
- |<
- <
- 1
- >
- >|
-
2023 年 109 巻 7 号 p. Cover-
発行日: 2023/07/01
公開日: 2023/06/30
ジャーナル オープンアクセスPDF形式でダウンロード (663K) -
2023 年 109 巻 7 号 p. Contents-
発行日: 2023/07/01
公開日: 2023/06/30
ジャーナル オープンアクセスPDF形式でダウンロード (3524K) -
2023 年 109 巻 7 号 p. Editorial-
発行日: 2023/07/01
公開日: 2023/06/30
ジャーナル オープンアクセスPDF形式でダウンロード (205K)
-
馬 喆, 丸岡 大佑, 村上 太一, 葛西 栄輝, 樋口 隆英原稿種別: 論文
2023 年 109 巻 7 号 p. 559-566
発行日: 2023/07/01
公開日: 2023/06/30
ジャーナル オープンアクセス HTMLDepletion trend of high-grade iron ore resources have become significant due to the recent increase in world steel production. Therefore, effective utilization of low-grade and low-quality ores has been and will be strongly required. Since iron ore concentrates obtained by beneficiation processes are usually fine, e.g., less than 100 µm in particles size, it is difficult to directly use a large amount of them to the sintering process. To cope with such a problem, the composite sintering process, namely MEBIOS (Mosaic EmBedding Iron Ore Sintering), has been proposed, which pre-granulates fine ores to be green pellets. The present study deals with the effects of limestone size and substituting ratio with burnt lime on the compression strength of the sintered pellets. The green pellets were prepared using fine hematite ores, limestone and burnt lime and sintered at up to 1300°C. The compression strength of the sintered pellet increased with a decrease in particles size of limestone when it was less than 500 µm, although relatively higher strength was obtained for the case using limestone having 500-1000 µm in size. The porosity of the pellets prepared by the partial substitution of limestone with burnt lime decreased and their compression strength increased with an increase in the substitution ratio. It seems due to the dispersion effect of extremely fine slacked lime particles formed by the hydration reaction of burnt lime.
 抄録全体を表示PDF形式でダウンロード (2764K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (2764K) HTML形式で全画面表示
-
中山 健一原稿種別: 論文
2023 年 109 巻 7 号 p. 567-572
発行日: 2023/07/01
公開日: 2023/06/30
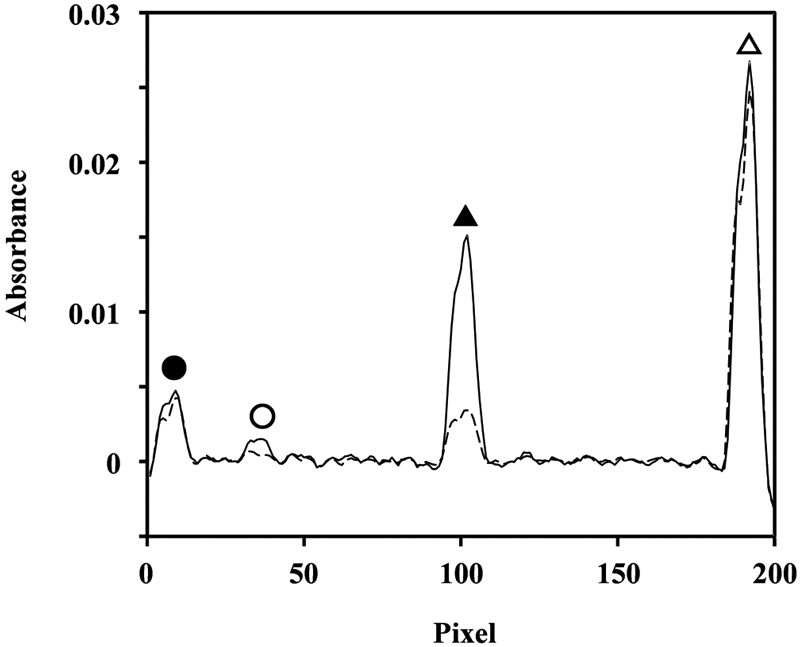
ジャーナル オープンアクセス HTMLThis article describes a procedure using flame atomic absorption spectrometry for specimen preparation to quantify tungsten in alloyed tool steel and high-speed steel. The sample solution was prepared using a microwave-assisted digestion with a mixed acid of hydrofluoric, nitric, and phosphoric acids. Vanadium was added as an internal standard element to the sample solution to measure tungsten and vanadium absorption lines simultaneously using a spectrometer equipped with a multi-wavelength system, which comprised a xenon lamp as a continuum light source and an echelle grating. The internal standard method, with its tungsten-to-vanadium absorbance ratio, contributed to accurate and precise quantification of high-content tungsten in tool steel samples.
