All issues

Volume 29, Issue 8
Displaying 1-16 of 16 articles from this issue
- |<
- <
- 1
- >
- >|
Rapid Communications
-
Toshio TAKAYANAGI, Masaki MORIMOTO, Tomoki YABUTANIArticle type: Rapid Communications
2013Volume 29Issue 8 Pages 769-771
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESSGraphene and chemically modified graphenes were resolved by micellar electrokinetic chromatography (MEKC) using sodium dodecylbenzenesulfonate (SDBS) as a micelle matrix. Graphene was successfully dispersed in a micellar solution, and a consecutive and broad signal was obtained by the MEKC with a 20 mmol dm−3 aqueous SDBS solution for Graphene Nanoplatelets. Chemically oxidized graphene was less retained to the micelle than the Graphene Nanoplatelets by the proposed MEKC, and they were resolved with each other. View full abstractDownload PDF (894K)
View full abstractDownload PDF (894K) -
Kazuaki OHARA, Masahide TOMINAGA, Isao AZUMAYA, Kentaro YAMAGUCHIArticle type: Rapid Communications
2013Volume 29Issue 8 Pages 773-776
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESS
Supplementary materialDiscrete and continuous coordination structures were obtained in single crystals of CoCl2 and an adamantane-based bidentate ligand bearing imidazolyl groups, depending on the methanol concentration in a methanol–chloroform mixture. Single-crystal X-ray structure analysis revealed that the metal centers exhibited a tetrahedral geometry in the discrete complex and an octahedral geometry in the continuous metal complex. Conventional analytical methods, including UV-vis and NMR spectroscopy, could not identify those two complexes in solution. In contrast, cold-spray ionization mass spectrometry could detect differences between the discrete complex and the continuous metal complex, and ion peaks due to continuous ligand adducts were found only in the spectrum of the continuous metal complex. View full abstractDownload PDF (948K)
View full abstractDownload PDF (948K)
Original Papers
-
Toshinori TANIUCHI, Yoshinori TAKANO, Kensei KOBAYASHIArticle type: Original Papers
2013Volume 29Issue 8 Pages 777-785
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESS
Supplementary materialThe organic haze of aerosols that shrouds the Saturnian moon Titan has previously been studied by both observations and laboratory simulation experiments. Here we report the abiotic formation of amino acid precursors in complex organic molecules during experimental simulation of the environment near Titan’s surface with proton irradiation. Pyrolysis of the organic molecules formed in the simulated Titan atmosphere by proton irradiation at 600°C yielded compounds that contained HCN and NH3 (m/z = 27 and 17). These experimental results are consistent with the molecular information obtained by pyrolysis