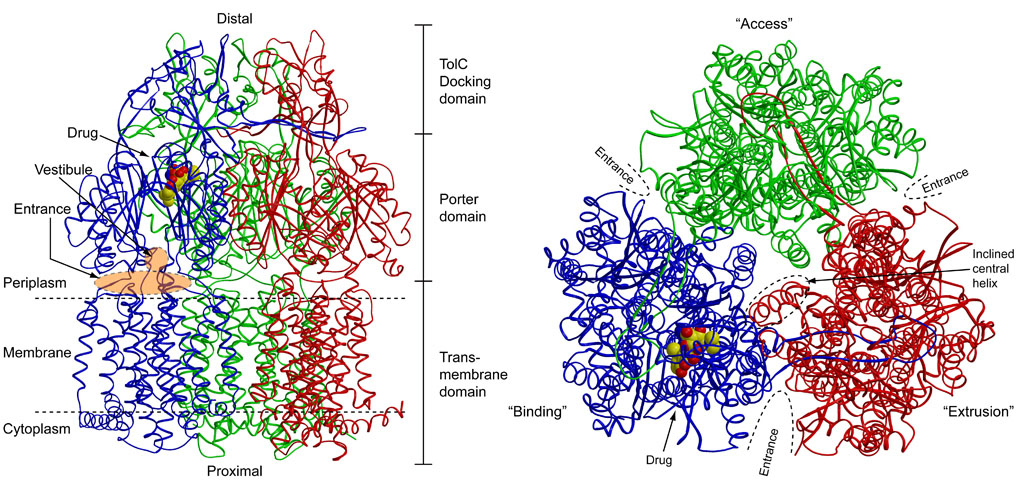
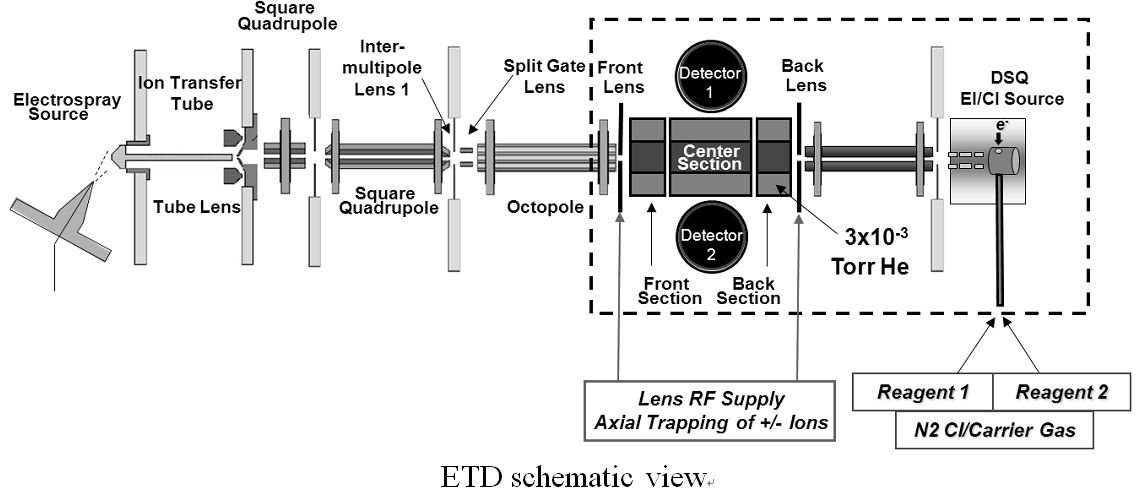
日本ヒトプロテオーム機構第5回大会
選択された号の論文の165件中1~50を表示しています
特別講演
-
セッションID: KL-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: KL-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: KL-3
発行日: 2007年
公開日: 2007/08/29
シンポジウム
細胞ネットワーク解析の最前線
-
セッションID: S1-1-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-1-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-1-3
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-1-4
発行日: 2007年
公開日: 2007/08/29
疾患・創薬マーカー探索の最前線
-
セッションID: S1-2-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-2-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-2-3
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-2-4
発行日: 2007年
公開日: 2007/08/29
ケミカルプロテオミクスの最前線
-
セッションID: S1-3-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-3-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-3-3
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-3-4
発行日: 2007年
公開日: 2007/08/29
グライコミクスの新展開
-
セッションID: S1-4-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-4-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-4-3
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-4-4
発行日: 2007年
公開日: 2007/08/29
メタボプロテオミクスの新展開
-
セッションID: S1-5-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-5-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-5-3
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-5-4
発行日: 2007年
公開日: 2007/08/29
プロテオミクスの新技術
-
セッションID: S1-6-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-6-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-6-3
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-6-4
発行日: 2007年
公開日: 2007/08/29
プロテオミクスの新技術・翻訳後修飾の解析
-
セッションID: S1-7-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-7-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-7-3
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-7-4
発行日: 2007年
公開日: 2007/08/29
細胞装置のプロテオミクス解析
-
セッションID: S1-8-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-8-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S1-8-3
発行日: 2007年
公開日: 2007/08/29
疾病メカニズムのプロテオミクス解析 (招待講演)
-
セッションID: S2-1-1
発行日: 2007年
公開日: 2007/08/29
疾病メカニズムのプロテオミクス解析
-
セッションID: S2-1-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S2-1-3
発行日: 2007年
公開日: 2007/08/29
構造プロテオミクスの最前線
-
セッションID: S2-2-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S2-2-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S2-2-3
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S2-2-4
発行日: 2007年
公開日: 2007/08/29
疾患プロテオミクスの最前線
-
セッションID: S2-3-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S2-3-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S2-3-3
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S2-3-4
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S2-3-5
発行日: 2007年
公開日: 2007/08/29
プロテオミクスの新技術・質量分析法を中心に
-
セッションID: S2-4-1
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S2-4-2
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S2-4-3
発行日: 2007年
公開日: 2007/08/29
-
セッションID: S2-4-4
発行日: 2007年
公開日: 2007/08/29