 抄録全体を表示PDF形式でダウンロード (738K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (738K) HTML形式で全画面表示 -
板橋 大輔, 水上 和実原稿種別: 論文
2023 年 109 巻 7 号 p. 573-580
発行日: 2023/07/01
公開日: 2023/06/30
ジャーナル オープンアクセス HTMLTo determine the number density of fine precipitates in steels by asymmetric flow field-flow fractionation (AF4) with inductively coupled plasma–mass spectrometry (ICP–MS), an analysis method employing flow injection was investigated. For accurate calibration, matrix matching was performed by mixing the standard solution and AF4 carrier solution in front of a nebulizer. Two surfactants were used for AF4 separation; it was found that the appropriate selection of surfactants based on their acidity constant is essential to avoid salt precipitation. In addition, the effect of the AF4 retention time on recovery was investigated. A long retention time led to the adsorption and aggregation of the samples in the AF4 separation channel. Results showed that an AF4 retention time within 20 min facilitated superior recovery. Moreover, five types of AuNPs were analyzed via AF4–ICP–MS and quantified using flow injection analysis. Good analytical performance was achieved for all AuNPs and the recoveries exceeded 93%, and the coefficient of variation was within 5%. The effect of particle size on the recovery was not confirmed.
Furthermore, the developed flow injection analysis for AF4–ICP–MS was applied to evaluate niobium carbide (NbC) precipitates in steels. The number density of nanometer-sized NbC was quantified to be within 1013 to 1014 particles per 1 g of Fe. It was quantitatively confirmed that the long-duration heat treatment led to an increase in the number density of nanometer-sized NbC. Hence, this method can be useful for quantitatively analyzing the size and number density of nanoprecipitates in steels.
 抄録全体を表示PDF形式でダウンロード (2545K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (2545K) HTML形式で全画面表示
-
小林 弘和, 武田 玄太郎, 加藤 健司, 脇本 辰郎原稿種別: 論文
2023 年 109 巻 7 号 p. 581-591
発行日: 2023/07/01
公開日: 2023/06/30
ジャーナル オープンアクセス HTMLGas jet cooling is widely used because the device is simple, it is possible to prevent oxidation and a uniform cooling ability can be obtained with thin steel sheets. Because the gas jet cooling ability is affected by physical properties of the gas such as the mixed gas ratio, quantitative evaluation of the influence of these factors is very important. However, few studies concerning prediction of the mixed gas jet cooling ability in atmospheres with different concentrations have been published.
In this research, the results of experiments and fluid analyses with an air-helium gas jet in an air atmosphere were compared with the results obtained with Martin's non-dimensional empirical equations. As the nozzle condition, a single round nozzle with a tapered shape was examined. The helium concentrations with respect to air were 0, 20, 50 and 100 vol%, and the pressure conditions were 3 and 5 kPa.
Compared with the experimental results, Martin's equations largely predicted improvement of cooling performance with increasing helium concentrations. In the analyses, mixing with air increased as the helium concentration decreased.
The trend of divergence between the experimental and predicted cooling ability was clearly presented in this research. The results of this study make it possible to improve the accuracy of predictions of the impinging jet cooling ability with different concentrations between an atmosphere and a gas jet.
 抄録全体を表示PDF形式でダウンロード (1739K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (1739K) HTML形式で全画面表示 -
建部 勝利, 藤田 俊輔, 藤本 仁原稿種別: 論文
2023 年 109 巻 7 号 p. 592-604
発行日: 2023/07/01
公開日: 2023/06/30
[早期公開] 公開日: 2023/02/16ジャーナル オープンアクセス HTMLThe spray cooling of moving hot solids is widely performed in the steel industry. Understanding flow and heat transfer when droplets impinge on moving hot solids is important. By simultaneous visualization with flash photography and temperature measurement using thermography, the flow and heat transfer of a droplet train obliquely impinging on a moving solid at high temperatures was experimentally investigated. A rectangular test piece (SUS303) was heated to 500 °C at a moving velocity of 0.25–1.5 m/s. The test liquid was water at approximately 25 °C. The pre-impact droplet diameter, impact velocity, and inter-spacing between two successive droplets were 0.69 mm, 2.2 m/s, and 2.23 mm, respectively. The tilt and torsional angles were 50° and −30–60°, respectively. No coalescence of droplets was observed; the droplets deformed independently on the moving solid, even though the torsional angle generated a velocity component along the width of the solid. The surface temperature of solid after droplet impingements depended on the experimental conditions. Wavy temperature profile was obtained when the moving distance of solid was large during two successive collisions. The temperature changed continuously for the small distances. In this regard, a simple model considering droplet movement, collisional deformation behavior, and solid migration can explain this phenomenon by the overlap of the cooling regions of the droplets. Furthermore, experimental and numerical analyses show that the heat removal rate of individual droplets is constant at approximately 12.5 MW/m2 and depends on the total contact time when multiple droplets collide.
