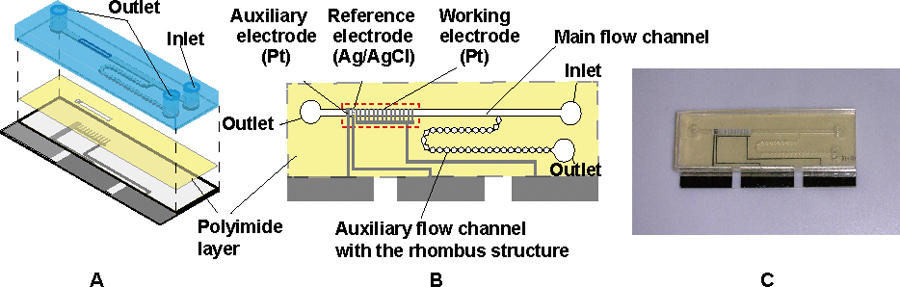
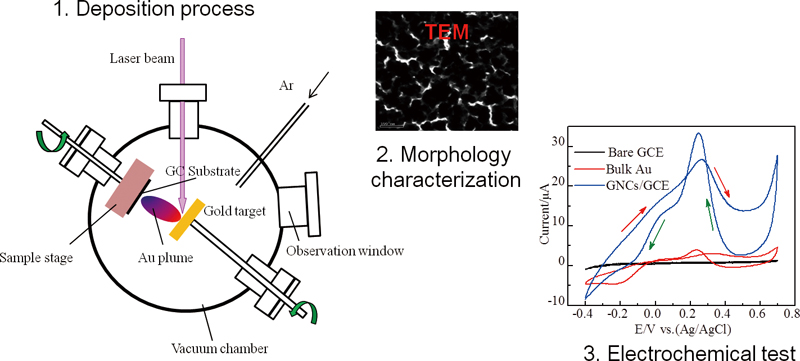
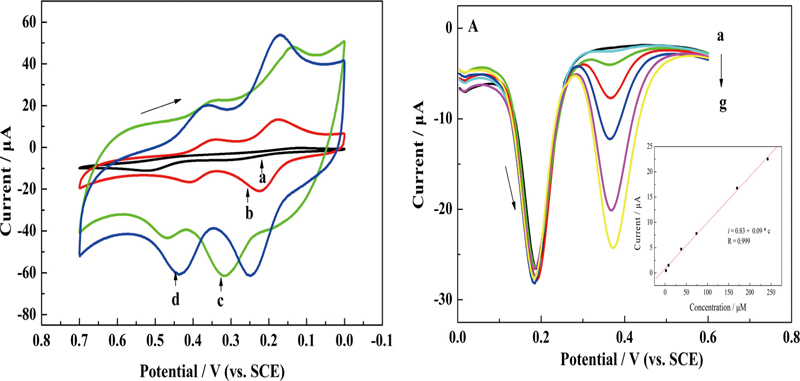
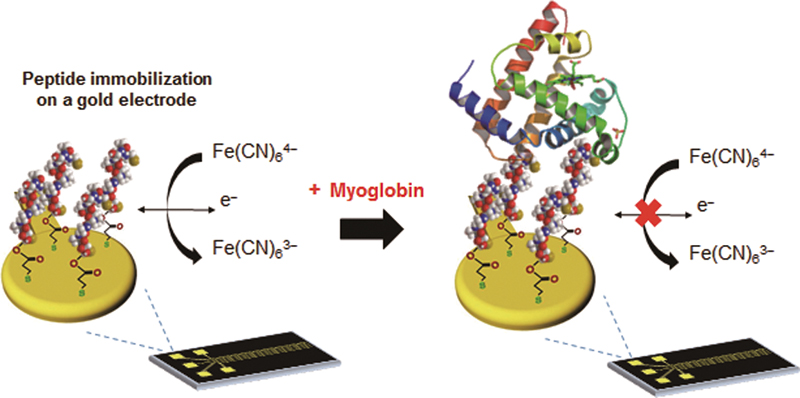
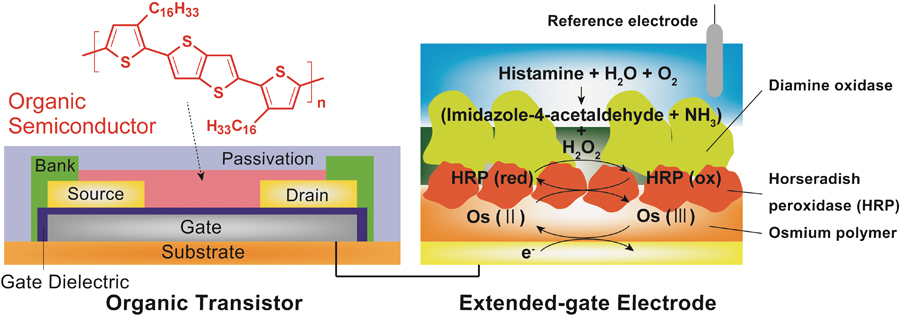
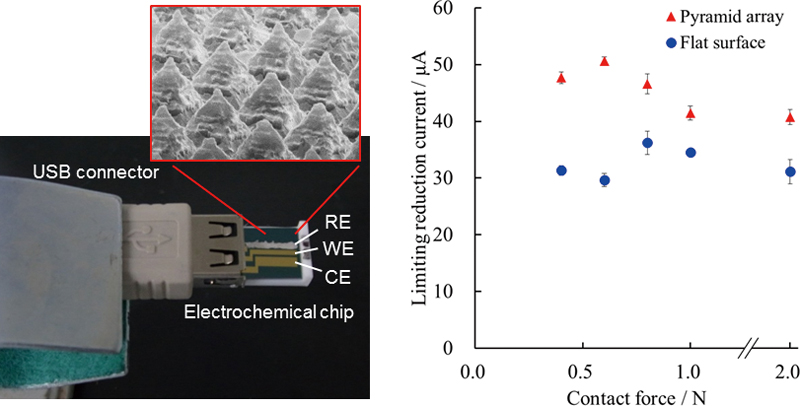
Volume 31, Issue 7
Displaying 1-30 of 30 articles from this issue
- |<
- <
- 1
- >
- >|
-
2015Volume 31Issue 7 Pages 571
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (334K)
Rapid Communications
-
Article type: Rapid Communications
2015Volume 31Issue 7 Pages 573-576
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (2590K) -
Article type: Rapid Communications
2015Volume 31Issue 7 Pages 577-579
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (897K)
Guest Editorial
-
Article type: Guest Editorial
2015Volume 31Issue 7 Pages 581
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (114K)
Original Papers
-
Article type: Original Papers
2015Volume 31Issue 7 Pages 583-589
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (4228K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 591-595
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (6419K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 597-602
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (995K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 603-607
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (746K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 609-616
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (7846K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 617-621
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (972K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 623-627
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (563K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 629-634
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (916K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 635-641
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (678K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 643-649
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (835K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 651-656
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (645K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 657-662
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (2032K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 663-667
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (821K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 669-675
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (1648K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 677-683
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (1630K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 685-691
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (1063K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 693-697
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (794K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 699-704
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (2502K) -
Interference Reduction in Glucose Detection by Redox Potential Tuning: New Glucose Meter DevelopmentArticle type: Original Papers
2015Volume 31Issue 7 Pages 705-710
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (1649K) -
Article type: Original Papers
2015Volume 31Issue 7 Pages 711-714
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (723K)
Notes
-
Article type: Notes
2015Volume 31Issue 7 Pages 715-719
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (12335K) -
Article type: Notes
2015Volume 31Issue 7 Pages 721-724
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (1152K) -
Article type: Notes
2015Volume 31Issue 7 Pages 725-728
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (824K) -
Article type: Notes
2015Volume 31Issue 7 Pages 729-732
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (2030K) -
Article type: Notes
2015Volume 31Issue 7 Pages 733-735
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (438K)
Announcements
-
Article type: Announcements
2015Volume 31Issue 7 Pages 737
Published: July 10, 2015
Released on J-STAGE: July 10, 2015
Download PDF (2739K)
- |<
- <
- 1
- >
- >|