
- |<
- <
- 1
- >
- >|
-
Kazuma Higashisaka, Kazuya Nagano, Yasuo Yoshioka, Yasuo Tsutsumi2017 年 40 巻 3 号 p. 243-248
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTMLIn the past decade, nanotechnology has advanced rapidly, and many products containing nanoparticles are now an important part of our daily lives. Despite our increasing exposure to nanoparticles, however, information regarding the absorption, distribution, metabolism, excretion, and toxicity of nanoparticles remains limited. In this review, we introduce our group’s ongoing research into the biological effects and toxicities of nanoparticles, which we broadly refer to as “nano-safety research.” In addition to determining the biological effects of nanoparticles and elucidating the underlying mechanisms of those effects, we are also exploring the associations among the physicochemical properties and kinetics of nanoparticles. Furthermore, we are currently developing a battery of biomarkers that we hope will be used to predict the biological effects of nanoparticles during the early stages of development. Our research provides valuable basic information on the safety of nanoparticles. We hope that this information will be used for the development of better assessments of nanoparticles safety and for the creation of more appropriate regulations to ensure not only the safety but also the sustainability of nanotechnology.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (602K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (602K) HTML形式で全画面表示
-
Yumei Li, Ting Zhang, Xiaojie Zhang, Wenying Zou, Xian Gong, Jianliang ...2017 年 40 巻 3 号 p. 249-255
発行日: 2017/03/01
公開日: 2017/03/01
[早期公開] 公開日: 2016/12/17ジャーナル フリー HTMLTo determine the combined effect of type 2 diabetes (T2D) and chronic cerebral hypoperfusion (CCH) on learning and spatial memory, we developed a rat model of CCH by permanent occlusion of bilateral common carotid arteries (2-vessel occlusion (2VO)) in high-fat diet (HFD)-fed rats injected with low-dose streptozotocin (STZ). Furthermore, we examined the effect of cinepazide maleate (CM) on cognitive deficits and brain damage in this rat model. Rats were maintained on HFD for 6 weeks and then injected with 35 mg/kg STZ to induce T2D. Sham or 2VO surgery was performed in non-diabetic or diabetic (DM) rats to obtain four groups: blank, DM, CCH, and DM-CCH groups. Cognitive function was tested by the Morris water maze (MWM) test. To determine the effects of the vasodilator cinepazide maleate (CM) on cognitive deficits and brain damage, DM-CCH rats were administered with 10 mg/kg CM or saline daily for 14 d. Neuronal damage in DM-CCH rats was associated with increased expression of glial fibrillary acidic protein (GFAP) and β-secretase 1 (BACE1), but decreased expression of choline acetyltransferase (ChAT). Moreover, the levels of all these proteins were significantly alleviated by CM treatment. These results suggest that T2D exacerbated CCH-induced brain damage and cognitive impairment, and CM ameliorated these effects.
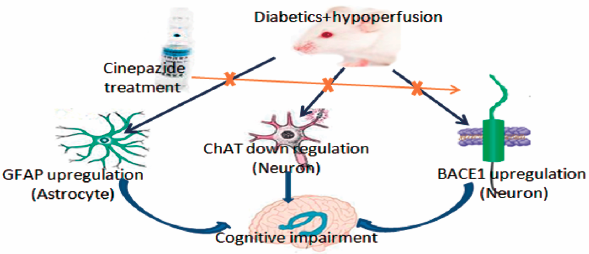 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (2644K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (2644K) HTML形式で全画面表示 -
Sunyoung Kim, Young-Won Chin, Jungsook Cho2017 年 40 巻 3 号 p. 256-265
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTMLLuteolin, one of the most common flavonoids present in many types of natural products, possesses diverse biological properties including anti-oxidant activity. In this study, we investigated neuroprotective effect of luteolin and its underlying signaling pathways using primary cultured rat cortical cells. Luteolin was demonstrated to attenuate H2O2- or xanthine/xanthine oxidase-induced oxidative damage and generation of intracellular reactive oxygen species (ROS). It enhanced the phosphorylation of Bad at Ser112 and attenuated H2O2-induced activation of caspase 3, indicating anti-apoptotic action. Terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling (TUNEL) assay confirmed this finding, showing inhibition of H2O2-induced DNA fragmentation. We also found that luteolin significantly up-regulated the expression of anti-oxidant enzyme heme oxygenase (HO)-1. Treatment with tin protoporphyrin IX, a selective HO-1 inhibitor, abolished neuroprotective and anti-apoptotic effects of luteolin, suggesting a critical role of HO-1 up-regulation. It was also shown to increase the phosphorylation of mitogen-activated protein kinase (MAPKs) such as extracellular signal-regulated