
- Issue 16 Pages 2157-
- Issue 15 Pages 1805-
- Issue 14 Pages 1613-
- Issue 13 Pages S1141-
- Issue 12 Pages S1037-
- Issue 11 Pages 1443-
- Issue 10 Pages 1273-
- Issue 9 Pages 1077-
- Issue 8 Pages 917-
- Issue 7 Pages 751-
- Issue 6 Pages 585-
- Issue 5 Pages S389-
- Issue 4 Pages S1-
- Issue 3 Pages 403-
- Issue 2 Pages 233-
- Issue 1 Pages 19-
- Issue 16 Pages 2153-
- Issue 15 Pages 1977-
- Issue 14 Pages 1813-
- Issue 13 Pages S1285-
- Issue 12 Pages S1043-
- Issue 11 Pages 1667-
- Issue 10 Pages 1481-
- Issue 9 Pages 1231-
- Issue 8 Pages 891-
- Issue 7 Pages 711-
- Issue 6 Pages 538-
- Issue 5 Pages S407-
- Issue 4 Pages S1-
- Issue 3 Pages 347-
- Issue 2 Pages 173-
- Issue 1 Pages 14-
- Issue 16 Pages 1837-
- Issue 15 Pages 1711-
- Issue 14 Pages 1569-
- Issue 13 Pages S1205-
- Issue 12 Pages S1034-
- Issue 11 Pages 1423-
- Issue 10 Pages 1269-
- Issue 9 Pages 1059-
- Issue 8 Pages 925-
- Issue 7 Pages 775-
- Issue 6 Pages 627-
- Issue 5 Pages S287-
- Issue 4 Pages S1-
- Issue 3 Pages 301-
- Issue 2 Pages 147-
- Issue 1 Pages 12-
- Issue 16 Pages 2179-
- Issue 15 Pages 1795-
- Issue 14 Pages 1631-
- Issue 13 Pages S1053-
- Issue 12 Pages S1023-
- Issue 11 Pages 1501-
- Issue 10 Pages 1315-
- Issue 9 Pages 987-
- Issue 8 Pages 767-
- Issue 7 Pages 621-
- Issue 6 Pages 473-
- Issue 5 Pages S305-
- Issue 4 Pages S1-
- Issue 3 Pages 299-
- Issue 2 Pages 151-
- Issue 1 Pages 16-
- Issue 16 Pages 1945-
- Issue 15 Pages 1699-
- Issue 14 Pages 1531-
- Issue 13 Pages S1055-
- Issue 12 Pages S1013-
- Issue 11 Pages 1367-
- Issue 10 Pages 1215-
- Issue 9 Pages 1087-
- Issue 8 Pages 887-
- Issue 7 Pages 721-
- Issue 6 Pages 507-
- Issue 5 Pages S317-
- Issue 4 Pages S1-
- Issue 3 Pages 343-
- Issue 2 Pages 187-
- Issue 1 Pages 17-
- Issue 16 Pages 2405-
- Issue 15 Pages 2067-
- Issue 14 Pages 1865-
- Issue 13 Pages 1675-
- Issue 12 Pages S1055-
- Issue 11 Pages S1015-
- Issue 10 Pages 1479-
- Issue 9 Pages 1129-
- Issue 8 Pages 895-
- Issue 7 Pages 711-
- Issue 6 Pages 545-
- Issue 5 Pages S325-
- Issue 4 Pages S1-
- Issue 3 Pages 369-
- Issue 2 Pages 193-
- Issue 1 Pages 16-
- Issue 16 Pages 2573-
- Issue 15 Pages 2261-
- Issue 14 Pages 2073-
- Issue 13 Pages S1111-
- Issue 12 Pages S1001-
- Issue 11 Pages 1867-
- Issue 10 Pages 1657-
- Issue 9 Pages 1409-
- Issue 8 Pages 1043-
- Issue 7 Pages 841-
- Issue 6 Pages 649-
- Issue 5 Pages S415-
- Issue 4 Pages S1-
- Issue 3 Pages 431-
- Issue 2 Pages 225-
- Issue 1 Pages 3-
- |<
- <
- 1
- >
- >|
-
2018 Volume 104 Issue 11 Pages Cover-
Published: November 01, 2018
Released on J-STAGE: October 31, 2018
JOURNAL OPEN ACCESSDownload PDF (658K) -
2018 Volume 104 Issue 11 Pages Contents-
Published: November 01, 2018
Released on J-STAGE: October 31, 2018
JOURNAL OPEN ACCESSDownload PDF (244K) -
2018 Volume 104 Issue 11 Pages Editorial-
Published: November 01, 2018
Released on J-STAGE: October 31, 2018
JOURNAL OPEN ACCESSDownload PDF (228K)
-
Atsushi Ishigaki, Keiji Okumura2018 Volume 104 Issue 11 Pages 613-619
Published: 2018
Released on J-STAGE: October 31, 2018
Advance online publication: June 16, 2018JOURNAL OPEN ACCESS FULL-TEXT HTMLExperiments were conducted to extract neodymium in bismuth by melting Fe-Nd-B magnets and bismuth together in a graphite crucible at 1200°C. Molten iron and bismuth separated into two phases, and neodymium in the magnet dissolved in the bismuth phase. Neodymium dissolved in bismuth was considered to form BiNd as a result of XRD. The neodymium concentration in iron phase after the dissolution treatment was 0.05 at.% or less,and the recovery rate was 99% and more. Compared to other metals(Mg, Ag, Cu) that separate into two phases with respect to molten iron, bismuth can be said to be a metal extraction material with a low melting point, safety and low energy cost.
