
- Issue 16 Pages 2157-
- Issue 15 Pages 1805-
- Issue 14 Pages 1613-
- Issue 13 Pages S1141-
- Issue 12 Pages S1037-
- Issue 11 Pages 1443-
- Issue 10 Pages 1273-
- Issue 9 Pages 1077-
- Issue 8 Pages 917-
- Issue 7 Pages 751-
- Issue 6 Pages 585-
- Issue 5 Pages S389-
- Issue 4 Pages S1-
- Issue 3 Pages 403-
- Issue 2 Pages 233-
- Issue 1 Pages 19-
- Issue 16 Pages 2153-
- Issue 15 Pages 1977-
- Issue 14 Pages 1813-
- Issue 13 Pages S1285-
- Issue 12 Pages S1043-
- Issue 11 Pages 1667-
- Issue 10 Pages 1481-
- Issue 9 Pages 1231-
- Issue 8 Pages 891-
- Issue 7 Pages 711-
- Issue 6 Pages 538-
- Issue 5 Pages S407-
- Issue 4 Pages S1-
- Issue 3 Pages 347-
- Issue 2 Pages 173-
- Issue 1 Pages 14-
- Issue 16 Pages 1837-
- Issue 15 Pages 1711-
- Issue 14 Pages 1569-
- Issue 13 Pages S1205-
- Issue 12 Pages S1034-
- Issue 11 Pages 1423-
- Issue 10 Pages 1269-
- Issue 9 Pages 1059-
- Issue 8 Pages 925-
- Issue 7 Pages 775-
- Issue 6 Pages 627-
- Issue 5 Pages S287-
- Issue 4 Pages S1-
- Issue 3 Pages 301-
- Issue 2 Pages 147-
- Issue 1 Pages 12-
- Issue 16 Pages 2179-
- Issue 15 Pages 1795-
- Issue 14 Pages 1631-
- Issue 13 Pages S1053-
- Issue 12 Pages S1023-
- Issue 11 Pages 1501-
- Issue 10 Pages 1315-
- Issue 9 Pages 987-
- Issue 8 Pages 767-
- Issue 7 Pages 621-
- Issue 6 Pages 473-
- Issue 5 Pages S305-
- Issue 4 Pages S1-
- Issue 3 Pages 299-
- Issue 2 Pages 151-
- Issue 1 Pages 16-
- Issue 16 Pages 1945-
- Issue 15 Pages 1699-
- Issue 14 Pages 1531-
- Issue 13 Pages S1055-
- Issue 12 Pages S1013-
- Issue 11 Pages 1367-
- Issue 10 Pages 1215-
- Issue 9 Pages 1087-
- Issue 8 Pages 887-
- Issue 7 Pages 721-
- Issue 6 Pages 507-
- Issue 5 Pages S317-
- Issue 4 Pages S1-
- Issue 3 Pages 343-
- Issue 2 Pages 187-
- Issue 1 Pages 17-
- Issue 16 Pages 2405-
- Issue 15 Pages 2067-
- Issue 14 Pages 1865-
- Issue 13 Pages 1675-
- Issue 12 Pages S1055-
- Issue 11 Pages S1015-
- Issue 10 Pages 1479-
- Issue 9 Pages 1129-
- Issue 8 Pages 895-
- Issue 7 Pages 711-
- Issue 6 Pages 545-
- Issue 5 Pages S325-
- Issue 4 Pages S1-
- Issue 3 Pages 369-
- Issue 2 Pages 193-
- Issue 1 Pages 16-
- Issue 16 Pages 2573-
- Issue 15 Pages 2261-
- Issue 14 Pages 2073-
- Issue 13 Pages S1111-
- Issue 12 Pages S1001-
- Issue 11 Pages 1867-
- Issue 10 Pages 1657-
- Issue 9 Pages 1409-
- Issue 8 Pages 1043-
- Issue 7 Pages 841-
- Issue 6 Pages 649-
- Issue 5 Pages S415-
- Issue 4 Pages S1-
- Issue 3 Pages 431-
- Issue 2 Pages 225-
- Issue 1 Pages 3-
- |<
- <
- 1
- >
- >|
-
2021 Volume 107 Issue 7 Pages Cover-
Published: July 01, 2021
Released on J-STAGE: June 30, 2021
JOURNAL OPEN ACCESSDownload PDF (663K) -
2021 Volume 107 Issue 7 Pages Contents-
Published: July 01, 2021
Released on J-STAGE: June 30, 2021
JOURNAL OPEN ACCESSDownload PDF (2343K) -
2021 Volume 107 Issue 7 Pages Editorial-
Published: July 01, 2021
