
- Issue 16 Pages 2157-
- Issue 15 Pages 1805-
- Issue 14 Pages 1613-
- Issue 13 Pages S1141-
- Issue 12 Pages S1037-
- Issue 11 Pages 1443-
- Issue 10 Pages 1273-
- Issue 9 Pages 1077-
- Issue 8 Pages 917-
- Issue 7 Pages 751-
- Issue 6 Pages 585-
- Issue 5 Pages S389-
- Issue 4 Pages S1-
- Issue 3 Pages 403-
- Issue 2 Pages 233-
- Issue 1 Pages 19-
- Issue 16 Pages 2153-
- Issue 15 Pages 1977-
- Issue 14 Pages 1813-
- Issue 13 Pages S1285-
- Issue 12 Pages S1043-
- Issue 11 Pages 1667-
- Issue 10 Pages 1481-
- Issue 9 Pages 1231-
- Issue 8 Pages 891-
- Issue 7 Pages 711-
- Issue 6 Pages 538-
- Issue 5 Pages S407-
- Issue 4 Pages S1-
- Issue 3 Pages 347-
- Issue 2 Pages 173-
- Issue 1 Pages 14-
- Issue 16 Pages 1837-
- Issue 15 Pages 1711-
- Issue 14 Pages 1569-
- Issue 13 Pages S1205-
- Issue 12 Pages S1034-
- Issue 11 Pages 1423-
- Issue 10 Pages 1269-
- Issue 9 Pages 1059-
- Issue 8 Pages 925-
- Issue 7 Pages 775-
- Issue 6 Pages 627-
- Issue 5 Pages S287-
- Issue 4 Pages S1-
- Issue 3 Pages 301-
- Issue 2 Pages 147-
- Issue 1 Pages 12-
- Issue 16 Pages 2179-
- Issue 15 Pages 1795-
- Issue 14 Pages 1631-
- Issue 13 Pages S1053-
- Issue 12 Pages S1023-
- Issue 11 Pages 1501-
- Issue 10 Pages 1315-
- Issue 9 Pages 987-
- Issue 8 Pages 767-
- Issue 7 Pages 621-
- Issue 6 Pages 473-
- Issue 5 Pages S305-
- Issue 4 Pages S1-
- Issue 3 Pages 299-
- Issue 2 Pages 151-
- Issue 1 Pages 16-
- Issue 16 Pages 1945-
- Issue 15 Pages 1699-
- Issue 14 Pages 1531-
- Issue 13 Pages S1055-
- Issue 12 Pages S1013-
- Issue 11 Pages 1367-
- Issue 10 Pages 1215-
- Issue 9 Pages 1087-
- Issue 8 Pages 887-
- Issue 7 Pages 721-
- Issue 6 Pages 507-
- Issue 5 Pages S317-
- Issue 4 Pages S1-
- Issue 3 Pages 343-
- Issue 2 Pages 187-
- Issue 1 Pages 17-
- Issue 16 Pages 2405-
- Issue 15 Pages 2067-
- Issue 14 Pages 1865-
- Issue 13 Pages 1675-
- Issue 12 Pages S1055-
- Issue 11 Pages S1015-
- Issue 10 Pages 1479-
- Issue 9 Pages 1129-
- Issue 8 Pages 895-
- Issue 7 Pages 711-
- Issue 6 Pages 545-
- Issue 5 Pages S325-
- Issue 4 Pages S1-
- Issue 3 Pages 369-
- Issue 2 Pages 193-
- Issue 1 Pages 16-
- Issue 16 Pages 2573-
- Issue 15 Pages 2261-
- Issue 14 Pages 2073-
- Issue 13 Pages S1111-
- Issue 12 Pages S1001-
- Issue 11 Pages 1867-
- Issue 10 Pages 1657-
- Issue 9 Pages 1409-
- Issue 8 Pages 1043-
- Issue 7 Pages 841-
- Issue 6 Pages 649-
- Issue 5 Pages S415-
- Issue 4 Pages S1-
- Issue 3 Pages 431-
- Issue 2 Pages 225-
- Issue 1 Pages 3-
- |<
- <
- 1
- >
- >|
-
2020 Volume 106 Issue 11 Pages Cover-
Published: November 01, 2020
Released on J-STAGE: October 31, 2020
JOURNAL OPEN ACCESSDownload PDF (658K) -
2020 Volume 106 Issue 11 Pages Contents-
Published: November 01, 2020
Released on J-STAGE: October 31, 2020
JOURNAL OPEN ACCESSDownload PDF (4205K) -
2020 Volume 106 Issue 11 Pages Editorial-
Published: November 01, 2020
Released on J-STAGE: October 31, 2020
JOURNAL OPEN ACCESSDownload PDF (226K)
-
Kohichi Isobe2020 Volume 106 Issue 11 Pages 777-787
Published: 2020
Released on J-STAGE: October 31, 2020
Advance online publication: July 21, 2020JOURNAL OPEN ACCESS FULL-TEXT HTMLImportant large components such as rotors for power generation steam turbines, pressure vessels and reaction vessels are manufactured by the ingot casting. However, it is difficult to manufacture the materials with sufficient properties due to macro-segregation of ingots.