kinase (ERK), p38 MAPK and c-Jun N-terminal kinases (JNK) and Akt. Treatment of the cells with specific inhibitors including SB203580, SP600125, and LY294002 suppressed the luteolin-induced HO-1 expression, suggesting the involvement of p38 MAPK, JNK, and Akt in HO-1 induction. In contrast, HO-1 expression was not reduced by U0126, implying that ERK may not be directly involved in HO-1 induction. These results indicate that luteolin exhibits neuroprotective effect through the inhibition of ROS and apoptotic cell death. Furthermore, up-regulation of HO-1 expression via p38 MAPK, JNK and Akt may contribute, at least in part, to luteolin-mediated neuroprotection. Based on these findings, luteolin may serve as a potential intervention for neurodegenerative diseases associated with oxidative stress.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (2229K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (2229K) HTML形式で全画面表示 -
Hiroko Awa, Akihiko Futamura, Takashi Higashiguchi, Akihiro Ito, Naoha ...2017 年 40 巻 3 号 p. 266-271
発行日: 2017/03/01
公開日: 2017/03/01
[早期公開] 公開日: 2017/01/26ジャーナル フリー HTMLA functional dietary supplement (FDS) containing Coenzyme Q10, branched-chain amino acids and L-carnitine was administered to tumor-bearing mice, investigating its effects on tumor and muscle tissues. Experiment (A): B16 melanoma cells were implanted subcutaneously into the right side of the abdomen of 8- to 9-week-old C57BL/6J mice. The mice were divided into two groups: a FDS group that received oral administration of FDS (n=10), and a control group that received oral administration of glucose (n=10). The moribund condition was used as the endpoint, and median survival time was determined. Experiment (B): On day 21 after tumor implantation, tumors, soleus muscle, gastrocnemius muscle, and suprahyoid muscles were collected. Tumor and muscle weight and other aspects were evaluated in each group: FDS group (n=15) and control group (n=15). The median survival time was comparable (21 d in the FDS group vs. 18 d in the control group, p=0.30). However, cumulative food intake was significantly higher in the FDS group than the control group (p=0.011). Metastasis of melanoma to the lung was observed in the control group but not in the FDS group (p=0.043). The weight of the suprahyoid muscles was significantly higher in the FDS group than in the control group (p=0.0045). The weight of the tumor was significantly lower in the FDS group than in the control group (p=0.013). The results possibly suggest oral administration of FDS in tumor-bearing mice enhances the maintenance of suprahyoid muscles, resulting in an extended feeding period and suppression of tumor growth and metastasis.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (663K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (663K) HTML形式で全画面表示 -
Mitsuhiro Machitani, Fuminori Sakurai, Keisaku Wakabayashi, Kosuke Nak ...2017 年 40 巻 3 号 p. 272-277
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTML
電子付録Clustered regularly interspaced short palindromic repeat (CRISPR)/Cas9-mediated genome engineering technology is a powerful tool for generation of cells and animals with engineered mutations in their genomes. In order to introduce the CRISPR/Cas9 system into target cells, nonviral and viral vectors are often used; however, such vectors trigger innate immune responses associated with production of type I interferons (IFNs). We have recently demonstrated that type I IFNs inhibit short-hairpin RNA-mediated gene silencing, which led us to hypothesize that type I IFNs may also inhibit CRISPR/Cas9-mediated genome mutagenesis. Here we investigated this hypothesis. A single-strand annealing assay using a reporter plasmid demonstrated that CRISPR/Cas9-mediated cleavage efficiencies of the target double-stranded DNA were significantly reduced by IFNα. A mismatch recognition nuclease-dependent genotyping assay also demonstrated that IFNα reduced insertion or deletion (indel) mutation levels by approximately half. Treatment with IFNα did not alter Cas9 protein expression levels, whereas the copy numbers of guide RNA (gRNA) were significantly reduced by IFNα stimulation. These results indicate that type I IFNs significantly reduce gRNA expression levels following introduction of the CRISPR/Cas9 system in the cells, leading to a reduction in the efficiencies of CRISPR/Cas9-mediated genome mutagenesis. Our findings provide important clues for the achievement of efficient genome engineering using the CRISPR/Cas9 system.