View full abstractDownload PDF (4865K) Full view HTML
-
Guowei Zhang, Hiromasa Sakai, Nao-Aki Noda, Yoshikazu Sano, Shun Oshir ...2018 Volume 104 Issue 11 Pages 620-627
Published: 2018
Released on J-STAGE: October 31, 2018
JOURNAL OPEN ACCESS FULL-TEXT HTMLCeramic roller can be used in the heating furnace conveniently because of its high temperature resistance. The roller consists of ceramic sleeve and steel shaft connected only under a small shrink fitting ratio because of the brittleness. However, the coming out of the shaft sometimes happens from the ceramic sleeve under repeated load. Therefore, it is important to find out the driving out force generation mechanism to prevent the coming out failure. Based on the previous studies, a two-dimensional shrink fitted structure is considered by replacing the shaft with the inner plate and by replacing the sleeve with the outer plate. Then, this research focuses on calculating the driving out force generated on the inner plate by introducing a newly designed stopper on the outer plate. The finite element simulation shows that the coming out phenomenon can be prevented due to the contact between the inner plate and the stopper installed on the outer plate. Then the mechanism of driving out force generation is clarified. Finally, the process of coming out in terms of the residual displacement is illuminated.
 View full abstractDownload PDF (4194K) Full view HTML
View full abstractDownload PDF (4194K) Full view HTML
-
Kakeru Ninomiya, Kazutaka Kamitani, Yusuke Tamenori, Kazuki Tsuruta, T ...2018 Volume 104 Issue 11 Pages 628-633
Published: 2018
Released on J-STAGE: October 31, 2018
Advance online publication: July 19, 2018JOURNAL OPEN ACCESS FULL-TEXT HTMLObservation of chemical state of solid-solution carbon in a low-carbon steel was tried by C-K near-edge x-ray absorption fine structure spectra measurement. In addition, the wavelength dependence of the photoelectron spectrum on the surface of the bulk steel was evaluated, and the contamination and oxidation layer of 3 nm in thickness on the surface of the steel was found. As a result, it was possible to observe the chemical state change of carbon existing in the bulk iron located deeper than the oxidation and contamination layer, by evaluating the difference spectra between the sample and the reference. Furthermore, by evaluating the shape change of the difference spectra depending on the heat treatment time, this study suggested that the chemical state of carbon in bulk iron changes with heat treatment.
View full abstractDownload PDF (1007K) Full view HTML -
Kazumi Mizukami, Daisuke Itabashi, Michihiro Aimoto, Masayuki Nishifuj ...2018 Volume 104 Issue 11 Pages 634-639
Published: 2018
Released on J-STAGE: October 31, 2018
JOURNAL OPEN ACCESS FULL-TEXT HTMLCopper sulfide (CuxS) has been frequently observed in steel samples, prepared using selective potentiostatic etching by electrolytic dissolution (SPEED). It is often the case that CuxS is detected unexpectedly from the precipitates extracted from steel samples by selective potentiostatic etching, although such CuxS formation during the heat treatment conducted is not anticipated by the thermodynamic equilibrium calculations.