For the purpose of developing effective and versatile macro-segregation countermeasures in the casting of large steel ingots for manufacturing large parts for power plants, the insert casting in vacuum atmosphere, in which a core material with the same composition as the base steel is placed at the center of the steel ingot, was studied. The effectiveness of the proposed insert casting as a macro-segregation countermeasure was verified in laboratory experiments. In addition, it has been clarified that good bonding between the core material and the base material can be realized even under conditions where bonding by normal insert casting in air atmosphere is difficult.
The smelting behavior of the core material and the solidification behavior of the molten steel in the experiments of macro-segregation reproduction casting and the insert casting were analyzed by the direct finite difference method. The mechanism by which this method suppresses the formation of macro-segregation and the solidification conditions for the suppression, the reasons and conditions for good bonding in this insert casting are clarified by the analyses.
Furthermore, in the experiments of insert casting, the cause of internal cracks generated in the solidified shell on the core material surface was considered, and guidelines for preventing the internal cracks were presented.
 View full abstractDownload PDF (10365K) Full view HTML
View full abstractDownload PDF (10365K) Full view HTML
-
Shigeto Yamasaki, Masatoshi Mitsuhara, Hideharu Nakashima2020 Volume 106 Issue 11 Pages 788-798
Published: 2020
Released on J-STAGE: October 31, 2020
Advance online publication: June 26, 2020JOURNAL OPEN ACCESS FULL-TEXT HTMLIn this study, the relation between the change of the magnetic properties and the creep strength with the addition of Co was investigated for ferritic steel containing 15 mass%Cr. Co addition up to 6 mass% hardly contributes to solid solution strengthening and precipitation strengthening at room temperature. However, in the range of 650ºC to 750ºC, it was confirmed that the addition of Co was effective for the creep strengthening because the steel with a larger amount of Co had higher creep strength. This creep strengthening is explained by a reduction in the diffusion rate associated with a change in magnetic properties by Co addition. The increase of the volume magnetization of the steel with increase of Co amount in the temperature range from room temperature to about 800ºC was confirmed. Comparing the difference in volume magnetization and the ratio of creep strain rate for steels with different amounts of Co, a clear correlation was found between the values. That is, at the temperature at which the difference in volume magnetization becomes maximum, the peak of the creep strain rate ratio was observed. This result is explained as follows. In a low temperature region where the magnetization is large or in a high temperature region above the Curie point of both steels, there is no significant difference in the creep strength between them. However, in the temperature where one steel loses the ferromagnetism but the other steel maintains ferromagnetism, a significant difference in the creep strength was observed.