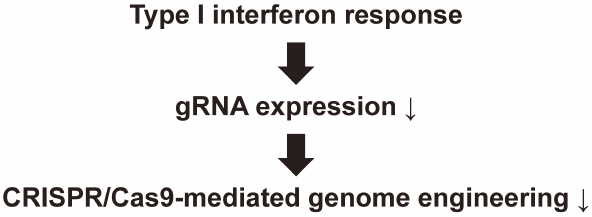 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (2491K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (2491K) HTML形式で全画面表示 -
Yoshihiro Uekuzu, Takashi Higashiguchi, Akihiko Futamura, Akihiro Ito, ...2017 年 40 巻 3 号 p. 278-283
発行日: 2017/03/01
公開日: 2017/03/01
[早期公開] 公開日: 2016/12/16ジャーナル フリー HTMLThere have been few detailed reports on respiratory depression due to overdoses of opioids in terminal cancer patients. We investigated the situation of treatment with opioid antagonists for respiratory depression that occurred after administration of opioid at optimal doses in terminal cancer patients, to clarify pathological changes as well as causative factors. In 2443 terminal cancer patients receiving opioids, 7 patients (0.3%) received opioid antagonists: 6, morphine (hydrochloride, 5; sulfate, 1); 1, oxycodone. The median dosage of opioids was 13.3 mg/d, as converted to morphine injection. Respiratory depression occurred on this daily dose in 4 patients and after changed dose and route in 3 patients. Opioids were given through the vein in 6 patients and by the enteral route in 1 patient. Concomitant drugs included nonsteroidal anti-inflammatory drugs in 3 patients and zoledronic acid in 2 patients. In morphine-receiving patients, renal functions were significantly worsened at the time of administration of an opioid antagonist than the day before the start of opioid administration. These findings indicate that the proper use of opioids was safe and acceptable in almost all terminal cancer patients. In rare cases, however, a risk toward respiratory depression onset is indicated because morphine and morphine-6-glucuronide become relatively excessive owing to systemic debility due to disease progression, especially respiratory and renal dysfunctions. At the onset of respiratory depression, appropriate administration of an opioid antagonist mitigated the symptoms. Thereafter, opioid switching or continuous administration at reduced dosages of the same opioids prevented the occurrence of serious adverse events.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (377K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (377K) HTML形式で全画面表示 -
Yukino Nagai, Satoko Kawano, Kenichiro Motoda, Masaaki Tomida, Chiye T ...2017 年 40 巻 3 号 p. 284-289
発行日: 2017/03/01
公開日: 2017/03/01
[早期公開] 公開日: 2016/12/20ジャーナル フリー HTMLWe investigated the solubility of 10 samples of sucrose esters of fatty acids (SEFA) products that are commercially available worldwide as food additives (emulsifiers). Although one sample dissolved transparently in both water and ethanol, other samples produced white turbidity and/or precipitates and did not meet the solubility criterion established by the Joint Food and Agriculture Organization of the United Nations (FAO)/WHO Expert Committee on Food Additives (JECFA). When the sample solutions were heated, the solubility in both water and ethanol increased. All of the samples dissolved transparently in ethanol, and dispersed and became white without producing precipitates in water. The present study suggests that the current solubility criterion of the JECFA SEFA specifications needs to be revised.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1285K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1285K) HTML形式で全画面表示 -
Ryuji Kato, Takaji Sato, Michiko Kanamori, Mizuho Miyake, Ayumi Fujimo ...2017 年 40 巻 3 号 p. 290-296
発行日: 2017/03/01
公開日: 2017/03/01
