- |<
- <
- 1
- >
- >|
-
2017Volume 40Issue 12 Pages 2015-2023
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (14112K) Full view HTML -
2017Volume 40Issue 12 Pages 2024-2037
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Advance online publication: October 07, 2017Download PDF (2885K) Full view HTML
-
2017Volume 40Issue 12 Pages 2038
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (153K) Full view HTML
-
2017Volume 40Issue 12 Pages 2039-2044
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (802K) Full view HTML -
2017Volume 40Issue 12 Pages 2045-2049
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (6320K) Full view HTML -
2017Volume 40Issue 12 Pages 2050-2060
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (891K) Full view HTML
-
2017Volume 40Issue 12 Pages 2061-2067
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (990K) Full view HTML
-
2017Volume 40Issue 12 Pages 2068-2074
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Advance online publication: September 22, 2017Download PDF (1999K) Full view HTML -
2017Volume 40Issue 12 Pages 2075-2080
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (1251K) Full view HTML -
2017Volume 40Issue 12 Pages 2081-2087
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Advance online publication: September 23, 2017Download PDF (1024K) Full view HTML -
2017Volume 40Issue 12 Pages 2088-2095
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (5446K) Full view HTML -
2017Volume 40Issue 12 Pages 2096-2104
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (1490K) Full view HTML -
2017Volume 40Issue 12 Pages 2105-2109
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (471K) Full view HTML -
2017Volume 40Issue 12 Pages 2110-2116
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (929K) Full view HTML -
2017Volume 40Issue 12 Pages 2117-2124
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Advance online publication: September 30, 2017Download PDF (2916K) Full view HTML -
2017Volume 40Issue 12 Pages 2125-2133
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Advance online publication: September 22, 2017Download PDF (2544K) Full view HTML -
2017Volume 40Issue 12 Pages 2134-2139
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (714K) Full view HTML -
2017Volume 40Issue 12 Pages 2140-2145
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (524K) Full view HTML -
2017Volume 40Issue 12 Pages 2146-2152
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Advance online publication: October 04, 2017Download PDF (3918K) Full view HTML -
2017Volume 40Issue 12 Pages 2153-2157
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Advance online publication: September 29, 2017Download PDF (423K) Full view HTML -
 2017Volume 40Issue 12 Pages 2158-2165
2017Volume 40Issue 12 Pages 2158-2165
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
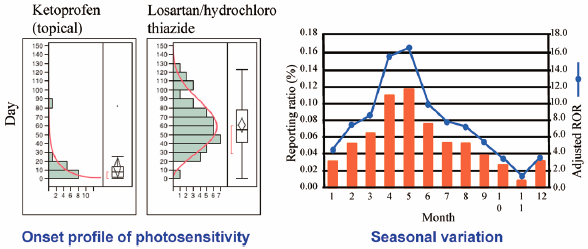
Editor's pickDrug-induced photosensitivity (DIP) is a cutaneous adverse event caused by the combined effects of a medication and exposure to light. The article by Nakao et al. evaluated the association between drugs and DIP by using the reporting odds ratio and time-to-onset analysis data from the Japanese Adverse Drug Event Report (JADER) database. More than half of the reports of DIP onset following ketoprofen administration were recorded within 10 days of initiation of treatment. The seasonal variation of DIP was shown to follow an annual sinusoidal pattern with peaks observed in April and May. The results of this study suggest close monitoring of patients taking suspected drugs, especially during the peak season of photosensitivity reactions.
Download PDF (738K) Full view HTML -
2017Volume 40Issue 12 Pages 2166-2174
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Advance online publication: September 30, 2017Download PDF (1082K) Full view HTML -
2017Volume 40Issue 12 Pages 2175-2182
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (661K) Full view HTML -
2017Volume 40Issue 12 Pages 2183-2190
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
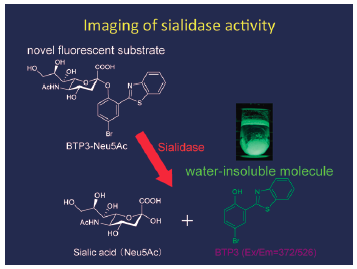
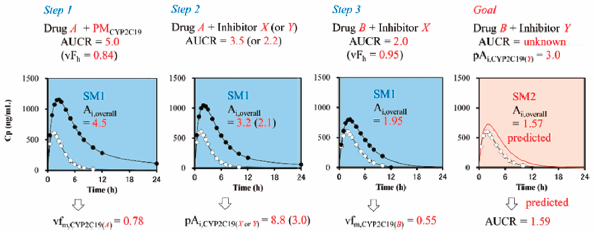
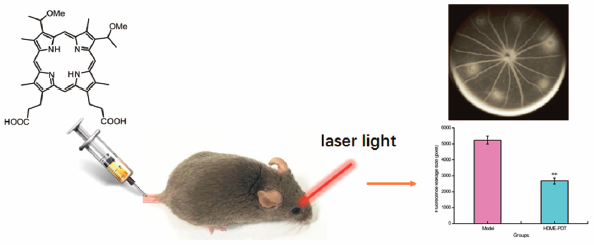
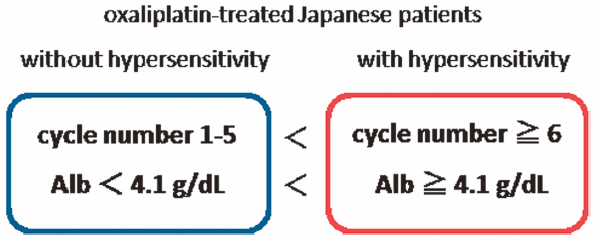
Download PDF (15544K) Full view HTML -
2017Volume 40Issue 12 Pages 2191-2198
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (1940K) Full view HTML
-
2017Volume 40Issue 12 Pages 2199-2204
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Advance online publication: September 28, 2017Download PDF (282K) Full view HTML -
2017Volume 40Issue 12 Pages 2205-2211
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (921K) Full view HTML
-
2017Volume 40Issue 12 Pages 2212
Published: December 01, 2017
Released on J-STAGE: December 01, 2017
Download PDF (147K) Full view HTML
- |<
- <
- 1
- >
- >|