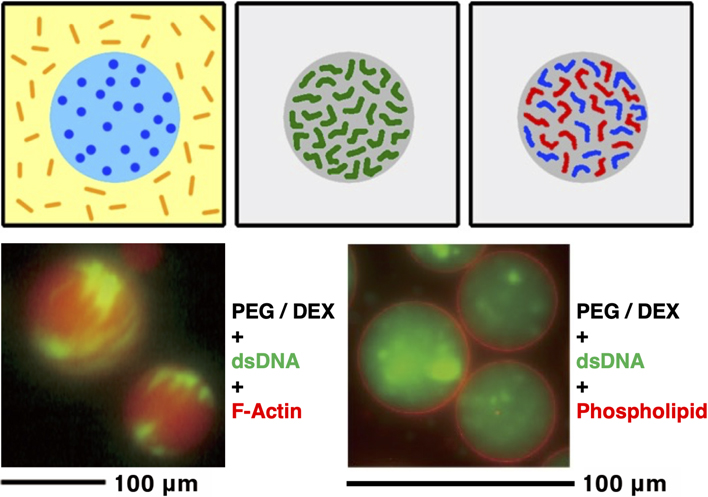
JOURNAL FREE ACCESS FULL-TEXT HTMLReproducing the dynamics of proteins and/or nucleic acids in a solution in which microdroplets of the similar size as cells or organelles are emerged by liquid-liquid phase separation, is an excellent model system. This is a powerful and rewarding strategy for understanding the properties of living cells. The targets of that research also include the formation of membraneless organelles, which has been gaining more attention in recent years. Here, the current results of our research using representative biopolymers, actin cytoskeleton and DNA, and phospholipids that are important components of biological membranes, would be introduced.
 View full abstractDownload PDF (3326K) Full view HTML
View full abstractDownload PDF (3326K) Full view HTML
-
Mirco DINDO, Alessandro BEVILACQUA, Paola LAURINO2023Volume 63Issue 1 Pages 12-15
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLLiquid-liquid phase separation (LLPS) is recognized as a mechanism for regulation of enzymatic activity. Biochemical mechanisms include concentrating reactants to enhance reaction rates or sequester enzymes and reactants from each other to reduce the reaction rate. On the other hand, LLPS might also regulate the diffusion of small molecules or important parameters for enzymatic activity (such as modulators, macromolecular crowding and changing the media physicochemical features) increasing or decreasing the reaction rate of the enzymes. Furthermore, the co-compartmentalization of specific enzymes can favour or speed up specific metabolic fluxes. Here, we discuss how LLPS contributed to generate a new era for enzyme regulation and the new possible subtle regulation mechanisms still unexplored.
 View full abstractDownload PDF (2198K) Full view HTML
View full abstractDownload PDF (2198K) Full view HTML -
Ryoji SUNO, Takuya KOBAYASHI2023Volume 63Issue 1 Pages 16-20
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLProstaglandins (PGs), lipid mediators, exert various effects in vivo through receptor-mediated signal transduction, and the elucidation of the molecular mechanisms of signal transduction by structural biology of PG receptors is expected to contribute to drug discovery. We have elucidated the molecular mechanisms of ligand binding mode, signal transduction mechanism, and G protein selectivity through structural analysis of PG receptors. In this paper, we would like to introduce the structure and function of GPCRs revealed through structural biology of PG receptors.