[早期公開] 公開日: 2016/12/14ジャーナル フリー HTMLCisplatin is the most widely used anticancer drug in the world. Mono-chloro and none-chloro complexes of cisplatin may be believed to be the activated compounds. The separation of these compounds using octa decyl silyl column or aminopropylsilyl silica gel column is difficult because of high-reactivity and structural similarity. In this study, cisplatin, hydroxo complexes, and OH-dimer were determined by HPLC using a naphthylethyl group bonded with silica gel (πNAP) column. The analytical conditions of HPLC were as follows: analytical column, πNAP column; wave length, 225 nm; column temperature, 40°C; mobile phase, 0.1 M sodium perchlorate, acetonitrile, and perchloric acid (290 : 10 : 3), flow rate, 1.0 mL/min. Sample (20 µL) was injected onto the HPLC system. Retention time of cisplatin, mono-chloride, OH-dimer, and none-chloride was 3.2, 3.4, 3.6, and, 4.3–6.6 min, respectively. Measurable ranges with this method were 1×10−5 to 4×10−3 M for cisplatin. Correlation coefficient of the calibration curves of cisplatin was 0.999 (p<0.01). The within- and between-day variations of coefficient of variation (CV) were 5% or lower. In this study, injectable formulations in physiological saline solution, water for injection, 5% glucose solution, and 7% sodium bicarbonate precisely were measured the stability and compositional changes upon mixing by πNAP column rather than C18 column. We successfully determined cisplatin, hydroxo complexes, and OH-dimer by HPLC using a πNAP column. Thus the measurement of cisplatin (cis-diamminedichloro-platinum(II), cis-[PtCl2(NH3)2]) (CDDP) should be done using a πNAP column rather than a C18 column or aminopropylsilyl silica gel column.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (772K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (772K) HTML形式で全画面表示 -
 Masayori Hagimori, Eri Hatabe, Kohei Sano, Hirotaka Miyazaki, Hitoshi ...2017 年 40 巻 3 号 p. 297-302
Masayori Hagimori, Eri Hatabe, Kohei Sano, Hirotaka Miyazaki, Hitoshi ...2017 年 40 巻 3 号 p. 297-302
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTMLSentinel lymph nodes (SLN) are the first lymph nodes (LN) where cancer cells metastasize from the primary tumor. We designed fluorophore-quencher-based activatable nanoparticles for SLN imaging. We selected TAMRA as a fluorophore and BHQ2 or QSY7 as a quencher. Ternary anionic complexes were constructed with generation 4th polyamidoamine dendrimer (G4) modified with TAMRA and p-SCN-Bn-DTPA (DTPA), polyethyleneimine (PEI) modified with BHQ2 or QSY7, and γ-polyglutamic acid (γ-PGA) by the electrostatic self-assembly system. TAMRA-G4-DTPA/PEI-BHQ2/γ-PGA and TAMRA-G4-DTPA/PEI-QSY7/γ-PGA complexes had a particle size of about 40 nm and a ζ-potential of −50 mV, and showed fluorescence resonance energy transfer (FRET) quenching. Fluorescence microscopy studies demonstrated that TAMRA-G4-DTPA/PEI-QSY7/γ-PGA complex produced intracellular fluorescent signals in the lysosome. During in vivo fluorescent imaging, TAMRA-G4-DTPA/PEI-QSY7/γ-PGA complex enabled the detection of mouse popliteal LN. The fluorophore-quencher conjugated γ-PGA complex based on FRET quenching would be useful for fluorescence-based optical imaging of SLN.
 Graphical Abstract Fullsize Image抄録全体を表示Editor's pick
Graphical Abstract Fullsize Image抄録全体を表示Editor's pickSentinel lymph nodes (SLN) are the first LN where cancer cells metastasize from the primary tumor. As an activatable fluorescence probe to detect the SLNs, Hagimori et al. developed ternary anionic nanoparticles constructed with fluorophore (TAMRA)-labeled polyamidoamine dendrimer conjugated with diethylenetriaminepentaacetic acid (TAMRA-G4-DTPA), quencher-labeled polyethyleneimine (PEI-QSY7 or PEI-BHQ2), and g-polyglutamic acid, namely TAMRA-G4-DTPA/PEI-QSY7/g-PGA and TAMRA-G4-DTPA/PEI-BHQ2/g-PGA by the electrostatic self-assembly system. The fluorescence of these complexes was quenched by a strong stacking interaction of TAMRA and quenchers, but was dequenched by dissociation of complexes when taken up by inflammatory cells (high populations in LN). They performed fluorescence imaging at 24 h after intradermal injection of TAMRA-G4-DTPA/PEI-QSY7/g-PGA into mouse footpads. Then, TAMRA fluorescence signal was clearly visualized in popliteal lymph node with high contrast.