gas chromatography/mass spectrometry (pyrolysis GC/MS) of samples collected by the Huygens probe to Titan. Scanning electron microscopy (SEM) and three-dimensional atomic force microscopy (AFM) images of the irradiation products reveal nanometer-scale filaments and globules in complex amorphous structures (approximately 1000 Da). Isotope probing experiments by matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF-MS) show that oxygen atoms were incorporated into the racemic amino acids by hydrolysis of 18O-labeled water. We suggest that the amino acid precursors possibly formed after water hydrolysis, as suggested in a previous observational study (C. A. Griffith, T. Owen, T. R. Geballe, J. Rayner, and P. Rannou, Science, 2003, 300, 628). We propose that cosmic rays are a significant and effective energy source for producing complex organics and amino acid precursors in Titan’s atmospheric haze. View full abstractDownload PDF (8260K)
View full abstractDownload PDF (8260K) -
Yasumi ANAN, Yoshiko HATAKEYAMA, Maki TOKUMOTO, Yasumitsu OGRAArticle type: Original Papers
2013Volume 29Issue 8 Pages 787-792
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESSTwo major selenoproteins are present in mammalian serum: extracellular glutathione peroxidase (eGPx) and selenoprotein P (Sel P). The chromatographic behaviors of the two serum selenoproteins were compared in four rodent species, and the selenoproteins in rat serum were identified by measuring enzyme activity and Western blotting. The selenoproteins in rat serum showed a specific chromatographic behavior. In particular, rat eGPx was eluted faster than eGPxs of the other rodent species, although the amino-acid sequences of the rodent species were identical. The elution profiles of Se in rat serum obtained by inductively coupled plasma tandem mass spectrometry (ICP-MS-MS) and ICP-MS were compared. The tandem quadrupoles and the O2 reaction/collision gas completely removed severe interferences with the Se speciation originating from the plasma source and the biological sample matrix. ICP-MS-MS under the O2 mass shift mode gave us more accurate abundance ratios of Se than ICP-MS. View full abstractDownload PDF (508K)
View full abstractDownload PDF (508K) -
Ying LIU, Susumu IMASHUKU, Jun KAWAIArticle type: Original Papers
2013Volume 29Issue 8 Pages 793-797
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESSMulti-element solutions containing the 11 elements S, K, Sc, V, Mn, Co, Cu, Ga, As, Br and Y were analyzed by a portable total reflection X-ray fluorescence (TXRF) spectrometer. The excitation parameters (glancing angle, operational voltage and current) and sample amount were optimized for the portable TXRF in order to realize the smallest possible detection limits for all elements. The excitation