In this study, we observed such artificial CuxS along with manganese sulfide (MnS) precipitates, which were extracted from steel materials by SPEED, using secondary electron microscope (SEM) equipped with energy dispersive X-ray spectroscopy (EDX) and Auger electron spectroscopy. These CuxS-MnS sulfide complex would be formed by the following mechanism: as the solubility of CuxS is far bigger (i.e. 10 times or more) than that of MnS, Cu2+ ion dissolved from steel matrix would be exchanged with Mn2+ ion on the MnS surface during the etching process, leading to a formation of CuxS-MnS sulfide complex.
In order to suppress the formation of such CuxS, we propose the use of following electrolyte: a non-aqaueous solutions of 4% methyl salicylate + 1% salicylic acid + 1% tetramethylammonium chloride (TMAC) + 5% Triethylenetetramine (TET) in volume fraction, in methyl alcohol (Cu ion selective hold etching by electrolytic dissolution, abridged as CUSH electrolyte). Then, this electrolyte was applied to precipitates in steel samples. It was effective to prevent the formation of sulfides in electrolyte, with the effect of metallic (Cu2+, Ag+, Pb+, etc.) chelating ability of TET.
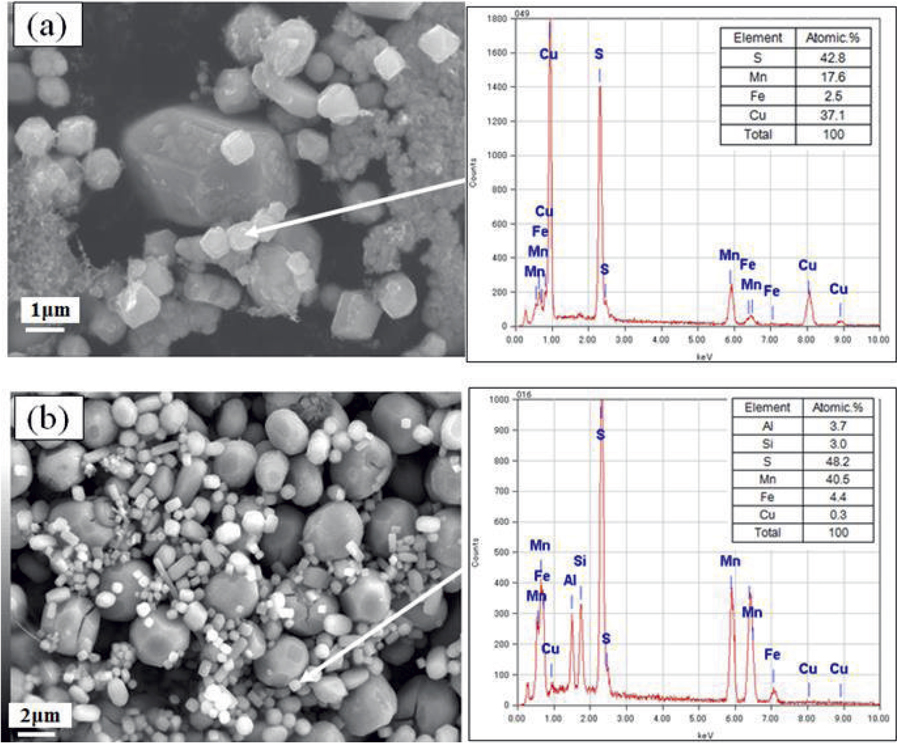 Comparison of sulfides by SEM-EDX after SPEED etching with (a) 4% MS (b) 4%MS+5%TET. Fullsize ImageView full abstractDownload PDF (5171K) Full view HTML
Comparison of sulfides by SEM-EDX after SPEED etching with (a) 4% MS (b) 4%MS+5%TET. Fullsize ImageView full abstractDownload PDF (5171K) Full view HTML
-
Yukihiro Matsubara, Yukio Kimura, Hiroshi Utsunomiya2018 Volume 104 Issue 11 Pages 640-645
Published: 2018
Released on J-STAGE: October 31, 2018
JOURNAL OPEN ACCESS FULL-TEXT HTMLIn hot rolling of ferritic stainless steel, prevention of sticking is very important from the viewpoint of productivity. However, the formation mechanism of sticking has not been clarified sufficiently. Therefore, in this work, rolling experiments were carried out using a tribo-simulator. The results clarified the following points: Sticking occurs more easily on ferritic stainless steel than on high strength steel. On ferritic stainless steel, the work roll sticks with the hot-rolled sheet at the entrance of roll-bite, and the work roll then moves forward on the hot-rolled sheet. Therefore, it is thought that the surface layer of the hot-rolled sheet is fractured by large shear strain, and the work roll stuck with the fractured layer advances further on the sheet, forming a defect with an accumulated fractured surface layer on the hot-rolled sheet. Applying a lubricant oil is effective for prevention of sticking between the work roll and hot-rolled sheet.