PDF形式でダウンロード (4245K) HTML形式で全画面表示 -
Keishi Ishida, Yaichiro Kotake, Seigo Sanoh, Shigeru Ohta2017 年 40 巻 3 号 p. 303-309
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTMLLead is a persistent environmental pollutant and exposure to high environmental levels causes various deleterious toxicities, especially to the central nervous system (CNS). The α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor that is devoid of the glutamate receptor 2 (GluR2) subunit is Ca2+-permeable, which increases the neuronal vulnerability to excitotoxicity. We have previously reported that long-term exposure of rat cortical neurons to lead acetate induces decrease of GluR2 expression. However, it is not clarified whether lead-induced GluR2 decrease is involved in neurotoxicity. Therefore, we investigated the contribution of GluR2 non-containing AMPA receptor to lead-induced neurotoxic events. Although the expression of four AMPA receptor subunits (GluR1, GluR2, GluR3, and GluR4) was decreased by lead exposure, the decrease in GluR2 expression was remarkable among four subunits. Lead-induced neuronal cell death was rescued by three glutamate receptor antagonists, 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX, a non-selective AMPA receptor blocker), MK-801 (N-methyl-D-aspartate (NMDA) receptor blocker), and 1-naphthyl acetyl spermine (NAS, a specific Ca2+-permeable AMPA receptor blocker). Lead exposure activated extracellular signal-regulated protein kinase (ERK) 1/2, which was significantly ameliorated by CNQX. In addition, lead exposure activated p38 mitogen-activated protein kinase (MAPK p38), and protein kinase C (PKC), which was partially ameliorated by CNQX. Our findings indicate that Ca2+-permeable AMPA receptors resulting from GluR2 decrease may be involved in lead-induced neurotoxicity.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (8831K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (8831K) HTML形式で全画面表示 -
Keishi Yamasaki, Hiroki Sato, Saori Minagoshi, Karin Kyubun, Makoto An ...2017 年 40 巻 3 号 p. 310-317
発行日: 2017/03/01
公開日: 2017/03/01
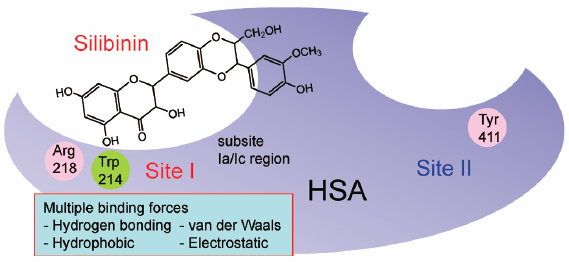
ジャーナル フリー HTMLSilibinin is the main constituent of silymarin, an extract from the seeds of milk thistle (Silybum marianum). Because silibinin has many pharmacological activities, extending its clinical use in the treatment of a wider variety of diseases would be desirable. In this study, we report on the binding of silibinin to plasma proteins, an issue that has not previously been extensively studied. The findings indicated that silibinin mainly binds to human serum albumin (HSA). Mutual displacement experiments using ligands that primarily bind to sites I and II clearly revealed that silibinin binds tightly and selectively to site I (subsites Ia and/or Ic) of HSA, which is located in subdomain IIA. Thermodynamic analyses suggested that hydrogen bonding and van der Waals interactions are major contributors to silibinin–HSA interactions. Furthermore, the binding of silibinin to HSA was found to be decreased with increasing ionic strength and detergent concentration of the media, suggesting that electrostatic and hydrophobic interactions are involved in the binding. Trp214 and Arg218 were identified as being involved in the binding of silibinin to site I, based on binding experiments using chemically modified- and mutant-HSAs. In conclusion, the available evidence indicates that silibinin binds to the region close to Trp214 and Arg218 in site I of HSA with assistance by multiple forces and can displace site I drugs (e.g., warfarin or iodipamide), but not site II drugs (e.g., ibuprofen).
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (667K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (667K) HTML形式で全画面表示 -
Yuki Naito, Yutaka Yoshikawa, Michiko Shintani, Shingo Kamoshida, Naem ...2017 年 40 巻 3 号 p. 318-326
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTML
電子付録Zinc (Zn) is a trace element with anti-diabetes mellitus (anti-DM) effects. Zn complexes exhibit stronger insulin-like activity than Zn ions. Bis(hinokitiolato)zinc complex ([Zn(hkt)2]) was recently reported to be a potent anti-DM candidate. We examined the effects of [Zn(hkt)2] on insulin resistance and pancreatic islet cells through in vivo long-term ingestion studies. In an in vivo study, we performed 4-month long-term [Zn(hkt)2] administration experiments in KK-Ay mice as a type 2 DM animal model. Ingestion of [Zn(hkt)2] resulted in lower blood glucose levels compared with the non-treated KK-Ay mice (control group). Additionally, [Zn(hkt)2] treatment decreased plasma insulin concentration compared with that of the non-treated KK-Ay group. [Zn(hkt)2] treatment resulted in a significant suppression of islet cell enlargement and a significantly decreased number of insulin-positive cells compared with the non-treated KK-Ay control group. The [Zn(hkt)2] treatment group showed the increasing tendency in the amount of Zn levels in peripheral organs; liver, muscle, adipose, and pancreas, compared with the non-treated KK-Ay control group. However, the Zn level in the pancreas of the [Zn(hkt)2] treatment group did not show the significant increase compared with the non-treated KK-Ay control group. This accumulation of Zn in pancreas suggested that [Zn(hkt)2] mainly effects on the peripheral tissue, and [Zn(hkt)2] has the less effect on the pancreas directly. Thus, we concluded that [Zn(hkt)2] exerted the main effect on peripheral organs by ameliorating insulin resistance.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (3527K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (3527K) HTML形式で全画面表示 -