Released on J-STAGE: June 30, 2021
JOURNAL OPEN ACCESSDownload PDF (205K)
-
Hideo Mizukami, Tomoyuki Kitaura, Yoshihisa Shirai2021 Volume 107 Issue 7 Pages 543-550
Published: 2021
Released on J-STAGE: June 30, 2021
JOURNAL OPEN ACCESS FULL-TEXT HTMLFor multi-component titanium alloy ingots that contain evaporable solute elements, the evaporation behavior of these elements is very important for optimizing the casting conditions. This study examined the evaporation of Al and Sn from molten Ti-6242, an alloy used for aircraft engines, after partial melting in a small electron beam furnace. The amount of Al and Sn evaporation depended on the melting time, there equilibrium vapor pressure of the elements and activity in the molten alloy. The concentration of Al changed in proportion to that of Sn in the molten alloy during evaporation process and the amount of evaporation of Al was twice as Sn. The rate control step of evaporation of Al and Sn were limited by evaporation from the molten alloy surface to the vacuum phase. The liquidus temperature of the alloy melt also changed with the evaporation of Al and Sn. These findings will help predict the amount of solute evaporation from molten titanium alloys and maintain the surface and internal quality of the ingots.
 View full abstractDownload PDF (1495K) Full view HTML
View full abstractDownload PDF (1495K) Full view HTML -
Saori Shinohara, Rie Endo, Takashi Watanabe, Miyuki Hayashi, Hiroshi T ...2021 Volume 107 Issue 7 Pages 551-557
Published: 2021
Released on J-STAGE: June 30, 2021
JOURNAL OPEN ACCESS FULL-TEXT HTMLDensity of oxide scale formed on steel is essential to derive thermal conductivity, which is used for the process simulation for cooling of steel plate in hot-rolling of steelmaking. Thus, the aim of this research was to propose the density determination method for the oxide scale. The samples used were prepared by the high-temperature oxidation of iron plate, consisting of 96% Fe1-xO and 4%Fe3O4, and there were voids in the Fe1-xO layer. The density was determined by four methods based on (1) thickness and mass of oxide scale, (2) the mass balance in the sample, (3) volume balance in the sample, and (4) single crystal density and volume of voids. All of the methods considered the non-stoichiometry of Fe1-xO because the perfect FeO has a much larger density. The density of the oxide scale was firstly determined by assuming that the oxide scale consisted only of Fe1-xO. The methods (1) and (2) did not show reasonable density and reproducibility. The results by methods (3) and (4) showed reasonable values with good reproducibility. The density was then determined again by methods (3) and (4), taking into account the presence of Fe3O4. It is also discussed the effect of density revision on the thermal diffusivity/conductivity determination of the oxide scale.