View full abstractDownload PDF (3070K) Full view HTML
-
Masaki Koba, Yusuke Fushiwaki, Yasunobu Nagataki2018 Volume 104 Issue 11 Pages 646-654
Published: 2018
Released on J-STAGE: October 31, 2018
JOURNAL OPEN ACCESS FULL-TEXT HTMLThe Fe-Zn alloying reaction and the selective oxidation behavior of 0.7 mass% Si - 1.15 mass% Mn added hot-rolled steel annealed at 600-800°C were investigated by comparison with those of cold-rolled steel. The Fe-Zn reactivity of the hot-rolled steel improved from 600°C to 700°C, while, it deteriorated from 700°C to 800°C. Above 700°C, the amount of Fe-Si-Mn oxide on the steel surface increased with increasing temperature and deteriorated Fe-Zn reactivity. Below 700°C, a thin layer of Fe oxide which existed on the steel surface deteriorated Fe-Zn reactivity. This thin oxide layer was reduced by Si and Mn diffused from the steel substrate. As the temperature increased from 600°C to 700°C, Fe-Zn reactivity improved due to the formation of reduced iron on the steel surface. In the case of the cold-rolled steel, the same selective oxidation behavior and reduction mechanism of the Fe oxide were also confirmed. As a result, the Fe-Zn reactivity of the cold-rolled steel showed the same behavior as that of the hot-rolled steel. However, the Fe-Zn reactivity of the cold-rolled steel improved at a lower temperature than that of the hot-rolled steel. This can be explained by the faster diffusion rates of Si and Mn in the cold-rolled steel than in the hot-rolled steel. At the surface of the cold-rolled steel, reduction of the Fe oxide layer was promoted and the Fe-Zn reactivity of the cold-rolled steel was improved at a lower temperature.
View full abstractDownload PDF (3079K) Full view HTML
-
Tomohiko Omura, Kota Tomatsu, Yuji Sakiyama, Kazuki Sugita, Masataka M ...2018 Volume 104 Issue 11 Pages 655-663
Published: 2018
Released on J-STAGE: October 31, 2018
JOURNAL OPEN ACCESS FULL-TEXT HTMLThe effects of hydrogen charge on the formation of vacancy-type defects in low alloy martensitic steel were investigated by using positron lifetime spectroscopy. During slow strain rate tensile tests (SSRT), dislocation and vacancy were introduced and increased with increasing tensile strains. Vacancy clustering was significantly promoted by hydrogen charge, although there was no remarkable difference in the dislocation density and mono-vacancy equivalent vacancy density between hydrogen-charged and uncharged samples under the same tensile strains. Increase in hydrogen concentration in the steel promoted vacancy clustering. Preferential conditions for the vacancy clustering showed a good agreement with the conditions for significant elongation loss by SSRT, implying the vacancy clustering has an big effect on hydrogen embrittlement. In the case of constant load tests under applied stress in elastic region, no increase in vacancy and dislocation density was observed. This implies plastic deformation is necessary for the formation of vacancy-type defects even under hydrogen charging.
View full abstractDownload PDF (2004K) Full view HTML -
Hiroshi Yuki, Shota Takada2018 Volume 104 Issue 11 Pages 664-672
Published: 2018
Released on J-STAGE: October 31, 2018
Advance online publication: July 21, 2018JOURNAL OPEN ACCESS FULL-TEXT HTMLThe effect of prior cold deformation on the dissolution speed of carbide in SUJ2 bearing steel subjected to induction heating austenitization at 920°C was examined. Microscopic observations using scanning electron microscopy and transmission electron microscopy, in addition to crystallinity measurements using powder X-ray diffraction, found no trace of the cold deformation effect in the carbide and few crystal defects in the spheroidal carbide particles of an as-received specimen. However, it is important to note that the cold deformation resulted in a refinement of austenite grains during austenitization, which accelerated the dissolution of chromium from the carbide compared to an unprocessed specimen. This led to enhancement of the resultant carbon diffusion in a matrix.