Takashi Kawakubo, Ryotaro Mori, Keiro Shirotani, Nobuhisa Iwata, Masas ...2017 年 40 巻 3 号 p. 327-333
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTMLAmyloid-β peptide (Aβ) accumulation is a triggering event leading to the Alzheimer’s disease (AD) pathological cascade. Almost all familial AD-linked gene mutations increase Aβ production and accelerate the onset of AD. The Swedish mutation of amyloid precursor protein (APP) affects β-secretase activity and increases Aβ production up to ca. 6-fold in cultured cells; the onset age is around 50. Down syndrome (DS) patients with chromosome 21 trisomy present AD-like pathologies at earlier ages (40s) compared with sporadic AD patients, because APP gene expression is 1.5-fold higher than that in healthy people, thus causing a 1.5-fold increase in Aβ production. However, when comparing the causal relationship of Aβ accumulation with the onset age between the above two populations, early DS pathogenesis does not appear to be accounted for by the increased Aβ production alone. In this study, we found that neprilysin, a major Aβ-degrading enzyme, was downregulated in DS patient-derived fibroblasts, compared with healthy people-derived fibroblasts. Treatment with harmine, an inhibitor of dual-specificity tyrosine phosphorylation-regulated kinase 1A (DYRK1A), which is located in the DS critical region of chromosome 21, and gene knockdown of DYRK1A, upregulated neprilysin in fibroblasts. These results suggest that a decrease in the Aβ catabolic rate may be, at least in part, one of the causes for accelerated AD-like pathogenesis in DS patients if a similar event occurs in the brains, and that neprilysin activity may be regulated directly or indirectly by DYRK1A-mediated phosphorylation. DYRK1A inhibition may be a promising disease-modifying therapy for AD via neprilysin upregulation.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (861K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (861K) HTML形式で全画面表示 -
Tomoko Nishikawa, Yutaro Tanaka, Makiya Nishikawa, Yuka Ogino, Kosuke ...2017 年 40 巻 3 号 p. 334-338
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTMLMulticellular spheroids are useful as three-dimensional cell culture systems and for cell-based therapies. Their successful application requires an understanding of the consequences of spheroid size for cellular functions. In the present study, we prepared multicellular spheroids of different sizes using the human hepatoblastoma HepG2 cells, as hepatocytes are frequently used for in vitro drug screening and cell-based therapy. Precise polydimethylsiloxane-based microwells with widths of 360, 450, 560, and 770 µm were fabricated using a micromolding technique. Incubation of HepG2 cells in cell culture plates containing the microwells resulted in the formation of HepG2 spheroids with average diameters of 195, 320, 493, and 548 µm. The cell number per spheroid positively correlated with its diameter, and the viability of HepG2 cells was 94% or above for all samples. The smallest HepG2 spheroids showed the highest albumin secretion. On the other hand, the metabolic activity of 7-ethoxyresorufin, a fluorometric substrate for CYP1A1, increased with increasing spheroid size. These results indicate that controlling spheroid size is important when preparing HepG2 spheroids and that the size of HepG2 spheroids greatly influences the cellular function of HepG2 cells in the spheroids.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1007K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1007K) HTML形式で全画面表示 -
Mi Young Song, Hyo Won Jung, Seok Yong Kang, Yong-Ki Park2017 年 40 巻 3 号 p. 339-344
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTMLTargeting energy expenditure provides a potential alternative strategy for achieving energy balance to combat obesity and the development of type 2 diabetes mellitus (T2DM). In the present study, we investigated whether atractylenolide III (AIII) regulates energy metabolism in skeletal muscle cells. Differentiated C2C12 myotubes were treated with AIII (10, 20, or 50 µM) or metformin (2.5 mM) for indicated times. The levels of glucose uptake, the expressions of key mitochondrial biogenesis-related factors and their target genes were measured in C2C12 myotubes. AIII significantly increased the glucose uptake levels, and significantly increased the expressions of peroxisome proliferator-activated receptor coactivator-1α (PGC1α) and mitochondrial biogenesis-related markers, such as, nuclear respiratory factor-1 (NRF-1), and mitochondrial transcription factor A (TFAM) and mitochondrial mass and total ATP contents. In addition, AIII significantly increased the phosphorylation of AMP-activated protein kinase (AMPK) and the expression of sirtuin1 (SIRT1). These results suggest that AIII may have beneficial effects on obesity and T2DM by improving energy metabolism in skeletal muscle.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (2627K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (2627K) HTML形式で全画面表示 -
