All issues

Volume 31, Issue 4
Displaying 1-14 of 14 articles from this issue
- |<
- <
- 1
- >
- >|
-
Article type: Guest Editorial
2015 Volume 31 Issue 4 Pages 243
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESSDownload PDF (110K)
Reviews
-
Sung-Jin PARK, Jun-Young KIM, Chai Lean TEOH, Nam-Young KANG, Young-Ta ...Article type: Reviews
2015 Volume 31 Issue 4 Pages 245-255
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESSAtherosclerosis is an inflammatory disease in which immune mechanisms interact with diverse metabolic risk factors to initiate, propagate and activate lesions in the arterial wall. This process starts and evolves in response to cholesterol accumulation in arterial intima of the large and medium arteries. A number of modalities for inflammatory imaging in the arterial wall are currently in the stages of pre-clinical and clinical development. However, the identification of new targets that are linked to atherosclerosis to allow for dynamic ranges of diagnosis and prognosis still remains a challenge. In this review, we summarize the current status of atherosclerosis imaging aimed at macrophage-related, direct macrophage and non-macrophage as target molecules during different stages of atherogenesis. We also present activated macrophage, biofilm and amyloid fibrils as possible new targets for the development of non-invasive imaging probes to assess atherosclerosis. View full abstractDownload PDF (936K)
View full abstractDownload PDF (936K) -
Toru KOMATSU, Yasuteru URANOArticle type: Reviews
2015 Volume 31 Issue 4 Pages 257-265
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESSIn this review, we aim to present an overview of how small-molecular fluorescent substrate probes for studying enzymatic functions are developed and how they are used in biological applications, under the following four headings: (1) History of Visual Detection of Enzymatic Activities, (2) Strategies to Design Fluorescent Substrate Probes to Measure Enzymatic Activities, (3) Development of Fluorescent Substrate Probes Suitable for Biological Studies, and (4) Biological Applications of Fluorescent Substrate Probes for Studying Enzymes. View full abstractDownload PDF (1597K)
View full abstractDownload PDF (1597K) -
Yoichiro FUJIOKA, Asuka NANBO, Shin-ya NISHIDE, Yusuke OHBAArticle type: Reviews
2015 Volume 31 Issue 4 Pages 267-274
