
- |<
- <
- 1
- >
- >|
-
Masaki Tsumadori2019 Volume 68 Issue 6 Pages 501
Published: 2019
Released on J-STAGE: June 06, 2019
JOURNAL FREE ACCESSDownload PDF (113K) -
Ken D. Stark2019 Volume 68 Issue 6 Pages 503-510
Published: 2019
Released on J-STAGE: June 06, 2019
Advance online publication: May 16, 2019JOURNAL FREE ACCESS FULL-TEXT HTMLAssessing dietary intake is critical for understanding the relationship between diet and health. Fatty acid blood biomarkers have been particularly useful in determining dietary intakes and assessing the risk of chronic disease. However, fatty acid analysis involves the removal of fatty acids from their complex lipid structures resulting in a loss of potentially useful biological information. “Lipidomics” involves the use of mass spectrometry to identify lipids in their native form. Lipidomic approaches present challenges as an alternative to fatty acid analysis. This includes different types of lipidomic approaches and a lack of consensus on the lipids reported in different studies. Distinguishing between macrolipidomic approaches to characterize highly abundant lipids and microlipidomic approaches examining low abundant bioactive lipids and the use of brutto, medio, genio, and infinio to describe the level of information of lipidomic data can provide clarity to the field. Using lipidomic measurements for understanding docosahexaenoic acid metabolism during pregnancy will also be examined.
 graphical abstract Fullsize ImageView full abstractDownload PDF (239K) Full view HTML
graphical abstract Fullsize ImageView full abstractDownload PDF (239K) Full view HTML -
Kei Hamazaki2019 Volume 68 Issue 6 Pages 511-515
Published: 2019
Released on J-STAGE: June 06, 2019
Advance online publication: May 16, 2019JOURNAL FREE ACCESS FULL-TEXT HTMLOmega-3 polyunsaturated fatty acids is a generic term that encompasses eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), which are abundant in fish, and α-linolenic acid, which is abundant in perilla oil. These fatty acids became well known following an epidemiological study conducted in Greenland about 50 years ago. EPA formulations, with their putative prophylactic effect against cardiovascular disease, first became clinically available in the 1990s in Japan. There were reports of their association with mental health, especially depression, and from around the year 2000 several epidemiological studies and clinical trials were conducted. This article introduces the epidemiological studies and clinical trials conducted in Japan and the knowledge gained.
View full abstractDownload PDF (344K) Full view HTML -
Bungo Shirouchi, Ryosuke Matsuoka2019 Volume 68 Issue 6 Pages 517-524
Published: 2019
Released on J-STAGE: June 06, 2019
JOURNAL FREE ACCESS FULL-TEXT HTMLAbdominal fat accumulation causes metabolic syndrome, which is a cluster of metabolic abnormalities such as dyslipidemia, glucose intolerance, insulin resistance or hyperinsulinemia, and hypertension, leading to the development of diabetes and cardiovascular disease. Diets are known to contribute to the development or prevention of metabolic syndrome. Several studies have reported that the quality of dietary proteins may be an important modulator of the risk of this syndrome. We investigated the effects of consuming egg white protein (EWP) or lactic-fermented egg white (LE), an easy-to-consume form of egg white, on the development of metabolic syndrome in animal models and humans. In comparison with casein, dietary EWP decreased lymphatic lipid transport in thoracic lymph duct-cannulated rats. In an in vitro experiment, EWP pepsin hydrolysate decreased the cholesterol micellar solubility and cholesterol transfer rate from micelles to oil phase, and increased water-holding capacity, settling volume in water, and relative viscosity compared with casein pepsin hydrolysate. The daily consumption of LE for 8 weeks reduced serum total cholesterol and LDL cholesterol levels in men with mild hypercholesterolemia. Furthermore, dietary EWP reduced the body fat mass of rats by increasing the body protein mass and accelerating hepatic β-oxidation. The daily consumption of LE for 12 weeks reduced the visceral fat area and improved the ratio of the visceral to subcutaneous fat area. Taken together, these results indicated that dietary EWP and LE would be useful for preventing or alleviating metabolic syndrome.