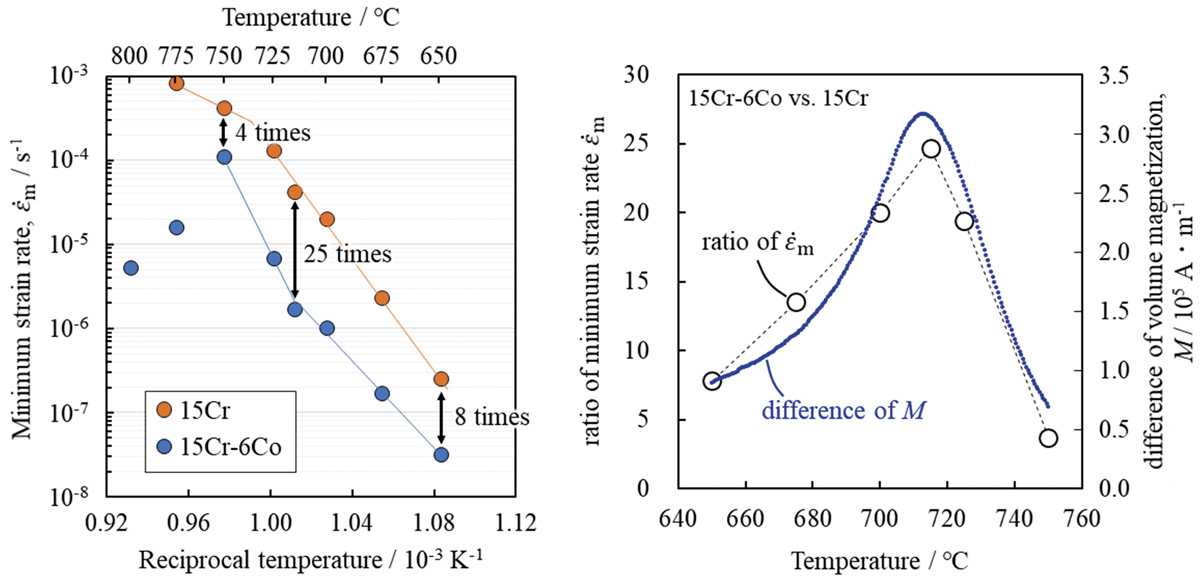 View full abstractDownload PDF (7668K) Full view HTML
View full abstractDownload PDF (7668K) Full view HTML -
Yoshiyuki Furuya2020 Volume 106 Issue 11 Pages 799-806
Published: 2020
Released on J-STAGE: October 31, 2020
JOURNAL OPEN ACCESS FULL-TEXT HTMLGigacycle fatigue tests were conducted on high-strength steels whose Mn and S contents were so high as to be close to upper limits of the JIS standard. Three types of materials were prepared with different working ratios. The fatigue strengths were in order of the working ratios, indicating that the high working ratios improved the fatigue strength. All specimens ended in internal fractures mostly originating from MnS. The MnS observed on the fracture surface was largely elongated in the cases of the materials with high wording ratios. ODAs (Optically Dark Areas) were then observed at around the center of the elongated MnS, meaning that small internal cracks were formed there. Although the shapes of the MnS were very complicated, the sizes were approximated by using ellipses which covered the whole MnS. When the MnS was largely elongated, the length l was cut off at four times of the width W, i.e., l = 4W, based on the results of Makino’s fracture mechanics analysis. The measured MnS sizes were applied to analysis by using a previously derived prediction, comparing the effects of MnS with those of oxides. As the result, it was suggested that the MnS was more harmful to the fatigue strength than the oxides. This was probably attributable to the properties of the MnS, while too large number of the MnS possibly reduced the fatigue strength.