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESSIn response to extracellular stimuli, cells display a variety of behaviors, including proliferation, differentiation, morphological changes and migration. The analysis of the spatiotemporal regulation of signal transduction in living cells is needed for a better understanding of such behaviors, and such investigations have been greatly accelerated by the development of fluorescent protein-based biosensors. Currently, by using these biosensors a range of molecular actions, including lipid metabolism, protein activation, and ion dynamics, can be visualized in living cells. We recently reported that intracellular calcium, with its relevant downstream signaling pathways consisting of the small GTPase Ras and the lipid kinase phoshoinositide-3-kinase (PI3K), can be exploited in an efficient incorporation of influenza A viruses into host cells via endocytosis using a set of biosensors based on fluorescent proteins and the principle of Förster resonance energy transfer. Here, we focus this review on fluorescent protein-based biosensors that have been utilized in our recent research reports. View full abstractDownload PDF (3933K)
View full abstractDownload PDF (3933K) -
Takakazu NAKABAYASHI, Nobuhiro OHTAArticle type: Reviews
2015 Volume 31 Issue 4 Pages 275-285
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESSFluorescence lifetime imaging (FLIM) has been recognized as a powerful microscopy technique to examine environments in living systems. The fluorescence lifetime does not depend on the photobleaching and optical conditions, which allows us to obtain quantitative information on intracellular environments by analyzing the fluorescence lifetime. A variety of exogenous fluorophores have been applied in FLIM measurements to examine cellular processes. Information on the correlation between the fluorescence lifetime and the physiological parameters is essential to elucidate the cellular environments from the fluorescence lifetime measurements of exogenous fluorophores. In this review, exogenous fluorophores used for lifetime-based sensing are summarized, with the expectation that it becomes a basis for selecting the fluorophore used to investigate the intracellular environment with FLIM. Experimental results of the intracellular sensing of pH, metal ions, oxygen, viscosity, and other physiological parameters on the basis of the FLIM measurements are described along with a brief explanation of the mechanism of the change in the fluorescence lifetime.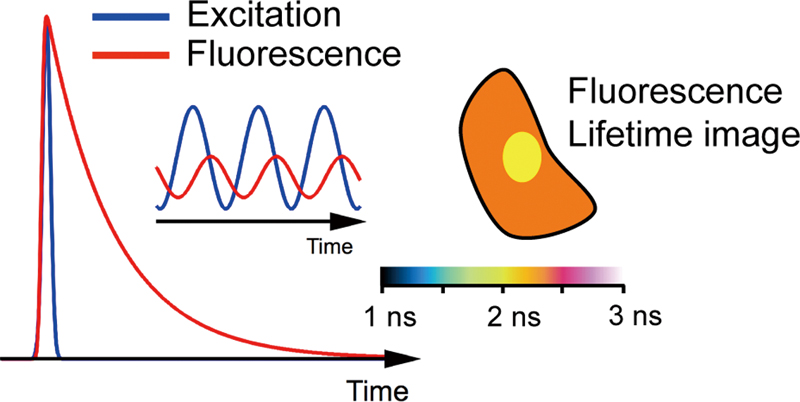 View full abstractDownload PDF (1681K)
View full abstractDownload PDF (1681K) -
Masafumi MINOSHIMA, Kazuya KIKUCHIArticle type: Reviews
2015 Volume 31 Issue 4 Pages 287-292