 View full abstractDownload PDF (3239K) Full view HTML
View full abstractDownload PDF (3239K) Full view HTML
-
Yoshimi KINOSHITA, Tomoko NISHIYAMA2023Volume 63Issue 1 Pages 21-23
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (1160K) Full view HTML -
Yukako NISHIMURA2023Volume 63Issue 1 Pages 24-26
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (2403K) Full view HTML -
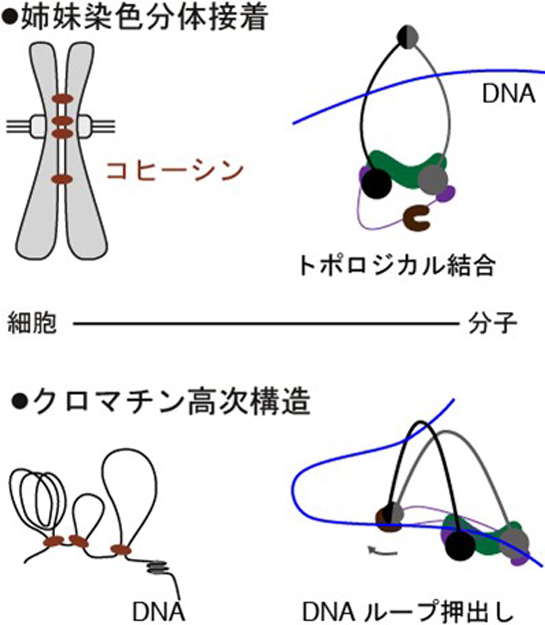
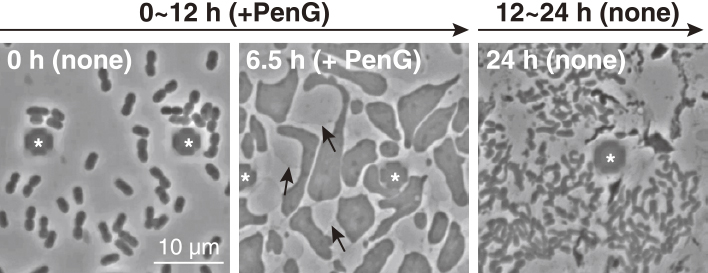
Daisuke SHIOMI, Taku OSHIMA2023Volume 63Issue 1 Pages 27-29
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (1079K) Full view HTML
-
Atsuhiro TOMITA2023Volume 63Issue 1 Pages 30-32
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (1752K) Full view HTML
-
Mutsuo NURIYA2023Volume 63Issue 1 Pages 33-37
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (1982K) Full view HTML
-
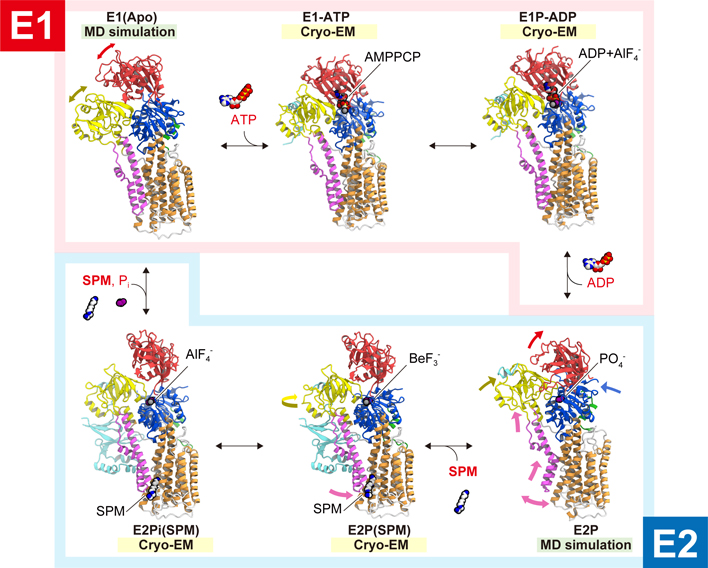
Yuki SUDO, Yoshie HARADA2023Volume 63Issue 1 Pages 39-40
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (1149K) Full view HTML -
Takashi NAGATA2023Volume 63Issue 1 Pages 41-42
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (1322K) Full view HTML -
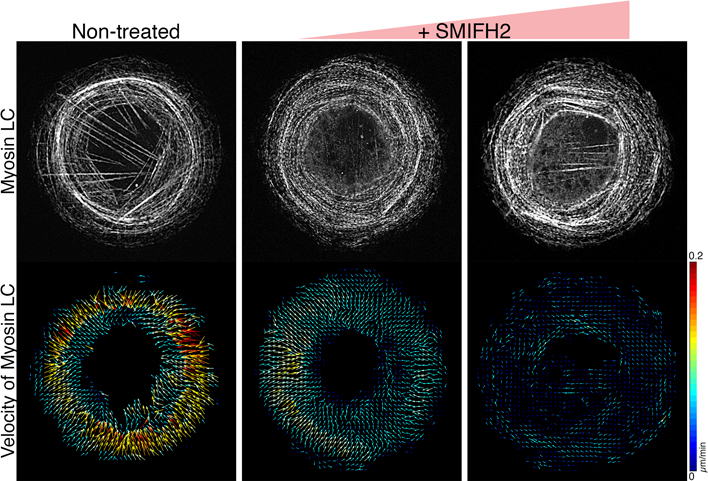
Sawako YAMASHIRO2023Volume 63Issue 1 Pages 43-44
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (853K) Full view HTML
-
[in Japanese]2023Volume 63Issue 1 Pages 45-46
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (2116K) Full view HTML
-
[in Japanese]2023Volume 63Issue 1 Pages 47-48
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (1114K) Full view HTML
-
[in Japanese]2023Volume 63Issue 1 Pages 49-50
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (2314K) Full view HTML
-
2023Volume 63Issue 1 Pages 38
Published: 2023
Released on J-STAGE: March 25, 2023
JOURNAL FREE ACCESS FULL-TEXT HTMLDownload PDF (127K) Full view HTML
- |<
- <
- 1
- >
- >|