 View full abstractDownload PDF (3086K) Full view HTML
View full abstractDownload PDF (3086K) Full view HTML
-
Keisuke Kinoshita, Yasuaki Tanaka, Hideaki Sawada, Kohsaku Ushioda2020 Volume 106 Issue 11 Pages 807-815
Published: 2020
Released on J-STAGE: October 31, 2020
JOURNAL OPEN ACCESS FULL-TEXT HTMLThe carbon solubility limit in bcc-iron in equilibrium with cementite at 973 K was estimated by exploiting the age-hardening behavior of a quenched specimen at 323 K and Bayesian inference. The cold-rolled steel containing 0.045 wt% carbon was subjected to decarburization annealing at 973 K in a salt bath for different times up to 10 hours; subsequently, they were water-quenched. These specimens showed carbon contents of 0.005 to 0.045 wt% depending on the annealing time. The quenched specimens were kept at 323 K in a silicone oil bath and the Vickers hardness was sequentially measured to evaluate an age-hardening curve. The maximum hardness of each aging curve showed the following changes with carbon content: when the carbon content decreased from 0.045 wt%, the hardness showed a constant value up to certain carbon content (Region I), decreased with further decreases in the carbon content (Region II), and finally showed a constant value again. Bayesian inference for the above relation was performed by a Markov chain Monte Carlo method. The boundary between Region I and Region II was rigorously estimated. The 95% confidence interval at the boundary between Region I and Region II was determined to be 0.023 ± 0.003 wt%. Based on the strengthening mechanism for each region, the above value was inferred to be the solubility limit of carbon in ferrite equilibrated with cementite at 973 K.
 View full abstractDownload PDF (1429K) Full view HTML
View full abstractDownload PDF (1429K) Full view HTML -
Yasuhito Kawahara, Ryo Teranishi, Chikako Takushima, Jun-ichi Hamada, ...2020 Volume 106 Issue 11 Pages 816-825
Published: 2020
Released on J-STAGE: October 31, 2020
JOURNAL OPEN ACCESS FULL-TEXT HTMLThe stacking-fault energies (SFE) were measured by weak-beam TEM method, and deformation mechanisms in room and high temperature were characterized in terms of the effect of nitrogen addition on SFE in Si-added austenitic stainless steel (Fe-19%Cr-13%Ni-0.05%C-3%Si-x%N). Nitrogen addition resulted in decrease of SFE and changing the dislocation configuration from tangled to planar in room temperature. In high temperature, nitrogen addition resulted in the increase of dislocation density in the subboundary by the decrease of SFE. These results indicated that addition of nitrogen contributed to room and high temperature strength because of the decrease of SFE.
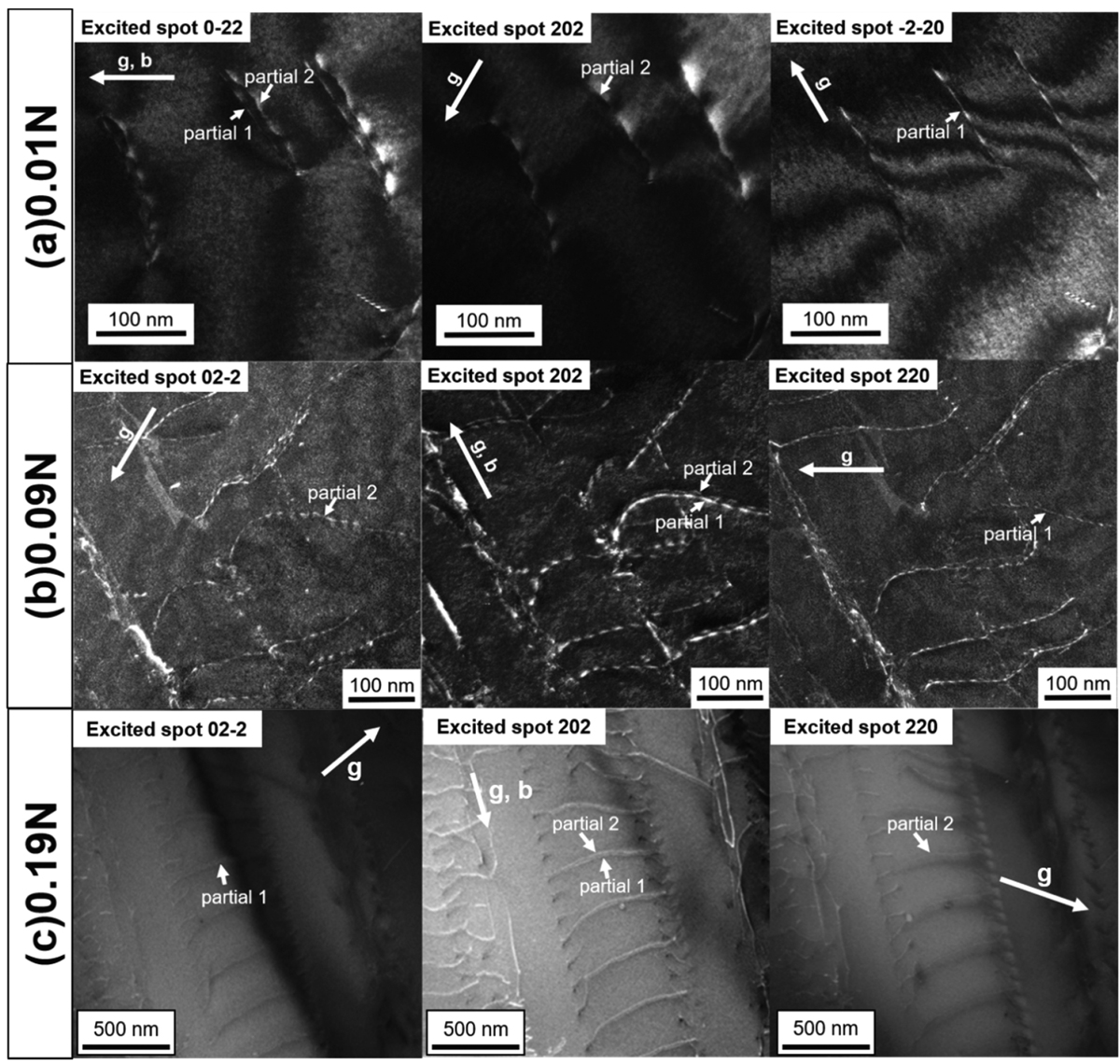 View full abstractDownload PDF (3175K) Full view HTML
View full abstractDownload PDF (3175K) Full view HTML -