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESSHistone deacetylases (HDACs) enzymes are responsible for removing epigenetic markers on histone proteins, which results in chromatin inactivation and gene repression. An evaluation of HDAC activity is essential for not only determining the physiological function of HDACs, but also for developing HDAC-targeting drugs. This review focuses on the chemical tools used to detect HDAC activity. We highlight activity-based probes and positron emission tomography probes based on the chemical structure of the inhibitors. We also summarize fluorogenic probes used in single-step methods for HDAC detection. These fluorogenic probes are designed based on the nucleophilicity of the amino group, aggregation via electrostatic interactions, and changes in the DNA binding properties. These fluorogenic systems may enable facile and rapid screening to evaluate HDAC inhibitors, which will contribute to the development of epigenetic drugs. View full abstractDownload PDF (1169K)
View full abstractDownload PDF (1169K) -
Muhammad AWAIS, Svetlana G VORONINA, Robert SUTTONArticle type: Reviews
2015 Volume 31 Issue 4 Pages 293-298
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESSGenetically encoded fluorescent and bioluminescent reporters are now widely used for imaging and understanding of intracellular signaling in response to extracellular stimuli in real time in single living cells. Primary cultures are a valuable tool, and are often preferred over transformed or immortalized cell lines, since they are biologically more relevant and important in biomedical research and therapeutic development. To incorporate genetically encoded reporters into the primary culture of non-dividing cells, such as mouse or human pancreatic acinar cells, is not an easy task. This short review discusses the different methods available to transfect cell lines and primary cultures while especially focusing on pancreatic acinar cells with genetically encoded optical reporters to advance knowledge of the pathophysiology of pancreatitis View full abstractDownload PDF (687K)
View full abstractDownload PDF (687K)
Original Papers
-
Hiroki SEGAWA, Masanari OKUNO, Philippe LEPROUX, Vincent COUDERC, Take ...Article type: Original Papers
2015 Volume 31 Issue 4 Pages 299-305
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESSThe subnanosecond “white-light laser” source has been applied to multimodal, multiphoton, and multiplex spectroscopic imaging (M3 spectroscopic imaging) with coherent anti-Stokes Raman scattering (CARS), third-order sum frequency generation (TSFG), and two-photon excitation fluorescence (TPEF). As the proof-of-principle experiment, we performed simultaneous imaging of polystyrene beads with TSFG and TPEF. This technique is then applied to live cell imaging. Mouse L929 fibroblastic cells are clearly visualized by CARS, TSFG, and TPEF processes. M3 spectroscopic imaging provides various and unique cellular information with different image contrast based on each multiphoton process.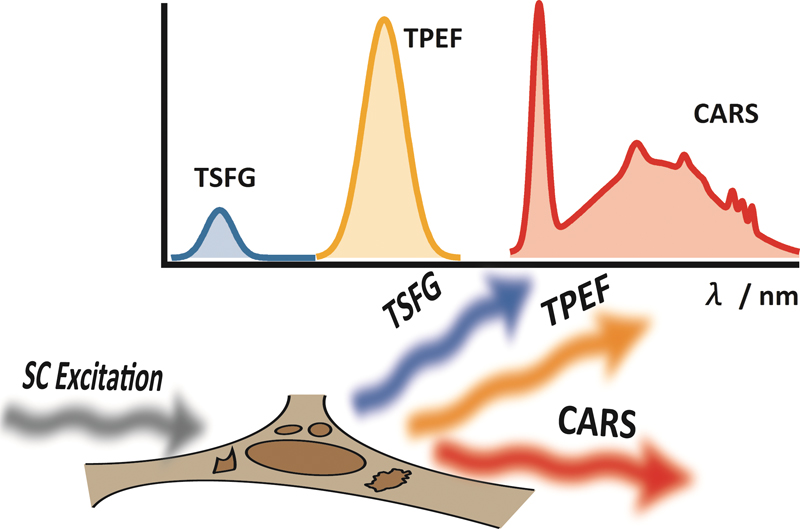 View full abstractDownload PDF (2808K)