 graphical abstract Fullsize ImageView full abstractDownload PDF (993K) Full view HTML
graphical abstract Fullsize ImageView full abstractDownload PDF (993K) Full view HTML -
Yasushi Kakizawa, Miyuki Miyake2019 Volume 68 Issue 6 Pages 525-539
Published: 2019
Released on J-STAGE: June 06, 2019
Advance online publication: May 16, 2019JOURNAL FREE ACCESS FULL-TEXT HTMLThe combination of polymers and surfactants is an important means to create various functions in recent detergents and personal care products. In particular, detergents mixing oppositely charged anionic surfactants and cationic polymers induce coacervation by the dilution of the washing and rinsing process, and the complexes effectively adsorb onto surfaces and can change their characteristics. The driving force of the coacervation is electrostatic interaction between the anionic groups of the surfactant and the cationic groups of the polymer. Normally, the coacervation is controlled by selecting the molecular structure or the amount of polymer and surfactant. In shampoo and body wash compositions, we studied the complex precipitation (CP) regions and the morphology and rheological properties of precipitated complexes by focusing on the number of ionic groups in the anionic surfactants and cationic polymers, the mixed electrolyte and the ionic strength as a whole. This clarified the factors related to complex functions. For coacervation in shampoo based on alkyl ethoxylate sulfate (AES), the degree of cationization of the cationic cellulose (CC) and coexisting electrolyte greatly contributed to these functions. In a combination of moderately cationically charged CC and AES mixed amphoteric surfactant, the precipitated complexes became a loose mesh-like morphology, which was also formed when the charge shielding effect was enhanced by adding electrolyte. The precipitated complexes with a looser mesh-like morphology gave a smooth texture to the hair surface during rinsing.
On the other hand, for coacervation in body wash based on fatty acid salt, the complexes were effectively precipitated in a combination with a synthetic polymer, poly diallyldimethylammonium chloride (PDADMAC), which has a higher cationic charge than CC. The precipitated complexes had high adsorbability onto skin and contributed to a moisturizing effect by lowering transepidermal water loss (TEWL).
In this review, we introduce the controllable factors of coacervation in shampoo and body wash systems by focusing on the relationship between dilution processes and precipitation behavior.
 graphical abstract Fullsize ImageView full abstractDownload PDF (3134K) Full view HTML
graphical abstract Fullsize ImageView full abstractDownload PDF (3134K) Full view HTML -
Charose Marie Ting Perez, Kenshi Watanabe, Yoshiko Okamura, Yutaka Nak ...2019 Volume 68 Issue 6 Pages 541-549
Published: 2019
Released on J-STAGE: June 06, 2019
Advance online publication: May 16, 2019JOURNAL FREE ACCESSThraustochytrids, a group of marine protists, are continuously gaining attention due to their capability in producing lipids for various biotechnological applications towards foods, medicines, chemicals, and biofuels. Although various substrates, predominantly glucose, have been used as carbon source for this microalga, it is desirable to adopt cheaper and more diversified substrate to expand their application range. In this study, we aimed to examine the ability of acetate, which can be easily generated from various resources by acetogenic microorganisms, as a substrate of Aurantiochytrium limacinum SR21. As a result of flask-scale analysis, specific growth rates (µ) of the strain SR21 grown in 3% acetate- or glucose-based medium were 0.55 and 0.98 h–1, respectively. The maximum yield of total fatty acid in acetate medium was 4.8 g/L at 48 h while that in glucose medium was 6.8 g/L at 30 h, indicating that acetate has potential as substrate. Metabolome analysis was performed to comprehensively elucidate characteristic metabolic fluctuations caused by acetate assimilation and identify targets to improve the fatty acid productivity from acetate. It was found that the use of glyoxylate cycle, which bypasses release of energy molecules such as NADH and GTP, and the inhibition of utilization of compounds from TCA cycle for anabolic reactions, may cause the slow growth in acetate which has an effect also in lipid productivity. The activity of the pentose phosphate pathway was found to be weak in acetate cultivation, thus NADPH was mainly produced in malate-pyruvate cycle. Lastly, mevalonate pathway was found to be activated in acetate cultivation which additionally competes with acetyl-CoA as starting material of fatty acid synthesis.