Takuro Masumura, Shohei Uranaka, Kyosuke Matsuda, Setsuo Takaki, Toshi ...2020 Volume 106 Issue 11 Pages 826-834
Published: 2020
Released on J-STAGE: October 31, 2020
JOURNAL OPEN ACCESS FULL-TEXT HTMLTo calculate the dislocation density of tempered low-carbon martensitic steels (Fe-0.15%C alloy) by the direct-fitting/modified Williamson-Hall (DF/mWH) method, the unknown parameter A in tempered martensite was investigated. In the DF/mWH method, the dislocation density ρ is defined as ρ=2φ2/(πA2b2). Here, φ and b correspond to the slope of the DF/mWH plot and the magnitude of the Burgers vector, respectively. In low-carbon martensitic steels tempered at 573 - 873 K, φ and ρ were estimated by the DF/mWH method and the modified Williamson-Hall/Warren-Averbach method, respectively. As a result, these parameters decreased with increasing tempering temperature. By substituting φ and ρ in the above equation, the parameter A can be obtained as a function of the tempering parameter λ as follows:
A = –1.53 × 10−5 × λ + 0.982
The parameter A in low-carbon martensitic steels tempered at 573 - 873 K for 18 ks was calculated to be 0.7 - 0.8. Using the parameters A and φ obtained by the DF/mWH method, dislocation density ρ can be easily estimated.
 View full abstractDownload PDF (1873K) Full view HTML
View full abstractDownload PDF (1873K) Full view HTML -
Estimation of Solute Carbon Concentration by Electrical Resistivity Measurement in Martensitic SteelTakuro Masumura, Taiga Taniguchi, Shohei Uranaka, Issei Hirashima, Tos ...2020 Volume 106 Issue 11 Pages 835-843
Published: 2020
Released on J-STAGE: October 31, 2020
JOURNAL OPEN ACCESS FULL-TEXT HTMLElectrical resistivity of low-carbon martensitic steels was measured to estimate the carbon concentration in solid solution. Since electrical resistivity is influenced not only by solute carbon but also by substitutional elements, lattice defects and second phase, the effects of these factors need to be subtracted from total electrical resistivity, in order to obtain the accurate solute carbon concentration by this method. As a result, the effects of dislocations and grain boundaries were much smaller than that of solute elements, being only 1 - 2% of the measured electrical resistivity, in martensitic steel. On the other hand, substitutional elements and retained austenite were found to be significantly effective. By subtracting these effects from the measured value, the change in electrical resistivity due to solute carbon, Δρsol.C, could be formulated as a function of the carbon concentration in solid solution of martensite, Csol, as follows:
Δρsol.C [mΩmm] = 0.25 × Csol [mass%]
The estimated solute carbon concentration was confirmed to correspond to the directly measured value by atom probe tomography.