 View full abstractDownload PDF (985K) Full view HTML
View full abstractDownload PDF (985K) Full view HTML
-
Tomohiro Nishimura, Ryota Matsubayashi, Kohei Morishita, Masato Yoshiy ...2021 Volume 107 Issue 7 Pages 558-565
Published: 2021
Released on J-STAGE: June 30, 2021
Advance online publication: April 02, 2021JOURNAL OPEN ACCESS FULL-TEXT HTMLIt has been realized that a massive-like transformation, in which δ phase (ferrite) transformed to γ phase (austenite) in the solid state during and after solidification, was selected in Fe-C steels. X-ray radiography confirmed that the massive-like transformation also occurred in Fe-18 mass%Cr-Ni alloys with Ni contents of 8, 11, 14 and 20 mass%Ni. According to the equilibrium phase diagram, δ phase is the primary phase in 8 and 11 mass%Ni alloys while γ phase in 14 and 20 mass%Ni alloys. Solidification was always initiated by nucleation of δ phase and consequently fine γ grains were formed by the massive-like transformation in 8 and 11 mass%Ni. On the other hand, nucleation of δ phase as a metastable phase was preferably selected at lower undercoolings (<50 K) in 14 and 20 mass%Ni and consequently the massive-like transformation occurred even in 14 and 20 mass%Ni alloys. Solidification of γ phase can be triggered by nucleation of δ phase followed by the massive-like transformation in the Fe-Cr-Ni with lower Cr/Ni values (the primary γ alloys). Moreover, the present study demonstrates that the massive-like transformation will be commonly observed in Fe-based alloys, in which δ and γ phases are competitive each other from a thermodynamic perspective.
 View full abstractDownload PDF (3283K) Full view HTML
View full abstractDownload PDF (3283K) Full view HTML
-
Yuto Kadowaki, Yuko Yanagihara, Arinori Inagawa, Nobuo Uehara2021 Volume 107 Issue 7 Pages 566-576
Published: 2021
Released on J-STAGE: June 30, 2021
Advance online publication: April 10, 2021JOURNAL OPEN ACCESS FULL-TEXT HTMLTotal iron contents in iron ores have been accurately determined by JIS M 8212, in which iron ions in digested solutions of iron ores are reduced to divalent prior to redox titration. It is necessary for the iron reduction process that no reducing chemicals other than iron(II) in the decomposition solutions must not remain after the reduction with titanium(III). However, the redox reactions concerning the chemical species present in the decomposition solution has not been completely elucidated at the present time. In this paper, the redox reactions that occurred in the decomposition solution during the iron reduction in JIS M 8212 were studied by potentiometry and spectrophotometry under nitrogen atmosphere. The redox reaction of tin(II)/(IV) was very slow, causing significant effects on identifying the end point of the indicator for the iron reduction. The copper chloro-complexes were reduced with titanium(III) at a potential higher than that of indigo carmine used as a redox indicator, so that the reduced copper(I) gave a positive error to the potassium dichromate titration. The pentavalent vanadium was reduced with titanium (III) to form a complex with titanium, which also interfered with the potassium dichromate titration positively. To avoid these interferences, titanium(III) chloride was stoichiometrically added to the reaction mixture after addition of tin(II) chloride under nitrogen atmosphere so as to reduce only iron to divalent prior to the following redox titration. Combination of the proposed protocol with the potassium dichromate titration could successfully determine the iron content of certified reference materials of iron ores.
 View full abstractDownload PDF (3180K) Full view HTML
View full abstractDownload PDF (3180K) Full view HTML
-
Hiroshi Niitani, Yuichi Mitsutake2021 Volume 107 Issue 7 Pages 577-582
Published: 2021
Released on J-STAGE: June 30, 2021
JOURNAL OPEN ACCESS FULL-TEXT HTMLThe inverse heat conduction problem (IHCP) with the Laplace transform technique is an exact solution method, and the surface heat flux and temperature can be obtained explicitly. However, if the temperature dependence of the thermophysical properties cannot be ignored, the nonlinear term of the heat conduction equation cannot be solved, and it is then necessary to approximate it to a linear equation. Therefore, the IHCP analysis error becomes remarkable under a high heat transfer coefficient condition where the temperature gradient near the surface is large. In this study, the converted temperature method was applied to the IHCP with the Laplace transform technique. Although in this method the error due to the temperature dependence of the thermal diffusivity remains, the nonlinear term, which is the main cause of the error, was possible to be solved exactly. When we evaluated this by numerical analysis, under a condition of using 0.08%C Al killed steel and SUS304, it was possible to accurately conduct analysis under a high cooling rate boiling heat transfer problem with a heat transfer coefficient of up to 12 kW/m2K. The standard deviations of the inverse analysis errors of the heat transfer coefficient were 0.096 kW/m2K and 0.156 kW/m2K, respectively. The error was reduced to 1/7 and 1/6, compared to the simple method (linear approximation).