Manal Alfuwaires, Abdallah Altaher, Mahmoud Kandeel2017 年 40 巻 3 号 p. 345-351
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTMLThe new emerging Middle East Respiratory Syndrome Coronavirus (MERS CoV) encodes several resistance proteins against the innate immune response of the host, including interferon (IFN) resistance. Monitoring of the status of such proteins will be important to track viral pathogenicity. In this study, molecular dynamics approaches were used to investigate MERS CoV Non-Structural Protein 3 (NSP3) specific proteins that resist host innate immunity. MERS CoV papain-like protease (Plpro) was more conformationally flexible than Severe Acute Respiratory Syndrome CoV (SARS) CoV Plpro. This flexibility was evident in either the free form or when bound with ubiquitin. There were marked changes in the root-mean-square deviation (RMSD) in the ubiquitin like domain (Ubl) and the fingers subdomain of the catalytic domain of Plpro. An interesting feature is the dynamic change in Ubl, which shows a rigid conformation in the free form of Plpro but is fully flexible upon the binding of ubiquitin. This increased flexibility could be important for the downstream effects of the interaction with other proteins and the inhibition of the innate immunity. Four major residues involved in deubiquitination, L106, P163, R168 and F265, were conserved in all MERS CoVs and differed from other Beta CoVs. These conserved CoV residues were associated with lower deubiquitinating activity and render MERS CoV Plpro with less potent deubiquitinating potential. The number of residues and total interactions with ubiquitin were lower for the MERS CoV Plpro than for the SARS CoV. These factors contribute to the lower deubiquitinating actions of MERS CoV NSP3 and its subsequently lower interaction with the host immune system.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1697K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1697K) HTML形式で全画面表示 -
Tomoharu Takeuchi, Aya Sugimoto, Nao Imazato, Mayumi Tamura, Sachie Na ...2017 年 40 巻 3 号 p. 352-356
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTMLOsteoclasts represent the only bone resorbing cells in an organism. In this study, we investigated the effect of glucosamine (GlcN), a nutrient used to prevent joint pain and bone loss, on the osteoclastogenesis of murine macrophage-like RAW264 cells. GlcN supplementation suppressed the upregulation of osteoclast-specific genes (tartrate-resistant acid phosphatase (TRAP), cathepsin K, matrix metallopeptidase 9, and nuclear factor of activated T cell c1 (NFATc1)), receptor activator of nuclear factor-κB ligand (RANKL)-dependent upregulation of TRAP enzyme activity, and the formation of TRAP-positive multinuclear cells more effectively than N-acetylglucosamine (GlcNAc), which we have previously shown to inhibit osteoclast differentiation. To clarify the mechanism by which GlcN suppresses osteoclastogenesis, we further investigated the effect of GlcN on O-GlcNAcylation by Western blotting and on other types of glycosylation by lectin blotting. We found that, upon addition of GlcN, the O-GlcNAcylation of cellular proteins was increased whereas α2,6-linked sialic acid modification was decreased. Therefore, these glycan modifications in cellular proteins may contribute to the suppression of osteoclastogenesis.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1175K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1175K) HTML形式で全画面表示 -
Tatsuaki Tagami, Kaori Fukushige, Emi Ogawa, Naomi Hayashi, Tetsuya Oz ...2017 年 40 巻 3 号 p. 357-364