 抄録全体を表示PDF形式でダウンロード (9142K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (9142K) HTML形式で全画面表示
-
松永 哲也, 本郷 宏通, 山﨑 政義, 田淵 正明原稿種別: 論文
2023 年 109 巻 7 号 p. 605-612
発行日: 2023/07/01
公開日: 2023/06/30
ジャーナル オープンアクセス HTMLA low carbon/ medium nitrogen 16Cr-8Ni-2Mo weld metal (16-8-2 WM) and a Mod. 316 WM were crept at 823 K-923 K to ensure long-term safety of components in fast breeder reactors (FBRs). For this purpose, creep tests were performed up to 238,064.7 hours in this study. Although the Mod. 316 WM showed longer time-to-rupture at the temperatures, the WM showed lower ductility and faster transition time to tertiary creep region relative to time-to-rupture. The behaviors were influenced by precipitation of σ phase during creep test. Matrix deformed easier because solid solution strengthening elements, i.e., Cr and Mo, diffused into the precipitate for the Mod. 316 WM with higher contents of the elements, and then WM became more brittle in association with precipitation of σ phase than the 16-8-2 WM. According to the above results, the precipitation of σ phase affects not only creep behavior but also time-to-rupture of the weld metals for 316FR steel.
 抄録全体を表示PDF形式でダウンロード (2929K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (2929K) HTML形式で全画面表示 -
中西 大貴, 内山 徹也, 白幡 浩幸, 髙橋 学原稿種別: 論文
2023 年 109 巻 7 号 p. 613-622
発行日: 2023/07/01
公開日: 2023/06/30
ジャーナル オープンアクセス HTMLThe microstructures of intra-granular bainite nucleated on Ti-oxide particles (IGB) and grain boundary upper bainite (GBB) in Ti-deoxidized steel (Fe-0.1mass%C-1.5mass%Mn-2mass%Ni-1mass%Cu) were investigated by EBSD analysis and 3-dimentional observation. The steel was austenitized at 1673 K for 23 s, held at 803 K for 7 s-20 ks and then quenched to room temperature. 803 K is just below the bainite transformation start temperature. IGB and GBB were observed at 5% bainite transformation. Despite the formation of IGB, part of GBB grew to a size of 100 μm at 17% bainite transformation, and coarsened to 130-200 μm at 85% bainite transformation. Mechanism of the GBB coarsening is discussed in terms of differences in (1) the microstructures and (2) the nucleation site of IGB and GBB. A single packet with many blocks was observed in GBB nucleated at whole surface of austenite grain boundaries with a size of 400-500 μm, while multiple packets with two blocks were observed in IGB nucleated on Ti-oxide particles with a size of 1-2 μm. IGB suppressed the growth of GBB by impingement. However, as the GBB was much larger than IGB, part of the GBB was not impinged by the IGB and continued to grow. GBB grew until all blocks of GBB were impinged by IGB and/or transformation was finished, resulted in GBB coarsening.
