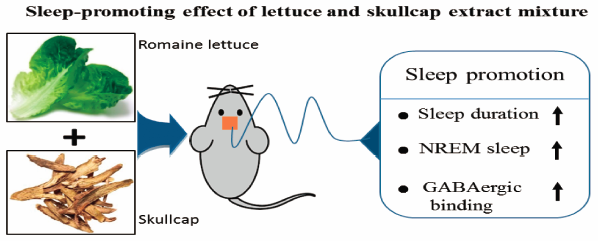
41 巻, 8 号
選択された号の論文の29件中1~29を表示しています
- |<
- <
- 1
- >
- >|
Review
-
2018 年 41 巻 8 号 p. 1119-1125
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (3355K) HTML形式で全画面表示
Current Topics - Ion Channels as Therapeutic Targets for the Immune, Inflammatory, and Metabolic Disorders
-
2018 年 41 巻 8 号 p. 1126
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (175K) HTML形式で全画面表示
Current Topics: Reviews
-
2018 年 41 巻 8 号 p. 1127-1134
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (2557K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1135-1144
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (596K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1145-1151
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (569K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1152-1157
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (836K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1158-1163
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (972K) HTML形式で全画面表示
Regular Articles
-
2018 年 41 巻 8 号 p. 1164-1169
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (3294K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1170-1177
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (833K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1178-1185
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (3484K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1186-1193
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (8574K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1194-1202
発行日: 2018/08/01
公開日: 2018/08/01
[早期公開] 公開日: 2018/05/29PDF形式でダウンロード (2857K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1203-1210
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (3772K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1211-1218
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (3608K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1219-1227
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (2348K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1228-1236
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (1563K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1237-1242
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (1240K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1243-1250
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (1536K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1251-1256
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (684K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1257-1268
発行日: 2018/08/01
公開日: 2018/08/01
[早期公開] 公開日: 2018/05/23PDF形式でダウンロード (11386K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1269-1276
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (598K) HTML形式で全画面表示
Notes
-
2018 年 41 巻 8 号 p. 1277-1281
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (530K) HTML形式で全画面表示 -
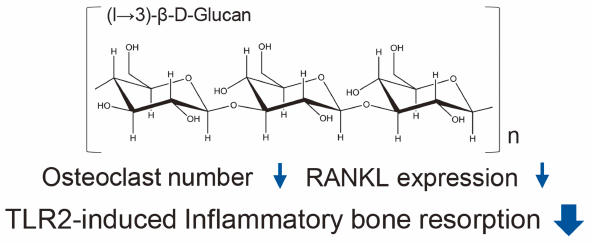
Low Molecular-Weight Curdlan, (1→3)-β-Glucan Suppresses TLR2-Induced RANKL-Dependent Bone Resorption2018 年 41 巻 8 号 p. 1282-1285
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (415K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1286-1290
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (508K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1291-1294
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (687K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1295-1298
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (780K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1299-1302
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (1888K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1303-1306
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (1803K) HTML形式で全画面表示 -
2018 年 41 巻 8 号 p. 1307-1310
発行日: 2018/08/01
公開日: 2018/08/01
PDF形式でダウンロード (663K) HTML形式で全画面表示
- |<
- <
- 1
- >
- >|