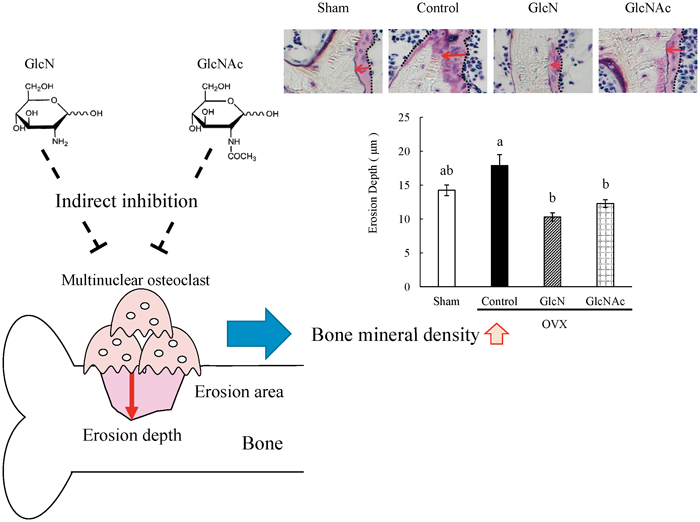
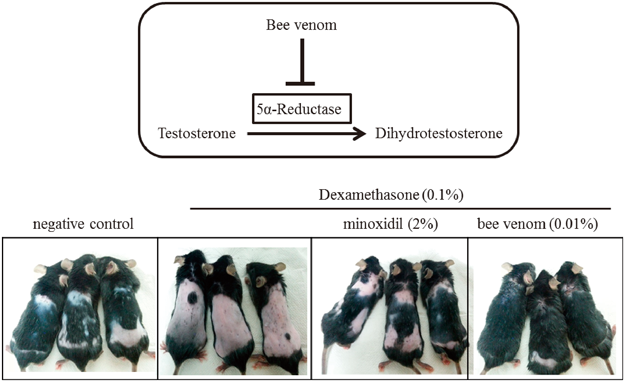
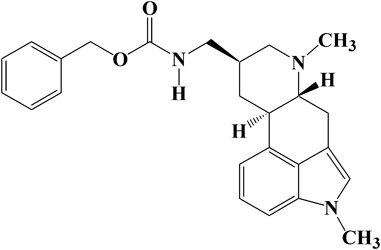
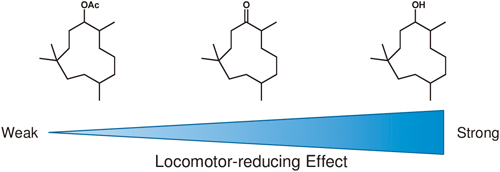
39 巻, 6 号
選択された号の論文の29件中1~29を表示しています
- |<
- <
- 1
- >
- >|
Review
-
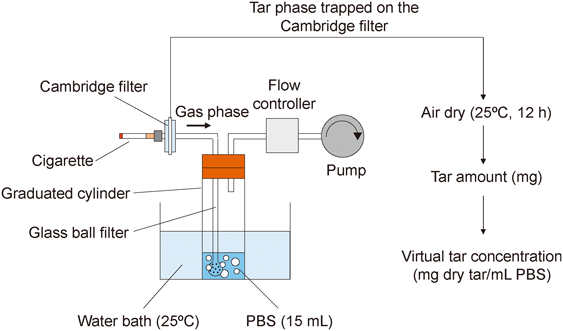
2016 年 39 巻 6 号 p. 891-896
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (892K) HTML形式で全画面表示
Current Topics: Recent Advances in Research on Bioactive Ingredients in Cigarette Smoke
-
2016 年 39 巻 6 号 p. 897
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (167K) HTML形式で全画面表示 -
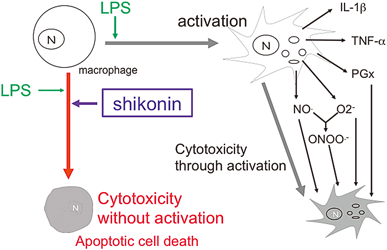
2016 年 39 巻 6 号 p. 898-902
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (523K) HTML形式で全画面表示 -
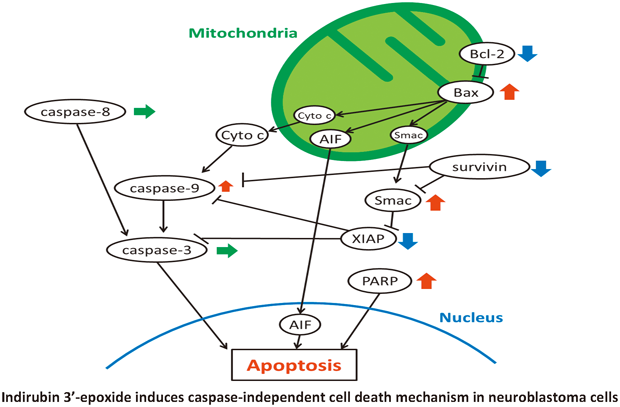
2016 年 39 巻 6 号 p. 903-908
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (642K) HTML形式で全画面表示 -
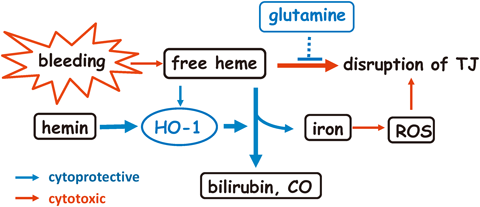
2016 年 39 巻 6 号 p. 909-914
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (428K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 915-919
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (823K) HTML形式で全画面表示
Regular Articles
-
2016 年 39 巻 6 号 p. 920-926
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (756K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 927-934
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (1445K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 935-945
発行日: 2016/06/01
公開日: 2016/06/01
[早期公開] 公開日: 2016/03/15PDF形式でダウンロード (7900K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 946-952
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (1472K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 953-958
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (1998K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 959-968
発行日: 2016/06/01
公開日: 2016/06/01
[早期公開] 公開日: 2016/03/31PDF形式でダウンロード (1092K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 969-976
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (1979K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 977-983
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (8022K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 984-992
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (1287K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 993-999
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (1859K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1000-1006
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (507K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1007-1012
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (808K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1013-1021
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (718K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1022-1028
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (4934K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1029-1034
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (12680K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1035-1041
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (4390K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1042-1046
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (3449K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1047-1054
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (913K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1055-1059
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (1248K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1060-1068
発行日: 2016/06/01
公開日: 2016/06/01
[早期公開] 公開日: 2016/04/02PDF形式でダウンロード (7408K) HTML形式で全画面表示
Notes
-
2016 年 39 巻 6 号 p. 1069-1072
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (438K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1073-1076
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (485K) HTML形式で全画面表示 -
2016 年 39 巻 6 号 p. 1077-1080
発行日: 2016/06/01
公開日: 2016/06/01
PDF形式でダウンロード (811K) HTML形式で全画面表示
- |<
- <
- 1
- >
- >|