 View full abstractDownload PDF (3513K) Full view HTML
View full abstractDownload PDF (3513K) Full view HTML
-
Daisuke Maruoka, Taichi Murakami, Eiki Kasai2020 Volume 106 Issue 11 Pages 844-850
Published: 2020
Released on J-STAGE: October 31, 2020
JOURNAL OPEN ACCESS FULL-TEXT HTMLSelf-healing function in ceramic-based composites is one of unique characteristics to improve the strength reliability. In this study, such a strength recovery phenomenon of 5 vol% Fe particle/2 mol% Y2O3/Mullite composite was investigated at various heat treatment temperatures.
Vickers indentation were conducted with dense rectangular samples. Samples were heat treated at 700 -1000°C for 1 h in air or Ar-3%H2 atmosphere and then 3-point bending test was carried out at room temperature. Peaks of hematite were identified in the XRD profiles of the samples after heat treatment in air. Surface cracks mostly disappeared due to formation of the oxidation product, i.e., particle-like hematite formed by the heat treatment at 900°C in air for 1 h. Further, needle-like hematite formed and penetrated into the sample surface by the heat treatment at 1000°C. While, oxide formation was not observed for the sample treated in Ar-3%H2 atmosphere. The bending strengths of the as-polished and as-cracked samples were 334 and 99 MPa, respectively. Recovery rate is increased with increasing heat treatment temperature up to 900°C. Heat-treatment at 900°C for 1 h resulted in a recovery of the bending strength up to 262 MPa. While, bending strengths of the samples treated at 1000°C for 1 h air was 204 MPa. It appears that needle like hematite degrades mechanical properties of the samples. Bending strengths of the samples treated at 900°C for 1 h air was 112 MPa as well as as-cracked sample due to no formation of oxidation product.
 View full abstractDownload PDF (1247K) Full view HTML
View full abstractDownload PDF (1247K) Full view HTML
-
Seiji Yokoyama, Masahiro Seyama, Shunya Yasuike, Junji Sasano2020 Volume 106 Issue 11 Pages 851-857
Published: 2020
Released on J-STAGE: October 31, 2020
JOURNAL OPEN ACCESS FULL-TEXT HTMLIt has been suggested that the eluate from steelmaking slag has a buffering effect. However the mechanism of the buffering action has not been elucidated. The slag eluate contains various elements such as Ca, Mg, Si, Al, and so on. CO2 in air influence pH. Therefore, in this study, we performed a model experiment in which an aqueous solution of sulfuric acid or nitric acid, which is a representative acid of the acid rain, was added to an aqueous solution containing Ca or Mg in Ar. Its solution was prepared from its hydroxide and ultrapure water. The pH behavior of the solution was investigated to clarify the factors which determined the pH. When the acid solution was added to the solution containing Ca or Mg, pH decreased gradually. At a certain amount of addition of the acid solution, the pH decreased steeply, and, subsequently, decreased gradually. The pH of the solution containing Ca or Mg was determined by the charge balance of ions in the solution. Those two gradual decreases in pH corresponded buffering action. This was caused by the reaction of water dissociation. The equivalence point was the point where concentration of Ca2+ or Mg2+ was equal to the concentration of SO42–, or twice that of NO3–. The pH at this point was seven. The sharp pH change occurred at pH of approximately seven.