parameter dependencies of the fluorescence signal and background for the detected elements are explained in detail. Background contributed by the sample carrier is also discussed. Consequently, nine elements were detectable at sub-nanogram levels in a single measurement of 10 min under the optimal experimental conditions. The portable TXRF spectrometer was found to be suitable for simultaneous multi-element analysis with low detection limits. The features of high sensitivity, small sample amount required, and fast detection of a wide range of elements make the portable TXRF a valuable tool in various applications, such as field studies in environmental and geological investigations. View full abstractDownload PDF (707K)
View full abstractDownload PDF (707K) -
Guangyu SHEN, Hui XU, Anant S. GURUNG, Yunhui YANG, Guodong LIUArticle type: Original Papers
2013Volume 29Issue 8 Pages 799-804
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESSIn order to amplify the signal in a gold nanoparticle-based lateral flow immunoassay, a simple and sensitive method utilizing gold nanoparticle aggregates as a colored reagent formed with a polyamidoamine dendrimer was developed. The results were compared with that achieved by employing the individual nanoparticles used in the conventional lateral flow immunoassay. Under the optimized experimental conditions, a detection limit of 0.1 ng mL−1 for rabbit immunoglobulin G was achieved, which is almost 20-fold lower than that of the traditional method using individual gold nanoparticles. We believe that this simple, practical bioassay platform will be of interest for use in areas such as disease diagnostics, pathogen detection, and quality monitoring of food and water. View full abstractDownload PDF (832K)
View full abstractDownload PDF (832K) -
Suwen QI, Qiaoliang LI, Wei RAO, Xinyu LIU, Li YIN, Huisheng ZHANGArticle type: Original Papers
2013Volume 29Issue 8 Pages 805-810
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESSOur objective is to develop an assay based on magnetic particles (MPs) to determine the concentration of procalcitonin (PCT) using a chemiluminescence immunoassay (CLIA). Fluorescein isothiocyanate (FITC) and N-(aminobutyl)-N-(ethylisoluminol) (ABEI) were used to label two different anti-procalcitonin (PCT) monoclonal antibodies. The labeled antibodies, the PCT antigen, and the anti-FITC antibody-coated MPs formed a double-sandwiched immunocomplex. The measured relative light units (RLUs) of ABEI in the substrate solution were directly proportional to the amount of PCT present in the samples. The proposed method was linear to 600 ng/mL with a detection limit of 0.03 ng/mL. The coefficient of variation (CV) was <5% and <6% for the intra- and inter-assay precision, respectively. The average recoveries were between 95 and 107%. The linearity-dilution effect gave a linear correlation coefficient of 0.9912. This proposed assay provided an alternative method to quantitatively measure PCT in serum for the diagnosis of sepsis.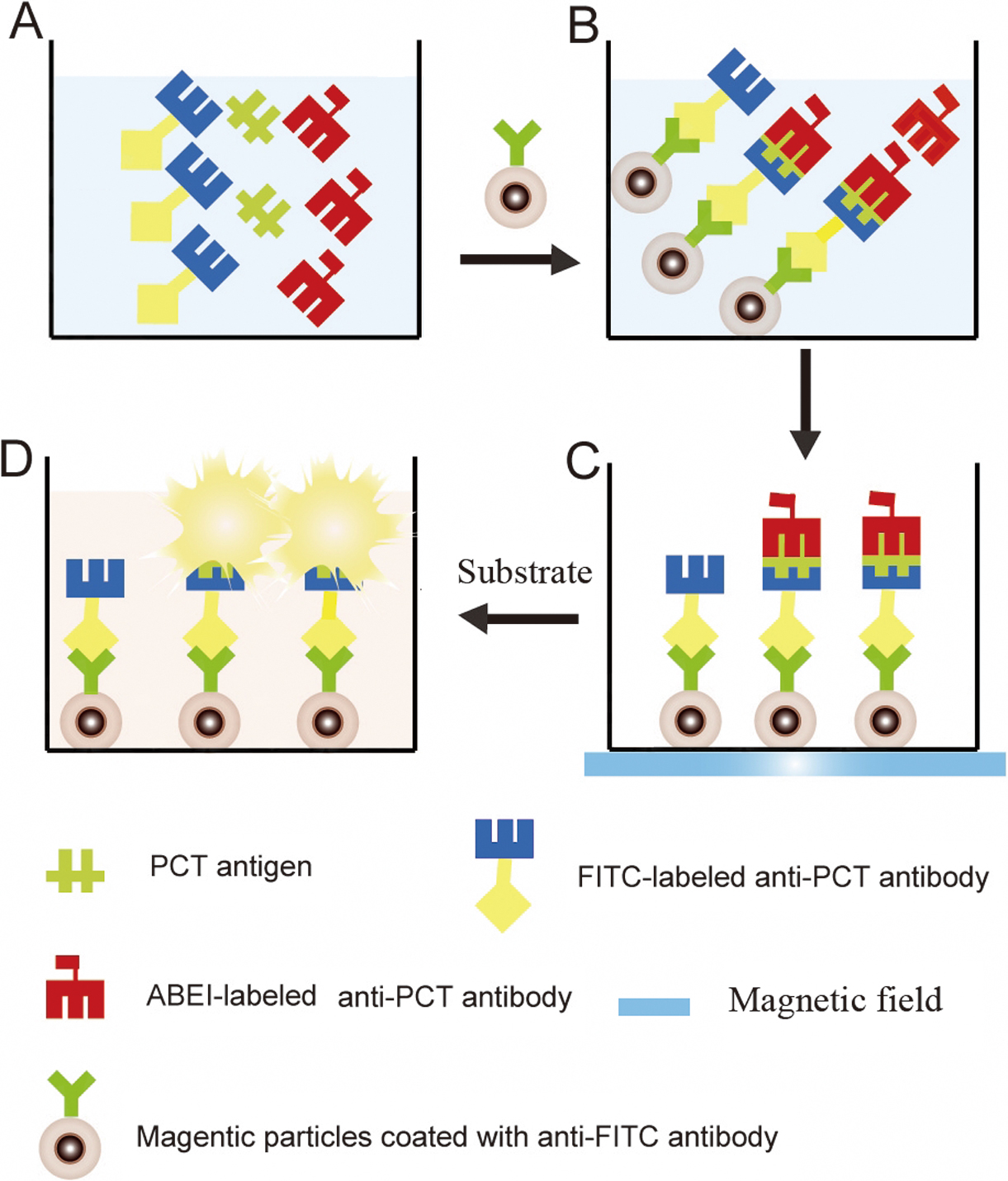 View full abstractDownload PDF (977K)