The volume fraction of carbide present at grain boundaries was estimated assuming a simple geometrical model. The relationship between the holding time and the volume fraction of undissolved carbide was derived based on a bulk diffusion model of chromium into an austenite matrix. Experimental results on the dissolution speed of carbide were qualitatively explained by considering the combination of grain boundary diffusion and bulk diffusion. The austenitization behavior of SUJ2 steel is remarkably influenced by the microstructure, in particular for short austenitization times.
View full abstractDownload PDF (2455K) Full view HTML -
Taketo Maejima, Mitsuharu Yonemura, Kaori Kawano, Goro Miyamoto2018 Volume 104 Issue 11 Pages 673-682
Published: 2018
Released on J-STAGE: October 31, 2018
Advance online publication: June 19, 2018JOURNAL OPEN ACCESS FULL-TEXT HTMLStrengthening effect by microalloyed vanadium (V) on eutectoid pearlite steel has been investigated from the perspective of nano-precipitation and lattice strain. 0.2% proof stress of specimens, isothermally transformed at 873 K, increases around 160-170 MPa with 0.1% V addition. However, interphase precipitation of vanadium carbide (VC), regarded as the principal strengthening factor, has not been detected by transmission electron microscopy or 3D atom probe microscopy (3D-AP). On the other hand, lattice strain in lamellar ferrite, analyzed by broadening of X-ray diffraction peak, has clear correlation with proof stress. The lattice strain data of 0.1% V added pearlite specimens are plotted on the same correlation line as of V free ones. In addition, elemental map by 3D-AP shows that vanadium atoms concentrate in lamellar cementite rather than ferrite, which could change cementite lattice parameters and gain ferrite/cementite misfit causing lattice strain increment. These results reveal that microalloyed V influences not only precipitation of VC in lamellar ferrite, but also the lattice strain increment in pearlite lamellar. As far as pearlite steels containing at most 0.1% V, lattice strain is considered to be the major factor of their yield behaviors. Furthermore, 0.1% V addition has not enhanced work-hardening behavior as notably as estimated by Ashby’s work-hardening theory of dispersion-hardened crystals. Therefore, VC precipitation should not necessary for V strengthening effect on pearlite steel.
View full abstractDownload PDF (2118K) Full view HTML -
Setsuo Takaki, Takuro Masumura, Fulin Jiang, Toshihiro Tsuchiyama2018 Volume 104 Issue 11 Pages 683-688
Published: 2018
Released on J-STAGE: October 31, 2018
JOURNAL OPEN ACCESS FULL-TEXT HTMLA low carbon ferritic steel (Fe-0.0056%C) was used for the estimation of dislocation density. Annealed steel sheet with the ferrite grain size of 50 μm was subjected to cold rolling up to 80% thickness reduction and then provided to the X-ray diffraction analysis. Dislocation density was evaluated by the Modified Williamson-Hall / Warren-Averbach method. It was confirmed in cold rolled specimens that the equation; σy [GPa] = 0.05+18 ρ /109 stands up in the relation between yield stress σy and dislocation density ρ [m–2]. On the other hand, the values of parameter φ in the Modified Williamson-Hall equation were accurately estimated in each cold rolled specimen and it was found that the equation ρ[m–2] = 4.5×1019φ2 stands up between φ and ρ. In addition, it is suggested that the screw dislocation component decreases with cold rolling and, in specimens with cold rolling more than 40%, dislocations are of almost full edge dislocation.
 View full abstractDownload PDF (891K) Full view HTML
View full abstractDownload PDF (891K) Full view HTML -
Makoto Akama, Akira Kiuchi2018 Volume 104 Issue 11 Pages 689-698
Published: 2018
Released on J-STAGE: October 31, 2018
Advance online publication: July 24, 2018JOURNAL OPEN ACCESS FULL-TEXT HTMLFatigue tests were performed to obtain co-planar and branch crack growth rates on rail and wheel steel under non-proportional mixed mode I/III cycles. In the experiments, sequential and overlapping mode I and mode III cycles that simulated the load experienced by the rolling contact fatigue cracks were applied to the crack in cylindrical specimens made of rail and wheel steel. Experiments showed that a long co-planar crack could be produced under certain loading conditions. Based on the fracture surface observations by SEM and the results of FEA, the long co-planar crack growth is thought to be driven mainly by mode III loading and the role of mode I is an assistant, keeping the crack face opened. It was observed that the cracks were apt to branch when the strength of the material increased. It was also observed that the crack branched when the degree of overlap between the mode I and mode III cycles increased. We proposed the equivalent stress intensity factor range for branch crack that can consider the crack face contact and successfully regressed the crack growth rate data. Comparing the fracture surfaces and the co-planar crack growth rates data under non-proportional mixed mode I/III loading with that under I/II loading, it is found that the mechanism of shear mode crack growth is essentially the same regardless of whether the main driving force is in-plane shear or out-of-plane shear.