 抄録全体を表示PDF形式でダウンロード (5950K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (5950K) HTML形式で全画面表示
-
林 宏太郎, 木之下 雄一, 藤村 里奈, 髙橋 学原稿種別: 論文
2023 年 109 巻 7 号 p. 623-637
発行日: 2023/07/01
公開日: 2023/06/30
ジャーナル オープンアクセス HTMLThe distribution of alloying elements was investigated to clarify latent features of the microstructure formation in TRIP-aided Multi-phase steels by using an unsupervised machine learning technique. The cold-rolled specimens with a composition of Fe-0.20mass%C-1.5mass%Si-1.5mass%Mn were heat-treated under various conditions. The FE-EPMA (field emission electron probe microanalysis) method was used to accurately measure the distribution of C, Si and Mn. The two-phase microstructure of ferrite and austenite was formed during intercritical annealing at 800 °C for 120 s. C and Mn were distributed in the austenite region while Si was distributed in the ferrite region. Part of the austenite transformed to bainite by subsequent heat-treatment of isothermal holding at 400 °C for 1200 s. The C concentration in the austenite region increased due to bainite formation. The microstructure formation factor was estimated by primary component analysis of big data on the elemental distribution. The cumulative contribution ratio of the second principal component was 0.85. Therefore, most of the information was contained in the first and second principal components. In the first principal component, the coefficients for the standardized C and Mn concentrations possessed positive values, while that for the standardized Si concentration possessed negative value. The tendency of each coefficient corresponds to the C, Si and Mn distribution caused by intercritical annealing. Moreover, the absolute value of the coefficient for the standardized C concentration was significantly larger than those of the other coefficients in the second principal component. The component represents the C enrichment of austenite due to bainite transformation.
 抄録全体を表示PDF形式でダウンロード (22104K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (22104K) HTML形式で全画面表示
-
木南 俊哉原稿種別: 論文
2023 年 109 巻 7 号 p. 638-648
発行日: 2023/07/01
公開日: 2023/06/30
ジャーナル オープンアクセス HTMLThe effect of combined addition of V and Mo on hydrogen trapping was investigated using tempered martensitic steels (0.3%C-1.2%Cr-0.5%Mo-0.4%V, 1.5%Mo-0.2%V or 2.0%Mo-0.1%V in mass%), for the purpose of improving hydrogen embrittlement strength. The trapped hydrogen content and the hydrogen trapping energy were evaluated under various tempering conditions and compared with the single addition 0.2%V and 1.0%Mo steels in the previous paper. The trapped hydrogen content and the hydrogen trapping energy of 0.5%Mo-0.4%V and 1.5%Mo-0.2%V steels were higher at 650°C tempering temperature for 1 hour than at 600°C. On the other hand, those values of 0.2%V and 1.0%Mo steels were lower at a tempering temperature of 650°C than at 600°C. This was presumed to be due to the some interaction between V and Mo at a tempering temperature of 650°C. In the 0.5%Mo-0.4%V and 1.5%Mo-0.2%V steels at tempering temperatures of 600°C and 650°C for 1 hour, the plate-shaped Cr enriched cubic (Mo, V)C fine carbides with a width of about 1nm and a length of about 20nm or less were precipitated, and there was no significant difference in the number density. The V/Mo peak ratio of the fine carbides by EDS analysis was higher at a tempering temperature of 650°C than at 600°C and the length of fine carbides was slightly longer. The 2.0%Mo-0.1%V steel was estimated to have less trapped hydrogen content at a tempering temperature of 650°C than at 600°C, similar to single addition steels, because of the smaller V/Mo mole ratio.
 抄録全体を表示PDF形式でダウンロード (4458K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (4458K) HTML形式で全画面表示 -
浦中 祥平, 平嶋 一誠, 前田 拓也, 増村 拓朗, 土山 聡宏, 川本 雄三, 白幡 浩幸, 植森 龍治原稿種別: 論文
2023 年 109 巻 7 号 p. 649-660
発行日: 2023/07/01
公開日: 2023/06/30
ジャーナル オープンアクセス HTMLThe relationship between hardness and solute carbon concentration estimated via electrical resistivity measurement was investigated in as-quenched and tempered martensitic steels containing carbon of 0.3-0.6 mass%. As a result of corelating the amount of hardening due to carbon in solid solution with the solute carbon concentration, by the calculation to subtract precipitation strengthening, grain refinement strengthening, dislocation strengthening, and softening due to retained austenite from the total strengthening, we derived an equation of solid solution strengthening, where the hardening increases proportionally to the 1 / 2 or 2 / 3 power of the solute carbon concentration. It was confirmed that the effects of the factors other than solid solution strengthening due to carbon on hardness are relatively small in tempered specimens when the tempering temperature is less than 673 K; therefore, the change in hardness in tempered martensitic steels can be mostly explained by solute carbon concentration regardless of carbon content.
 抄録全体を表示PDF形式でダウンロード (8867K) HTML形式で全画面表示
抄録全体を表示PDF形式でダウンロード (8867K) HTML形式で全画面表示
- |<
- <
- 1
- >
- >|