View full abstractDownload PDF (977K) -
Miho SUZUKI, Yoshihiro ISHIMARU, Ayumu SAITO, Koichi NISHIGAKIArticle type: Original Papers
2013Volume 29Issue 8 Pages 811-814
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESS
Supplementary materialWe have site-directedly linked a green fluorescent protein (GFP) variant and a β-cyclodextrin (β-CD) with a simple method to develop a basic complex for sophisticated supramolecules. We have confirmed β-CD grafting on GFP with several methods including matrix-assisted laser desorption/ionization linear time-of-flight mass spectrometry (MALDI-TOF MS) without protease digestion and characterized the complex as well. In consideration of the resulting properties, the product we plainly and efficiently obtained could have applications related to sensing devices and drug delivery systems. View full abstractDownload PDF (1682K)
View full abstractDownload PDF (1682K) -
Mohammad Javad CHAICHI, Tahereh KHAJVAND, Jalil MEHRZAD, Sakineh ASGHA ...Article type: Original Papers
2013Volume 29Issue 8 Pages 815-821
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESSA novel, simple, cheap, and high sensitivity batch chemiluminescent method for the determination of catecholamine drugs, epinephrine (E), dopamine (DA) and methyldopa (MD) at microgram levels in pharmaceutical formulations is described. The method is based on a chemiluminescence (CL) system arising from the reaction of bis(2,4,6-trichlorophenyl) oxalate (TCPO) with H2O2 in the presence of a novel fluorescer, furandicarboxylate, and is proposed as a new analytical method for the determination of catecholamines. The method is based on the inhibition of CL emission by DA and its enhancement by E and MD. Under optimal conditions, good linear ranges were obtained, 0.5 – 12.7, 0.06 – 1.83 and 0.069 – 3.52 μg/mL with detection limits of 0.30, 0.03 and 0.04 μg/mL (S/N = 3) for DA, E and MD, respectively. Moreover, a pooled-intermediate model was used to determine the kinetic parameters of CL with and without catecholamines and a possible CL mechanism was discussed.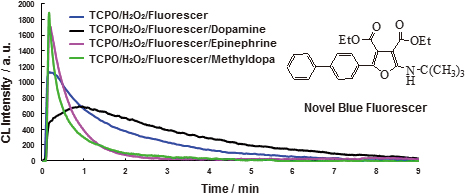 View full abstractDownload PDF (877K)
View full abstractDownload PDF (877K) -
Sarenqiqige, Masaki ASHITOMI, Kazuhisa YOSHIMURAArticle type: Original Papers
2013Volume 29Issue 8 Pages 823-829
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESS
Supplementary materialA cation-exchange column (TSK IC-Cation, 4.6 mm i.d., 10 mm) was used as a reaction/concentration/separation medium in an HPLC system. Cr(VI) and 1,5-diphenylcarbazide (DPC) were on-line mixed and reacted in a flow tube. When the reagents flowed into the column, both DPC and the complex were concentrated on the column, and the higher DPC concentration accelerated the remaining Cr(VI) to quickly complete the reaction with the DPC on the column. After the complexation and preconcentration, the complex and the extra DPC were eluted by a mixed solution containing lanthanum chloride and 1-propanol. The absorbance of the Cr(III)-DPCO complex at 540 nm was continuously monitored. In addition, the dissolved Cr(III) could be oxidized to Cr(VI) by 185-nm irradiation using a low pressure ultraviolet (UV) lamp for 8 min, and then analyzed by the present method as total chromium, thus the quantitative speciation analysis of Cr(III) and Cr(VI) was realized. In the range of 1 ng dm−3 – 100 μg dm−3 Cr(VI) concentration, the calibration curve was linear. The analytical time was 8 min for Cr(VI) and 24 min for total Cr. The detection limit (3σ) of the method was 0.6 ng dm−3 for Cr(VI) and 0.8 ng dm−3 for total chromium when using a 3.9-cm3 water sample. The present method could be successfully applied to the speciation analysis of dissolved chromium in natural water, and the leaching test of Cr(III) and Cr(VI) from stainless steel. View full abstractDownload PDF (1347K)
View full abstractDownload PDF (1347K) -
Sachio YAMAMOTO, Yumi NAKATANI, Shigeo SUZUKIArticle type: Original Papers
2013Volume 29Issue 8 Pages 831-835
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESSAn online preconcentration technique, large volume sample stacking with an electroosmotic flow pump, was combined with partial filling affinity capillary electrophoresis (PFACE) to create a highly sensitive analysis of the interaction of glycoprotein-derived oligosaccharides with plant lectins. Oligosaccharides were derivatized with 8-aminonaphthalene-1,3,6-trisulfonic acid (ANTS) for use in a blue light emitting diode-induced fluorescence detection capillary electrophoresis system. ANTS-labeled oligosaccharides were delivered to an entire neutrally coated capillary, and lectin solution was then hydrodynamically introduced from the outlet of the capillary as a short plug. When negative voltage was then applied, a low concentration sample solution caused a significant flow by electroosmosis from anode to cathode and the ANTS-labeled oligosaccharides moved quickly towards the anode and concentrated in the lectin phase. Finally, when the electroosmotic flow became negligible, ANTS-labeled saccharides passed through the lectin plug and were detected at the anodic end. The sensitivity was enhanced by a factor of roughly 200 compared to typical hydrodynamic injection (13.8 kPa, 5 s).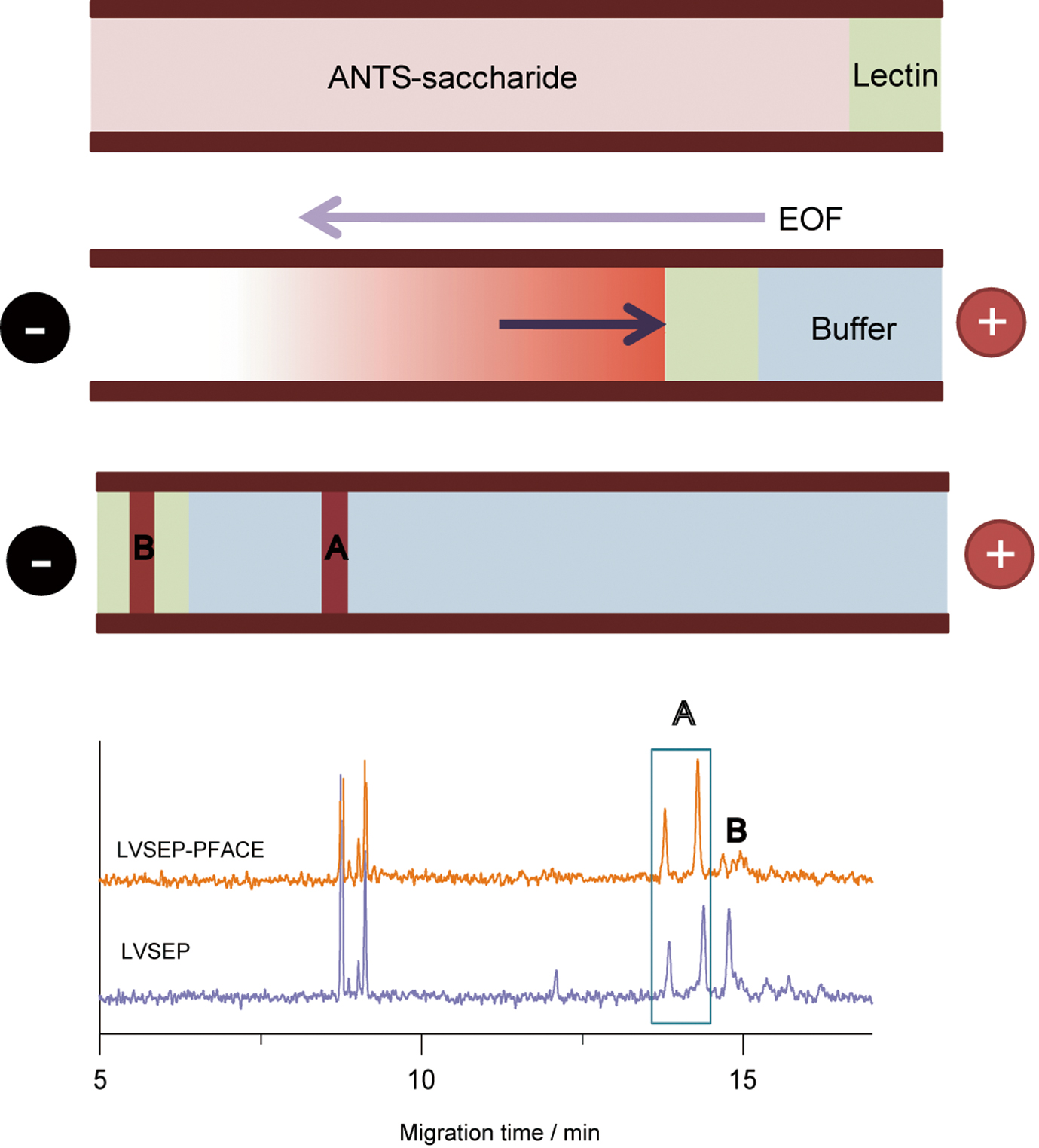 View full abstractDownload PDF (1208K)
