- |<
- <
- 1
- >
- >|
-
 2020Volume 43Issue 3 Pages 361
2020Volume 43Issue 3 Pages 361
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Editor's pickCutting-edge contributions from invited poster presentations providing significant research works in the fifth International Symposium for Medicinal Sciences (ISMS) in the 139th Chiba annual meeting in 2019 are assembled for the Current Topics section in this issue of the Biological and Pharmaceutical Bulletin.
Download PDF (150K) Full view HTML
-
2020Volume 43Issue 3 Pages 362-365
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (576K) Full view HTML -
2020Volume 43Issue 3 Pages 366-374
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (831K) Full view HTML -
2020Volume 43Issue 3 Pages 375-383
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (1050K) Full view HTML
-
2020Volume 43Issue 3 Pages 384-392
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Advance online publication: November 02, 2019Download PDF (690K) Full view HTML -
2020Volume 43Issue 3 Pages 393-398
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Advance online publication: December 05, 2019Download PDF (790K) Full view HTML -
2020Volume 43Issue 3 Pages 399-403
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (674K) Full view HTML
-
2020Volume 43Issue 3 Pages 404-408
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (492K) Full view HTML
-
2020Volume 43Issue 3 Pages 409-417
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Advance online publication: December 24, 2019Download PDF (2994K) Full view HTML -
2020Volume 43Issue 3 Pages 418-423
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Advance online publication: December 20, 2019Download PDF (563K) Full view HTML -
2020Volume 43Issue 3 Pages 424-431
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Advance online publication: December 14, 2019Download PDF (2062K) Full view HTML -
2020Volume 43Issue 3 Pages 432-439
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Advance online publication: December 26, 2019Download PDF (1868K) Full view HTML -
2020Volume 43Issue 3 Pages 440-449
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (2536K) Full view HTML -
 2020Volume 43Issue 3 Pages 450-457
2020Volume 43Issue 3 Pages 450-457
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
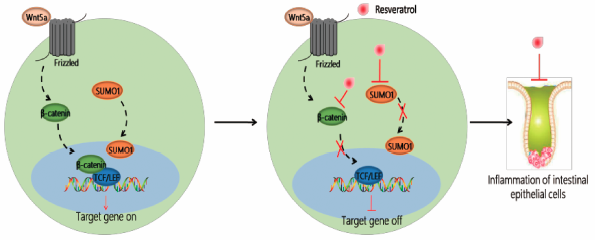
Editor's pickMao et al. found that resveratrol can significantly inhibit the expression of SUMO1. They demonstrate that resveratrol alleviates inflammatory bowel disease (IBD) in mice by inhibiting the expression of SUMO1 molecule, and by modulating the activation of wnt/β-catenin signaling pathway. Clinical analysis also proves that the expression of SUMO1 and β-catenin molecules increased with the worsening of the disease, which also provides a new method for clinical diagnosis and treatment of IBD.
Download PDF (11179K) Full view HTML -
 2020Volume 43Issue 3 Pages 458-462
2020Volume 43Issue 3 Pages 458-462
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Advance online publication: December 18, 2019Editor's pickEdoxaban is an oral anticoagulant used for preventing and treating stroke or systemic embolism. Bleeding is the most common complication associated with anticoagulants. In particular, severe bleeding is assumed to be related to mortality in patients treated with anticoagulation therapy. However, few studies have examined the risk factors for bleeding in Japanese patients receiving edoxaban. The article by Takase et al. revealed that a low baseline hemoglobin level was a significant risk factor for major and clinically relevant bleeding in Japanese patients receiving edoxaban.
Download PDF (315K) Full view HTML -
2020Volume 43Issue 3 Pages 463-473
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (2404K) Full view HTML -
2020Volume 43Issue 3 Pages 474-479
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (430K) Full view HTML -
2020Volume 43Issue 3 Pages 480-487
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (1963K) Full view HTML -
2020Volume 43Issue 3 Pages 488-492
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (324K) Full view HTML -
2020Volume 43Issue 3 Pages 493-502
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (1584K) Full view HTML -
2020Volume 43Issue 3 Pages 503-508
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (1768K) Full view HTML -
2020Volume 43Issue 3 Pages 509-515
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (2758K) Full view HTML -
 2020Volume 43Issue 3 Pages 516-525
2020Volume 43Issue 3 Pages 516-525
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Advance online publication: December 21, 2019Editor's pickThe article by Tanaka et al. proposed a novel mechanism of radioresistance and candidate for use as radiosensitizers in radiation therapy of melanoma. A2B receptor was involved in radioresistance via DNA damage response in B16 cells. A2B receptor antagonist enhances tumor growth-inhibitory effect by gamma-ray and shows radiosensitizing effect in vivo. These findings proposed that A2B receptor contributes to radioresistance, and could be a new target for the development of agents to increase the efficacy of radiotherapy.
Download PDF (1750K) Full view HTML -
2020Volume 43Issue 3 Pages 526-532
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (2468K) Full view HTML -
2020Volume 43Issue 3 Pages 533-539
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (4740K) Full view HTML -
 2020Volume 43Issue 3 Pages 540-545
2020Volume 43Issue 3 Pages 540-545
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Advance online publication: December 27, 2019Editor's pickCD81 is important for regulating biological processes such as B cell receptor signaling and B cell differentiation. However, little is known about degradation mechanism of CD81. Hosokawa et al. demonstrated that CD81 on the cell surface is degraded by lysosome via K63- and K29-linked poly-ubiquitination. The poly-ubiquitinated CD81 is translocated from the cell surface into endosomes and is degraded by lysosomes. This is the first report showing that the lysosomal degradation of CD81 requires poly-ubiquitination and clathrin-mediated endocytosis.
Download PDF (2973K) Full view HTML
-
2020Volume 43Issue 3 Pages 546-549
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (483K) Full view HTML -
2020Volume 43Issue 3 Pages 550-553
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (429K) Full view HTML -
2020Volume 43Issue 3 Pages 554-557
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Advance online publication: January 07, 2020Download PDF (430K) Full view HTML -
2020Volume 43Issue 3 Pages 558-564
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (2548K) Full view HTML -
2020Volume 43Issue 3 Pages 565-568
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Advance online publication: December 18, 2019Download PDF (481K) Full view HTML -
2020Volume 43Issue 3 Pages 569-573
Published: March 01, 2020
Released on J-STAGE: March 01, 2020
Download PDF (646K) Full view HTML
- |<
- <
- 1
- >
- >|