日本ヒトプロテオーム機構第7回大会
選択された号の論文の136件中1~50を表示しています
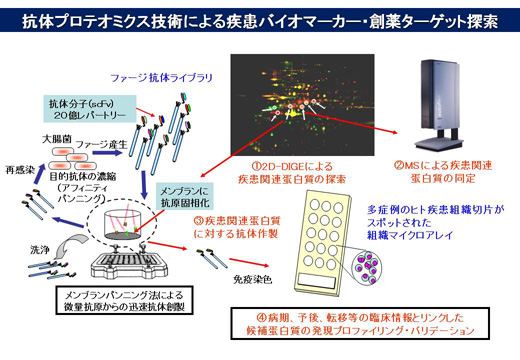
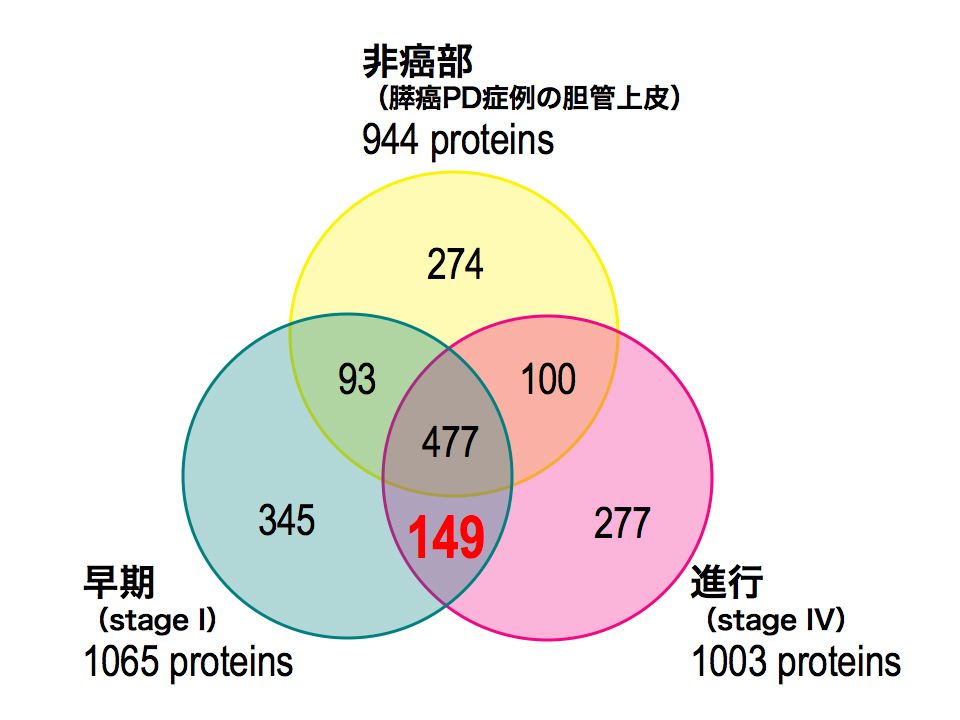
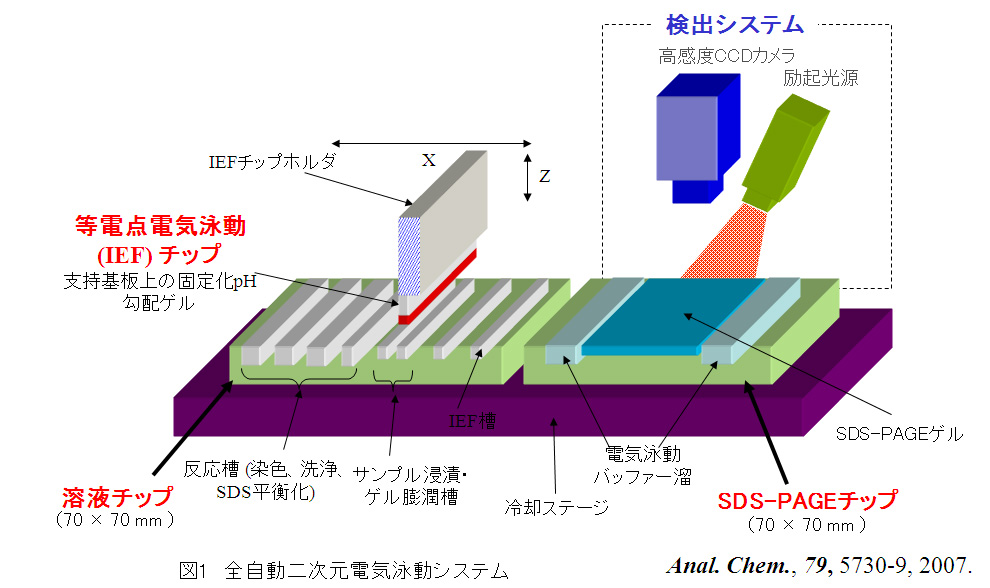
特別招待講演
-
セッションID: SIL1
発行日: 2009年
公開日: 2009/09/14
招待講演
-
セッションID: IL-1
発行日: 2009年
公開日: 2009/09/14
-
セッションID: IL-2
発行日: 2009年
公開日: 2009/09/14
-
セッションID: IL-3
発行日: 2009年
公開日: 2009/09/14
教育講演
-
セッションID: EL1
発行日: 2009年
公開日: 2009/09/14
-
セッションID: EL2
発行日: 2009年
公開日: 2009/09/14
シンポジウム1
プロテオミクスの医学への応用(その1) マーカー探索(組織)
-
セッションID: S1-1
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S1-2
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S1-3
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S1-4
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S1-5
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S1-6
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S1-7
発行日: 2009年
公開日: 2009/09/14
シンポジウム2
タンパク質研究から医薬品開発への道
-
セッションID: S2-1
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S2-2
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S2-3
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S2-4
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S2-5
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S2-6
発行日: 2009年
公開日: 2009/09/14
シンポジウム3
グライコミクスの医学への応用(糖鎖)
-
セッションID: S3-1
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S3-2
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S3-3
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S3-4
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S3-5
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S3-6
発行日: 2009年
公開日: 2009/09/14
シンポジム4
基礎生物学とプロテオミクス(翻訳後修飾、リン酸化)
-
セッションID: S4-1
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S4-2(P-45)
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S4-3
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S4-4
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S4-5
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S4-6
発行日: 2009年
公開日: 2009/09/14
シンポジウム5
プロテオミクスの医学への応用(その2) マーカー探索(血液・尿)
-
セッションID: S5-1
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S5-2
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S5-3
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S5-4
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S5-5
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S5-6
発行日: 2009年
公開日: 2009/09/14
シンポジウム6
プロテオミクスの医学への応用(その3) 疾患メカニズム解析
-
セッションID: S6-1
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S6-2
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S6-3
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S6-4
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S6-5
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S6-6
発行日: 2009年
公開日: 2009/09/14
シンポジウム7
プロテオミクスの薬学への応用
-
セッションID: S7-1
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S7-2
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S7-3
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S7-4
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S7-5
発行日: 2009年
公開日: 2009/09/14
-
セッションID: S7-6
発行日: 2009年
公開日: 2009/09/14
シンポジウム8
プロテオミクスの農学への応用
-
セッションID: S8-1
発行日: 2009年
公開日: 2009/09/14