View full abstractDownload PDF (1208K) -
Hossein Ali MASHAYEKHIArticle type: Original Papers
2013Volume 29Issue 8 Pages 837-841
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESSHomogeneous liquid–liquid extraction via flotation assistance (HoLLE-FA) and gas chromatography–flame ionization detection (GC-FID) was presented for the extraction and determination of fenitrothion in water samples. In this work, a rapid, simple and efficient HoLLE-FA method was developed based on applying low-density organic solvents without employing centrifugation. A special extraction cell was designed to facilitate the collection of low-density solvent extraction in the determination of fenitrothion in water samples. The water sample solution was added into an extraction cell that contained an appropriate mixture of extraction and homogeneous solvents. By using air flotation, the organic solvent was collected at the conical part of the designed cell. Under the optimum conditions, the method performance was studied in terms of the linear dynamic range (LDR from 1.0 up to 100 μg L−1), linearity (r2 > 0.998), and precision (repeatability < 8.0%). Also, limit of detection (LOD) of 0.4 μg L−1 was obtained for fenitrothion. The applicability of the HoLLE-FA method was evaluated by the extraction and determination of fenitrothion in three different water samples. View full abstractDownload PDF (1474K)
View full abstractDownload PDF (1474K) -
Mayumi ETOU, Tsutomu KURISAKI, Yoshihiro OKAUE, Hisanobu WAKITA, Takus ...Article type: Original Papers
2013Volume 29Issue 8 Pages 843-848
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESS
Supplementary materialDue to a consideration of Al detoxicification by simple carboxylic acid, the interaction between aluminium ion (Al3+) and three dicarboxylic acids (oxalic acid (OX), malonic acid (MA) and succinic acid (SU)) under an acidic condition was investigated using 13C and 27Al NMR techniques. Based on deconvolution of the 27Al NMR spectra and quantitative 13C NMR spectra, the structure of each complex was elucidated. Especially, we focused on the peak assignments of 27Al NMR spectra by combining of the results of quantitative 13C NMR spectra. In the OX system, the peak at 16 ppm in the 27Al NMR spectrum originates from Al(OX)33− and Al(OX)2−, and the ratio of each complex depends on the OX/Al molar ratio. In the MA system, the three complexes (Al(MA)2−, Al(MA)33− and Al(MA)+) are represented in the peak at 2 ppm in the 27Al NMR spectrum. The assignment of peaks in the 27Al NMR spectra in this study differs from those described in previous papers.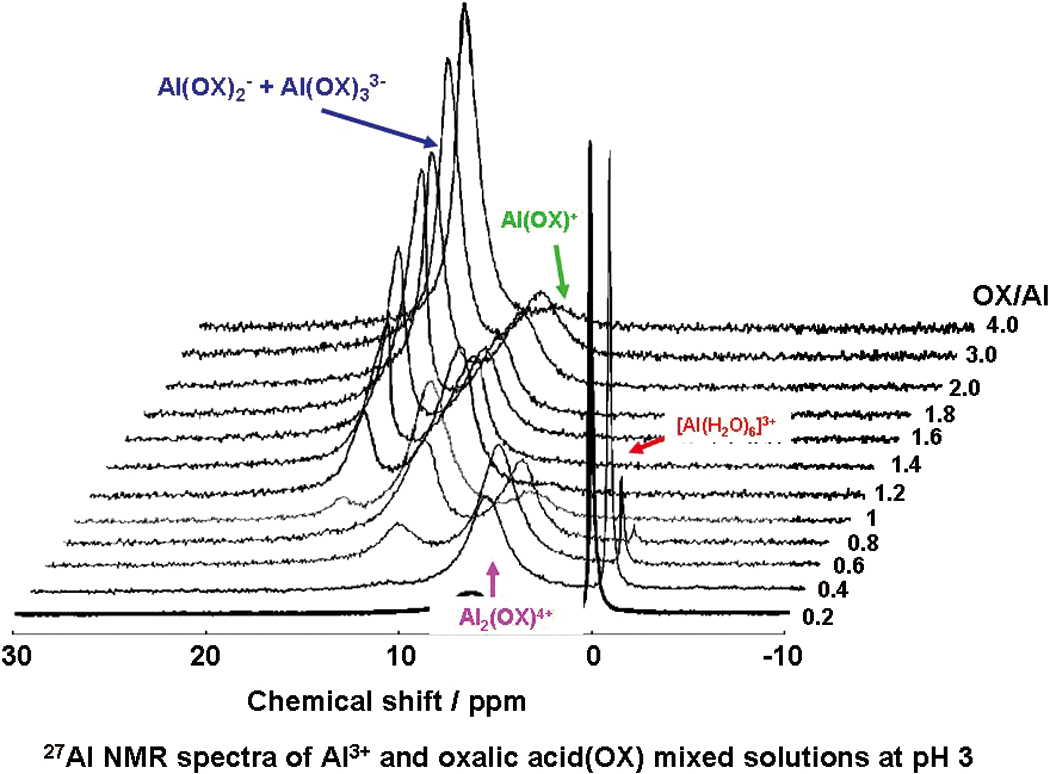 View full abstractDownload PDF (622K)
View full abstractDownload PDF (622K) -