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTMLThree-dimensional (3D) printers have been applied in many fields, including engineering and the medical sciences. In the pharmaceutical field, approval of the first 3D-printed tablet by the U.S. Food and Drug Administration in 2015 has attracted interest in the manufacture of tablets and drugs by 3D printing techniques as a means of delivering tailor-made drugs in the future. In current study, polyvinylalcohol (PVA)-based tablets were prepared using a fused-deposition-modeling-type 3D printer and the effect of 3D printing conditions on tablet production was investigated. Curcumin, a model drug/fluorescent marker, was loaded into PVA-filament. We found that several printing parameters, such as the rate of extruding PVA (flow rate), can affect the formability of the resulting PVA-tablets. The 3D-printing temperature is controlled by heating the print nozzle and was shown to affect the color of the tablets and their curcumin content. PVA-based infilled tablets with different densities were prepared by changing the fill density as a printing parameter. Tablets with lower fill density floated in an aqueous solution and their curcumin content tended to dissolve faster. These findings will be useful in developing drug-loaded PVA-based 3D objects and other polymer-based articles prepared using fused-deposition-modeling-type 3D printers.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (5808K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (5808K) HTML形式で全画面表示 -
Khandaker Ahtesham Ahmed, Tianli Zhang, Katsuhiko Ono, Hiroyasu Tsutsu ...2017 年 40 巻 3 号 p. 365-374
発行日: 2017/03/01
公開日: 2017/03/01
[早期公開] 公開日: 2016/12/16ジャーナル フリー HTML
電子付録Guanosine 3′,5′-cyclic monophosphate (cGMP)-dependent protein kinases (PKG) are kinases regulating diverse physiological functions including vascular smooth muscle relaxation, neuronal synaptic plasticity, and platelet activities. Certain PKG inhibitors, such as Rp-diastereomers of derivatives of guanosine 3′,5′-cyclic monophosphorothioate (Rp-cGMPS), have been designed and used to study PKG-regulated cell signaling. 8-Nitroguanosine 3′,5′-cyclic monophosphate (8-nitro-cGMP) is an endogenous cGMP derivative formed as a result of excess production of reactive oxygen species and nitric oxide. 8-Nitro-cGMP causes persistent activation of PKG1α through covalent attachment of cGMP moieties to cysteine residues of the enzyme (i.e., the process called protein S-guanylation). In this study, we synthesized a nitrated analogue of Rp-cGMPS, 8-nitroguanosine 3′,5′-cyclic monophosphorothioate Rp-isomer (Rp-8-nitro-cGMPS), and investigated its effects on PKG1α activity. We synthesized Rp-8-nitro-cGMPS by reacting Rp-8-bromoguanosine 3′,5′-cyclic monophosphorothioate (Rp-8-bromo-cGMPS) with sodium nitrite. Rp-8-Nitro-cGMPS reacted with the thiol compounds cysteine and glutathione to form Rp-8-thioalkoxy-cGMPS adducts to a similar extent as did 8-nitro-cGMP. As an important finding, a protein S-guanylation-like modification was clearly observed, by using Western blotting, in the reaction between recombinant PKG1α and Rp-8-nitro-cGMPS. Rp-8-Nitro-cGMPS inhibited PKG1α activity with an inhibitory constant of 22 µM in a competitive manner. An organ bath assay with mouse aorta demonstrated that Rp-8-nitro-cGMPS inhibited vascular relaxation induced by acetylcholine or 8-bromo-cGMP more than Rp-8-bromo-cGMPS did. These findings suggest that Rp-8-nitro-cGMPS inhibits PKG through induction of an S-guanylation-like modification by attaching the Rp-cGMPS moiety to the enzyme. Additional study is warranted to explore the potential application of Rp-8-nitro-cGMPS to biochemical and therapeutic research involving PKG1α activation.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1276K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1276K) HTML形式で全画面表示 -
Makiko Fujiwara, Kenshi Ohbori, Akihiro Ohishi, Kentaro Nishida, Yoshi ...2017 年 40 巻 3 号 p. 375-380
発行日: 2017/03/01
公開日: 2017/03/01
ジャーナル フリー HTMLP2X7 receptor (P2X7R), a purinergic receptor, is involved in pathophysiological events such as inflammation and cell death, and thus is an attractive target for therapeutic approaches. It is reported that divalent metal cations (DMCs) inhibit P2X7R activation and that there are species differences in their inhibitory effects. To extrapolate the findings in experimental animals to humans, these species differences have to be clarified, but species differences in the sensitivity of P2X7R to DMCs between man and mouse have not been demonstrated. Here we performed direct comparison of the inhibitory effects of DMCs on human and mouse P2X7R activation. Cell lines constitutively expressing human and mouse P2X7R were used, and their P2X7R activation was evaluated as means of YO-PRO-1 dye uptake. MgCl2, NiCl2, ZnCl2, CuCl2 and CaCl2 dose-dependently decreased agonist-induced YO-PRO-1 uptake via both human and mouse P2X7Rs. Apparent differences in the inhibitory profiles for NiCl2 and CaCl2 between them were found, and the IC50 values of DMCs were in the order of CaCl2>MgCl2>NiCl2≈ZnCl2>CuCl2 for both human and mouse P2X7Rs. In this study, we demonstrate that human P2X7R exhibits different sensitivity to nickel and calcium compared with the case of the mouse one, while there is no species difference in the sensitivity of their P2X7Rs to magnesium, zinc and copper, suggesting that the effects of magnesium, zinc and copper on P2X7R-associated pathophysiological events in humans might be predicted from those in mice.
 Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1514K) HTML形式で全画面表示
Graphical Abstract Fullsize Image抄録全体を表示PDF形式でダウンロード (1514K) HTML形式で全画面表示
- |<
- <
- 1
- >
- >|