View full abstractDownload PDF (699K)
-
Lixia Hou, Yujin Zhang, Xuede Wang2019 Volume 68 Issue 6 Pages 551-558
Published: 2019
Released on J-STAGE: June 06, 2019
JOURNAL FREE ACCESSSesame paste is used as a condiment in many parts of the world because of its unique flavor. In the study, the volatile compounds and flavor characteristics of sesame pastes processed at different temperatures (140°C - 190°C) were analysed by application of solid phase microextraction - gas chromatography/mass spectrometry analysis, odor activity value evaluation and sensory evaluation. For all the sesame pastes processed at the different temperatures, the pyrazine group had the highest content. The amount of the volatiles increased by 4.15-fold when the roasting temperature increased from 140°C to 180°C, which was mainly attributed to the increase of pyrazines, alkenes, ketones, thiazoles and pentylfuran. And accordingly, sesame paste prepared at 180°C had the highest Odor Activity Value (OAV). Samples differed significantly in terms of aroma and total acceptability (p < 0.05). The sesame paste processed at 180°C had a moderate roasted and sweet aroma, and the highest acceptability, followed by that processed at 170°C. These results can be valuable for manufacturers to adjust the roasting process according to market demand for a particular flavor profile.
View full abstractDownload PDF (323K) -
Jinfeng Qi, Xiaosan Wang, Xingguo Wang, Casimir C. Akoh, Qingzhe Jin2019 Volume 68 Issue 6 Pages 559-566
Published: 2019
Released on J-STAGE: June 06, 2019
Advance online publication: May 16, 2019JOURNAL FREE ACCESSThe effects of four different frying oils and three emulsifiers on oil absorption by steam-and-fried instant noodles were evaluated. The blended oil (high oleic sunflower oil/soybean oil/palm oil = 24:25:1 (v/v/v)) containing approximately 50% oleic acid was chosen as the proper frying oil due to lower oil absorption by instant noodle compared to palm, soybean, and high oleic sunflower oils. Among the four oils, the interfacial tension between high oleic sunflower oil and instant noodle was the lowest (0.073 mN/m), resulting in the highest oil uptake (15.47%), while the lowest interfacial tension (0.30 mN/m) between blended oil and instant noodle resulted in the lowest oil uptake by the fried product (12.63%). Scanning electron microscopy (SEM) and confocal laser scanning microscopy (CLSM) were used to observe surface properties and oil distribution. The instant noodle fried in blended oil was found to have uniform oil distribution and smooth surface. After selecting the proper frying oil, three emulsifiers (soybean lecithin, Tween-80, Span-80, at 0.2% (v/v)) were added to the blended frying oil. Adding emulsifier into frying oil significantly decreased the interfacial tension between frying oil and instant noodle. Among the three emulsifiers, addition of soybean lecithin resulted in the lowest interfacial tension (0.010 mN/m) and the highest oil uptake (18.36%). Therefore, from this study, we do not recommend adding emulsifier into frying oil.