Chang-Jin MA, Kyung-Chul HWANG, Ki-Hyun KIMArticle type: Original Papers
2013Volume 29Issue 8 Pages 849-853
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESSThis study was carried out to describe the chemical characteristics of individual residual particles in hexagonal snow crystals, which can provide a clue to the aerosol removal mechanism during snowfall. In the present study, to collect snow crystal individually and to identify the elemental composition of individual residues retained in a hexagonal crystal, an orchestration of the replication technique and micro-particle induced X-ray emission (micro-PIXE) analysis was carried out. Information concerning the elemental compositions and their abundance in the snow crystals showed a severe crystal-to-crystal fluctuation. The residues retained in the hexagonal snow crystals were dominated primarily by mineral components, such as silica and calcium. Based on the elemental mask and the spectrum of micro-PIXE, it was possible to presume the chemical inner-structure as well as the elemental mixing state in and/or on the individual residues retained in single snow crystals. View full abstractDownload PDF (10673K)
View full abstractDownload PDF (10673K) -
Elham ARKAN, Ziba KARIMI, Mojtaba SHAMSIPUR, Reza SABERArticle type: Original Papers
2013Volume 29Issue 8 Pages 855-860
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESSA graphene-based carbon ionic liquid electrode modified with gold nanoparticles was fabricated. The electrochemical response of the modified electrode toward celecoxib was studied by means of cyclic voltammetry and differential pulse voltammetry. The structural morphology of the modified electrode was characterized by a scanning electron microscopy technique and electrochemical impedance spectroscopy. The prepared electrode showed excellent electrocatalytic activity in the reduction of celecoxib in a phosphate buffer solution, leading to remarkable enhancements in the corresponding peak currents and lowering of the peak potential. The advantages are related to the unique properties of graphene and gold nanoparticles such as a large surface area and increased electron-transfer abilities. Differential pulse voltammetry was applied to the quantitative determination. The calibration curve was linear in the concentration range of 0.5 to 15 μM and the detection limit was about 0.2 μM (Sb/N = 3). The proposed electrochemical sensor was successfully applied to the determination of celecoxib in real samples.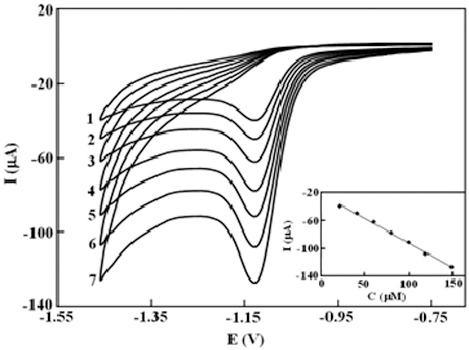 View full abstractDownload PDF (913K)
View full abstractDownload PDF (913K)
Announcements
-
Article type: Announcements
2013Volume 29Issue 8 Pages 861
Published: August 10, 2013
Released on J-STAGE: August 10, 2013
JOURNAL FREE ACCESSDownload PDF (1689K)
- |<
- <
- 1
- >
- >|