 Comparison of crack growth rates against ΔKs for RP, RF and WT (ΔKIII/ΔKI=1.0). Fullsize ImageView full abstractDownload PDF (3124K) Full view HTML
Comparison of crack growth rates against ΔKs for RP, RF and WT (ΔKIII/ΔKI=1.0). Fullsize ImageView full abstractDownload PDF (3124K) Full view HTML -
Hiroshi Yuki2018 Volume 104 Issue 11 Pages 699-707
Published: 2018
Released on J-STAGE: October 31, 2018
Advance online publication: July 21, 2018JOURNAL OPEN ACCESS FULL-TEXT HTMLThe applicability of the finite element method coupled with electromagnetic field analysis and heat conduction analysis to optimization of induction heating of a bearing raceway was assessed. In order to obtain temperature uniformity in a heated raceway, precise design of the coil arrangement and heating schedule are required. It is important to know how the internal temperature changes with time for the development stage.
The one-dimensional skin current model predicts that the temperature difference between the outer and inner surfaces decreases and that the temperature becomes uniform as the heating region moves inward when the outer surface temperature nears the Curie point. This phenomenon was reproduced in simulations and experiments for a ring sample, but it depended on the balance between the thickness of the ring and the penetration depth of the electromagnetic field. It is necessary to set an appropriate rate of temperature increase and frequency for the power source according to the thickness of ring.
High accuracy analysis was possible by using temperature-dependent B-H characteristics when a heated ring was in the ferromagnetic state. However, in the paramagnetic state, the agreement between simulation and experiment became worse. This may be due to the high rate of temperature increase used in this study, which suggests that the shift in the transformation point to high temperature must be taken into account.
View full abstractDownload PDF (2069K) Full view HTML
-
Yasutaka Ta, Takeru Hoshino, Hiroyuki Tobo, Keiji Watanabe2018 Volume 104 Issue 11 Pages 708-716
Published: 2018
Released on J-STAGE: October 31, 2018
Advance online publication: July 19, 2018JOURNAL OPEN ACCESS FULL-TEXT HTMLA continuous blast furnace slag solidification process was developed to promote the use of air-cooled slag coarse aggregate for concrete. In this process, molten slag can be solidified in only 120 seconds, and the thickness of the slag is about 25 mm. After crushing the slag, the water absorption ratio is much lower than that achieved in the past because gas generation is suppressed. With this apparatus, most of the slag is crystalline, but part of the slag has a glassy surface. Therefore, EPMA and XRD were used to study the glass transition phenomenon. It found that the thickness of the glass layer is about 2 mm. To discuss the glass transition and crystallization phenomena, the thermal history was simulated by heat transfer analysis. The results clarified the fact that all the slag on the mold has a glassy surface layer of about 2 mm, and good agreement between the calculation and experimental data concerning the layer was obtained. It was also shown that most of the slag crystallizes in the slag pit because the temperature inside the piled slags rises to more than 1173 K. The measured slag temperature and calculated temperature were also in good agreement.
View full abstractDownload PDF (7643K) Full view HTML
-
Takuro Masumura, Setsuo Takaki, Fulin Jiang, Toshihiro Tsuchiyama2018 Volume 104 Issue 11 Pages 717-719
Published: 2018
Released on J-STAGE: October 31, 2018
JOURNAL OPEN ACCESS FULL-TEXT HTMLIn Modified Williamson-Hall / Warren-Averbach (MWH/WA) method, the estimation of parameter α is of vital importance for obtaining other parameters accurately. Almost all researchers use the procedure for determining parameter α proposed by Ungár et al. (route 1), which includes the rough approximation. On the other hand, Takebayashi et al. suggested an improved procedure (route 2) which could give more precise results. By comparing above two routes, it is found that route 1 can result in overestimated (about twice) α values and significantly impacts further analysis in MWH/WA method due to ignored terms. Since the difference of α value affects the crystallite size D and parameter φ, the application of route 2 is recommended in MWH/WA method.
View full abstractDownload PDF (556K) Full view HTML
- |<
- <
- 1
- >
- >|