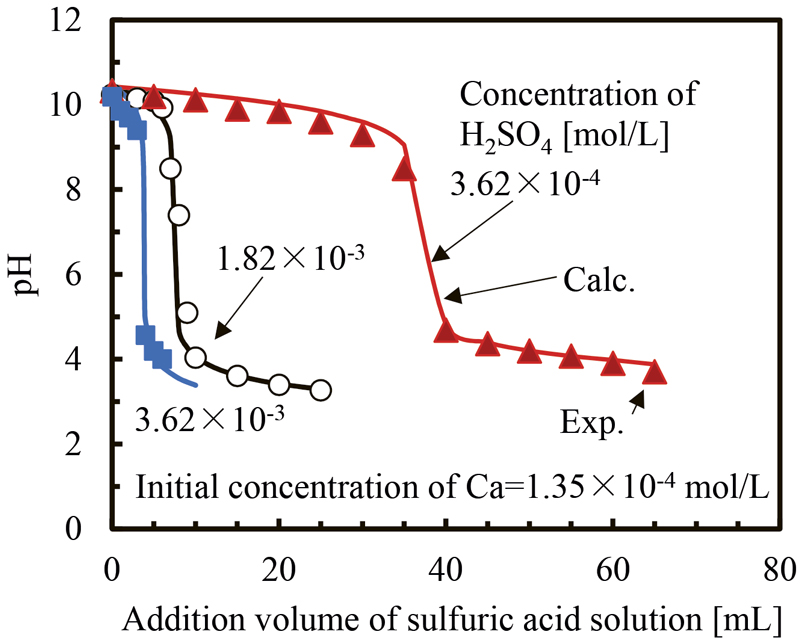 View full abstractDownload PDF (1462K) Full view HTML
View full abstractDownload PDF (1462K) Full view HTML -
Hiroshi Sato, Keiji Okumura2020 Volume 106 Issue 11 Pages 858-866
Published: 2020
Released on J-STAGE: October 31, 2020
Advance online publication: July 01, 2020JOURNAL OPEN ACCESS FULL-TEXT HTMLThe chlorination and volatilization behavior of zinc was investigated by the reaction between ZnO, ZnFe2O4 and CaCl2. Experiments were conducted in a nitrogen atmosphere by using horizontal electric furnace. As a result of changing the ratio of zinc oxide to zinc ferrite at 1000ºC, it was expected that ZnO and ZnFe2O4 were chloride volatilized at the same time. In the kinetic study at 950ºC-1030ºC, Jander’s equation showed the experimental result well in the reaction system of this study, and the activation energy was 237 ± 18 kJ / mol. It was also found that carbon influenced the reaction product determination. FeO and CaFe2O4 were formed in the presence of carbon, and Ca2Fe2O5 was formed in the absence of carbon.
 View full abstractDownload PDF (1423K) Full view HTML
View full abstractDownload PDF (1423K) Full view HTML
-
Setsuo Takaki, Takuro Masumura, Toshihiro Tsuchiyama2020 Volume 106 Issue 11 Pages 867-870
Published: 2020
Released on J-STAGE: October 31, 2020
JOURNAL OPEN ACCESS FULL-TEXT HTMLIn pure iron, the Young’s modulus ratio E100/Ehkl in single crystal is expressed as follows as a function of the orientation parameter Γ: E100/Ehkl=1–1.6063Γ. Under the condition that this relationship stands up on commercial ferritic and martensitic steels with various chemical composition and Poisson’s ratio is also identical at 0.291, the elastic stiffness cij was calculated as follows in connection with Young’s modulus Ep of poly crystal ferritic steels: c11=1.0970Ep, c12=0.6477Ep, c44=0.5424Ep. As a result, the elastic stiffness ratio c12/c44 and the anisotropy parameter Ai (=2c44/(c11–c12)) are given at 1.194 and 2.414 independent on the value of Ep, respectively. The contrast factor Ch00 and the parameter q, that are required for the dislocation analysis by the modified Williamson-Hall method, are represented as follows as a function of the screw component S in dislocation: Ch00=0.263+0.044×S and q=1.382+1.27×S.
 View full abstractDownload PDF (593K) Full view HTML
View full abstractDownload PDF (593K) Full view HTML
- |<
- <
- 1
- >
- >|