 View full abstractDownload PDF (1436K) Full view HTML
View full abstractDownload PDF (1436K) Full view HTML
-
Makoto Kawamori, Fumio Yuse, Yosuke Fujita, Hideyuki Ikegami2021 Volume 107 Issue 7 Pages 583-593
Published: 2021
Released on J-STAGE: June 30, 2021
Advance online publication: March 08, 2021JOURNAL OPEN ACCESS FULL-TEXT HTMLThe effect of blasting on hydrogen analysis was investigated with the aim of establishing a hydrogen analysis method for precisely measuring hydrogen that entered steel in a corrosive environment. The hydrogen existing states of the specimens blasted under various conditions were analyzed using thermal desorption analysis and the hydrogen visualization method by secondary ion mass spectrometry. The phenomenon of hydrogen entry into steel by blasting was demonstrated for the first time. It should be noted that the effect is remarkable in the case of a specimen with a large specific surface area, and the blasting becomes an inhibitory agent in the measurement of the hydrogen content in steel. The hydrogen source for increasing the hydrogen content due to blasting is mainly the water contained in the abrasive. The mechanism of increasing the hydrogen content in steel by blasting is that the fresh surface of the steel exposed by blasting reacts with the water in the abrasive, which results in the hydrogen generation and entry into steel. Additionally, the water in the abrasive remaining on the steel surface reacts with steel during the thermal desorption analysis to release hydrogen. To suppress the increase of hydrogen content by blasting, it is effective to use abrasive with low water content and to remove rust by repeating a short blasting time in order to suppress the temperature rise of the specimen.
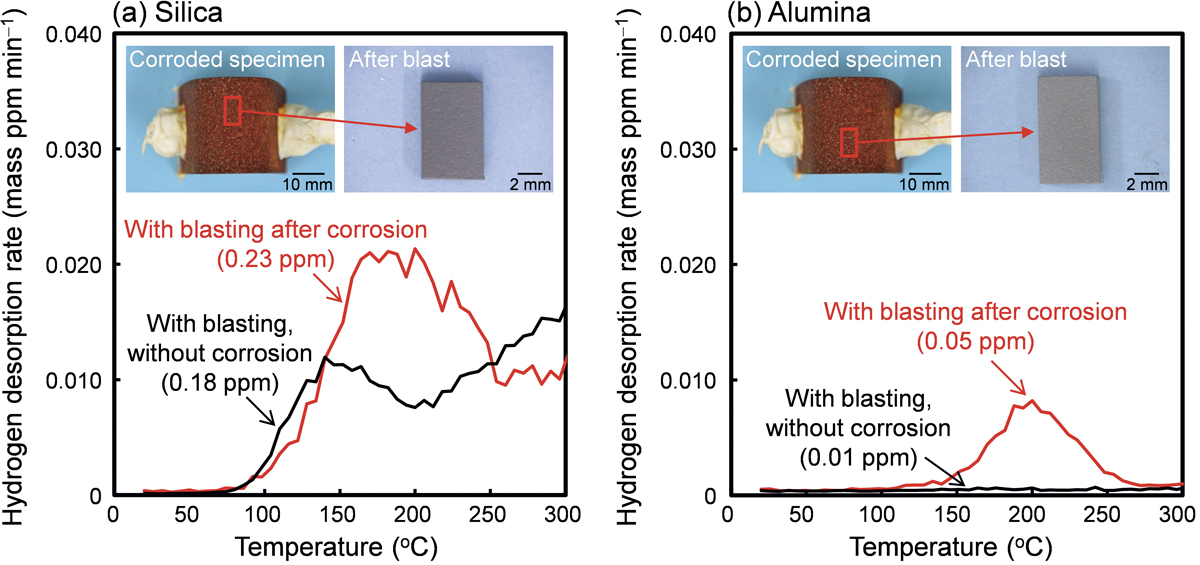 View full abstractDownload PDF (10996K) Full view HTML
View full abstractDownload PDF (10996K) Full view HTML
-
Gaku Kitahara, Aya Tsuji, Takashi Asada, Tomohiro Suzuki2021 Volume 107 Issue 7 Pages 594-603
Published: 2021
Released on J-STAGE: June 30, 2021
Advance online publication: March 05, 2021JOURNAL OPEN ACCESS FULL-TEXT HTMLThe application of high-strength steel sheets in automobiles has been increased to achieve low bodyweight and simultaneously enhance crashworthiness. High-strength steel sheets are susceptible to hydrogen embrittlement and it is essential to evaluate their delayed fracture resistance for appropriate use. Delayed fracture resistance is typically evaluated using the relationship between the amount of diffusible hydrogen and fracture strength obtained from a constant load test and the slow strain rate technique (SSRT). It is difficult to monitor the amount of diffusible hydrogen invading from the environment; the thermal desorption analysis is not a non-destructive analysis to obtain the amount of diffusible hydrogen and diffusible hydrogen