View full abstractDownload PDF (1033K)
-
Kenichi Sakai, Ryo Ishimado, Yuichiro Takamatsu, Masaaki Akamatsu, Hid ...2019 Volume 68 Issue 6 Pages 567-572
Published: 2019
Released on J-STAGE: June 06, 2019
Advance online publication: May 16, 2019JOURNAL FREE ACCESS
Supplementary materialWe characterized the friction and adsorption properties of an oleic acid-based gemini amphiphile having two carboxylic acid headgroups. We employed silica as a solid material, and diethyl sebacate and bis (2-ethylhexyl) sebacate as polar ester oils. Oleic acid and stearic acid were used as comparative amphiphilic materials. These amphiphiles were soluble in the ester oils, and the solubility of the gemini amphiphile was lower than that of the other two amphiphiles. Quartz crystal microbalance with dissipation monitoring measurements suggested that the gemini amphiphile had greater adsorption capability than the two comparative amphiphiles. The greater adsorption density of the gemini amphiphile resulted in the formation of a rigid interfacial film, as suggested by the normal force curves obtained by atomic force microscopy (AFM). We assessed the friction property of these systems using a ball-on-plate-type friction analyzer and by friction-mode AFM (friction force curve). These measurements confirmed that the gemini amphiphile had a smaller kinetic friction coefficient than that of the other two amphiphiles. These results suggest the potential of the gemini amphiphile as a friction modifier in polar oils.
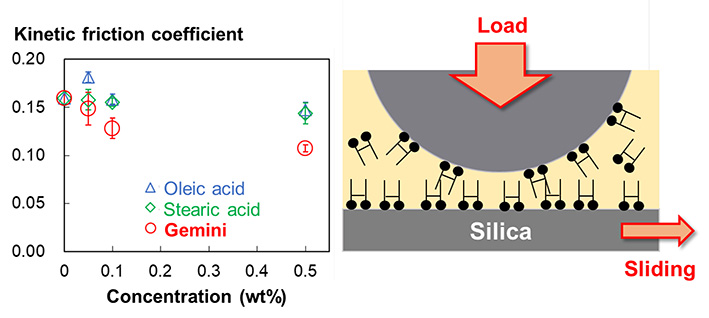 graphical abstract Fullsize ImageView full abstractDownload PDF (313K)
graphical abstract Fullsize ImageView full abstractDownload PDF (313K) -
Tsuyoshi Asakawa, Akina Fujii, Nodoka Yoneda, Akio Ohta, Hitoshi Asaka ...2019 Volume 68 Issue 6 Pages 573-580
Published: 2019
Released on J-STAGE: June 06, 2019
Advance online publication: May 16, 2019JOURNAL FREE ACCESSThioester surfactants, [C12H25N(CH3)2(CH2)mSCOCH3] Br (C12mSAc, m = 4, 11, 12), yielded thiol surfactants via thiol-thioester exchange upon addition of dithiothreitol in aqueous solution. The thiolthioester exchange reaction was enhanced in a micellar system owing to the concentration effect. The enhanced aggregation upon deprotection of the thioacetate group was observed by dynamic light scattering measurements. The thiol surfactants yielded disulfide-linked gemini surfactants upon air oxidation and incubation with hydrogen peroxide. In contrast, the thiol surfactants yielded thioester double-tailed products by esterolytic reactions with p-nitrophenyl hexanoate. The introduction of an alkyl chain to the second side chain significantly decreased the critical micelle concentration and induced the micellar growth.
 graphical abstract Fullsize ImageView full abstractDownload PDF (1096K)
graphical abstract Fullsize ImageView full abstractDownload PDF (1096K)
-
Tunyu Jian, Chen Yu, Xiaoqin Ding, Jian Chen, Jiawei Li, Yuanyuan Zuo, ...2019 Volume 68 Issue 6 Pages 581-589
Published: 2019
Released on J-STAGE: June 06, 2019
Advance online publication: May 16, 2019JOURNAL FREE ACCESSNon-alcoholic fatty liver disease (NAFLD), a common chronic liver disease characterized by hepatic steatosis, affects 30-40% of the population in the world. The seed of Euryale ferox salisb. possesses several pharmacological actions, including metabolic syndrome. However, the seed coat of E. ferox was usually discarded as waste, which contains comparatively abundant polyphenols, and its biological activity has been rarely investigated. In this work, we evaluate the hepatoprotective effect of E. ferox seed coat extract (EFSCE), in NAFLD mice induced by high-fat diet (HFD). The HPLC-MS analysis indicated that the main components of EFSCE were polyphenols. And then, mice were treated with HFD for 4 weeks to induce NAFLD. The result showed that the body weight, weight of adipose tissue, the ratio of liver to body weight in NAFLD mice increased compared with control group. In addition, blood lipids parameters including total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL), low-density lipoprotein cholesterol (LDL) also increased in NAFLD mouse model. It was showed that, after treated with EFSCE (15 and 30 mg/kg/day) for 4 weeks, the body weight, lipids deposition in the liver and blood lipids in HFD-induced NAFLD mice markedly reduced. Compared with NAFLD mice, EFSCE administration could also prevent malondialdehyde (MDA) overproduction and strengthen Superoxide Dismutase (SOD) activity to counteract oxidative stress. Moreover, EFSCE was also found effective in reducing alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activity in HFD-induced NAFLD model, which indicated liver injury in NAFLD. Therefore, EFSCE (rich in polyphenols) is indicated as bioactive nature product for HFD-induced NAFLD treatment, by eliminating lipid accumulation and oxidative stress via regulation of IRs-1 and CYP2E1.