View full abstractDownload PDF (2808K) -
Kohei OTOMO, Terumasa HIBI, Takashi MURATA, Hirotaka WATANABE, Ryosuke ...Article type: Original Papers
2015 Volume 31 Issue 4 Pages 307-313
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESS
Supplementary materialThe temporal resolution of a two-photon excitation laser scanning microscopy (TPLSM) system is limited by the excitation laser beam’s scanning speed. To improve the temporal resolution, the TPLSM system is equipped with a spinning-disk confocal scanning unit. However, the insufficient energy of a conventional Ti:sapphire laser source restricts the field of view (FOV) for TPLSM images to a narrow region. Therefore, we introduced a high-peak-power Yb-based laser in order to enlarge the FOV. This system provided three-dimensional imaging of a sufficiently deep and wide region of fixed mouse brain slices, clear four-dimensional imaging of actin dynamics in live mammalian cells and microtubule dynamics during mitosis and cytokinesis in live plant cells.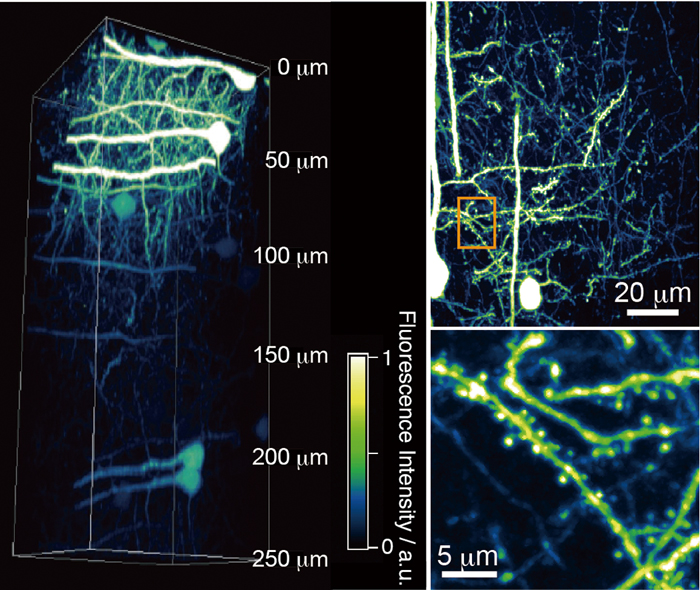 View full abstractDownload PDF (1086K)
View full abstractDownload PDF (1086K) -
Takaya SATO, Yusuke SATO, Kenta IWAI, Shusuke KUGE, Norio TERAMAE, Sei ...Article type: Original Papers
2015 Volume 31 Issue 4 Pages 315-320
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESS
Supplementary materialWe report on the use of a peptide nucleic acid (PNA)-based fluorescent probe for the analysis of siRNA delivery to living cells. The probe, Py-AA-TO, possesses thiazole orange (TO) and pyrene moieties in the C- and N-termini of PNA, and can function as a light-up probe capable of selective binding to 3′-overhanging nucleotides of target siRNAs. The affinity-labeling of the siRNAs with Py-AA-TO facilitates fluorescence imaging of cellular uptake of polymer-based carriers encapsulating the siRNAs (polyplexes) through endocytosis and subsequent sequestration into lysosome. In addition, flow cytometric measurements reveal that the monitoring of Py-AA-TO fluorescence inside the cells is successfully applicable to the analysis of the polyplex disassembly. These promising functions of Py-AA-TO are presented and discussed as a basis for the design of molecular probes for fluorescent imaging and quantitative analysis of the siRNA delivery process.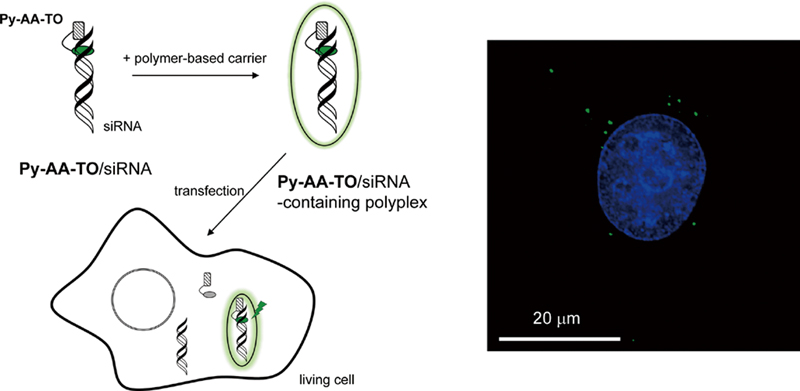 View full abstractDownload PDF (627K)
View full abstractDownload PDF (627K) -
Kazuhisa TANAKA, Atushi SHOJI, Masao SUGAWARAArticle type: Original Papers
2015 Volume 31 Issue 4 Pages 321-325
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESS
Supplementary materialAn agarose gel slip containing L-glutamate oxidase (GluOx), horseradish peroxidase (HRP) and a dye DA-64 is proposed as a tool for visualizing ischemia-induced L-glutamate release in hippocampal slices in a flow system. The agarose slip with a detection limit of 6.0 ± 0.8 μmol L−1 for L-glutamate enabled us to visualize L-glutamate fluxes in a flow system. The leak of a dye from the agarose gel was negligible and a diffusion blur due to spreading of Bindshedler’s Green (BG) within the gel was suppressed. Monitoring the time-dependent change of ischemia-induced L-glutamate fluxes at neuronal regions CA1, DG and CA3 of hippocampal slices is demonstrated. View full abstractDownload PDF (1238K)
View full abstractDownload PDF (1238K)
Notes
-
Mitsuru HATTORI, Takeaki OZAWAArticle type: Notes
2015 Volume 31 Issue 4 Pages 327-330
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESS
Supplementary materialFor the design of therapeutic drugs, G protein coupled receptors (GPCRs) are notable targets. Many screening methods have been developed to identify effective agents for GPCR signaling. However, analyses of temporal variations of GPCR activity with specific ligands remain insufficient because of monitoring method limitations and difficulties. We previously developed a high-throughput bioluminescence measuring system to detect interactions of GPCR with β-arrestin based on split luciferase fragment complementation. By newly introducing a bioluminescence imaging technique into the system, we demonstrate a method for the temporal monitoring of GPCR–β-arrestin interactions in living cells during stimulation by different ligands.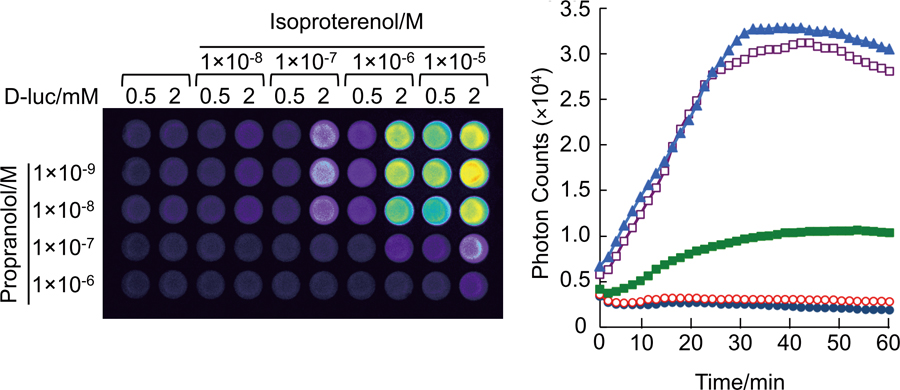 View full abstractDownload PDF (1481K)
View full abstractDownload PDF (1481K) -
Hiroshi NONAKA, Qi AN, Fuminori SUGIHARA, Tomohiro DOURA, Akira TSUCHI ...Article type: Notes
2015 Volume 31 Issue 4 Pages 331-335
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESS
Supplementary materialHerein, we report on a new 19F MRI probe for the detection and imaging of H2O2. Our designed 2-fluorophenylboronic acid-based 19F probe promptly reacted with H2O2 to produce 2-fluorophenol via boronic acid oxidation. The accompanying 19F chemical-shift change reached 31 ppm under our experimental conditions. Such a large chemical-shift change allowed for the imaging of H2O2 by 19F chemical-shift-selective MRI.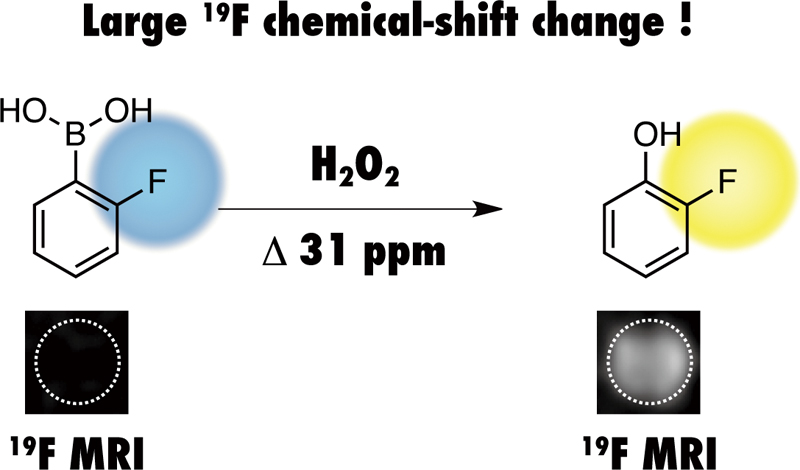 View full abstractDownload PDF (1126K)
View full abstractDownload PDF (1126K)
Announcements
-
Article type: Announcements
2015 Volume 31 Issue 4 Pages 337
Published: April 10, 2015
Released on J-STAGE: April 10, 2015
JOURNAL FREE ACCESSDownload PDF (2640K)
- |<
- <
- 1
- >
- >|