is easily desorbed from the specimens. The hydrogen permeation test easily monitors the invasion of diffusible hydrogen. In this study, we evaluated the delayed fracture resistance of high-strength steel sheets using the hydrogen permeability obtained from the hydrogen permeation test. As a result, relationships between hydrogen permeability, mechanical properties obtained from SSRT, and the brittle fracture surface ratio were found to be consistent among various hydrogen invasion conditions, such as under hydrogen charging and corrosive environments. Furthermore, little diffusible hydrogen was detected using the hydrogen permeation test. Thus, delayed fracture resistance obtained from the relationship between hydrogen permeability and its mechanical properties proves the effectiveness of this method.
 View full abstractDownload PDF (3312K) Full view HTML
View full abstractDownload PDF (3312K) Full view HTML
-
Hironari Kubo, Masahiro Nakanishi2021 Volume 107 Issue 7 Pages 604-615
Published: 2021
Released on J-STAGE: June 30, 2021
Advance online publication: March 08, 2021JOURNAL OPEN ACCESS FULL-TEXT HTMLMetallic Fe (hereinafter abbreviated as M.Fe) is suspended in steelmaking slags due to the stirring action during blowing and is mainly recovered via pulverization, classification, and magnetic separation. However, steelmaking slags are hard, and it is difficult to transform irregular-shaped and fine M.Fe in slags into free particles through the conventional pulverization method, which requires a large energy consumption. In this study, pulverization and separation experiments of steelmaking slags were performed using electrical pulse disintegration, which is completely different from the conventional pulverization method and capable of causing preferential fracture at the heterophase interface. As a result, several free particles of M.Fe with almost no slag attached were obtained from the coarse and fine pulverized particles. In addition, the electric field analysis results of a system where spherical M.Fe exists in a slag show that electric field concentration occurs in the front and back directions of the external magnetic field. The findings also show that a fracture can occur at the interface between the M.Fe and slag due to the combination of increased discharge probability, concentration of thermal energy, and generation of the Maxwell stress. Furthermore, the larger the pulverized mass, the higher the pulverization efficiency. In sum, electrical pulse disintegration may be advantageous for actual operations, where large quantities of oxides employed in the steel industry, such as steelmaking slag, spent refractories, and raw materials, should be treated in a short time with low energy consumption.
 View full abstractDownload PDF (3525K) Full view HTML
View full abstractDownload PDF (3525K) Full view HTML
- |<
- <
- 1
- >
- >|