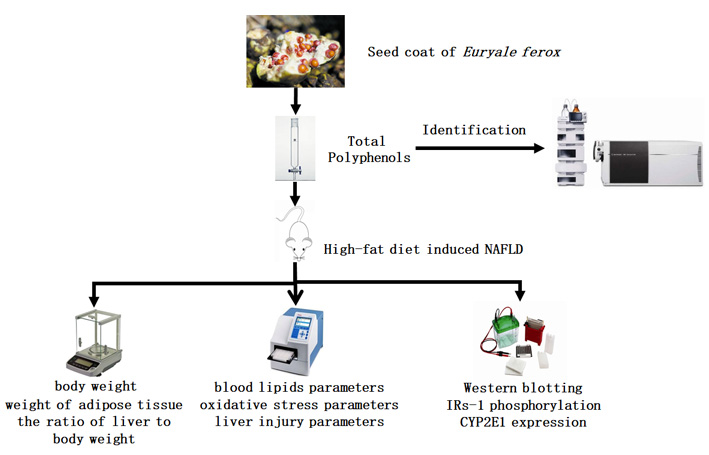 graphical abstract Fullsize ImageView full abstractDownload PDF (678K)
graphical abstract Fullsize ImageView full abstractDownload PDF (678K)
-
Kazuaki Yoshinaga, Fumiaki Beppu, Yoshio Yamatani, Atsushi Kubo, Aya Y ...2019 Volume 68 Issue 6 Pages 591-598
Published: 2019
Released on J-STAGE: June 06, 2019
Advance online publication: May 16, 2019JOURNAL FREE ACCESSFatty acids in triacylglycerol (TAG) are catabolized after digestion. However, the catabolic rates of several fatty acids bound to the α (sn-1, 3) or β (sn-2) position of TAG have not been thoroughly compared. In this study, the catabolic rates of 13C-labeled palmitic acid, oleic acid, linoleic acid, α-linolenic acid, eicosapentaenoic acid (EPA), or docosahexaenoic acid (DHA) bound to the α and β position of TAG were compared using isotope ratio mass spectrometry. The catabolic rates of the studied fatty acids were evaluated using the ratio of 13C and 12C in carbon dioxide expired from mice. The results indicated that palmitic acid, oleic acid, or α-linolenic acid bound to the β position was slowly catabolized for a long duration compared to that when bound to the α position. In contrast, EPA bound to the β position was quickly catabolized, and EPA bound to the α position was slowly catabolized for a long time. For linoleic acid or DHA, no difference in the catabolic rates was detected between the binding positions in TAG. Furthermore, EPA and DHA were less catabolized than the other fatty acids. These results indicate that the catabolic rates of fatty acids are influenced by their binding positions in TAG and that this influence on the catabolic rate differed depending on the fatty acid species.
 graphical abstract Fullsize ImageView full abstractDownload PDF (1040K)
graphical abstract Fullsize ImageView full abstractDownload PDF (1040K)
- |<
- <
- 1
- >
- >|