
- |<
- <
- 1
- >
- >|
-
 Hiromitsu Takayama2020Volume 68Issue 2 Pages 103-116
Hiromitsu Takayama2020Volume 68Issue 2 Pages 103-116
Published: February 01, 2020
Released on J-STAGE: February 01, 2020
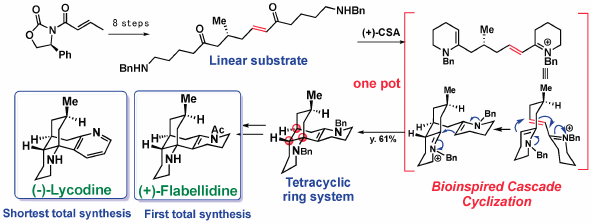
JOURNAL FREE ACCESS FULL-TEXT HTMLThe merits of biogenetic considerations in the chemical syntheses of natural products have been emphasized by describing the total syntheses of Lycopodium alkaloids; lycodine, flabellidine, lycopodine, and flabelliformine, as well as monoterpenoid indole alkaloids; C-mavacurine, kopsiyunnanine K, koumine, and 11-methoxy-19R-hydroxygelselegine.
 Graphical Abstract Fullsize ImageView full abstractEditor's pick
Graphical Abstract Fullsize ImageView full abstractEditor's pickThe lessons from nature on biosynthesis of natural products might be beneficial for synthetic organic chemists to design unique synthetic approaches as well as to facilitate development of new synthetic methodologies. This review emphasized the merits of biosynthetic consideration in the chemical synthesis of complex natural products by describing the total syntheses of Lycopodium alkaloids and monoterpenoid indole alkaloids conducted in author’s laboratory.
Download PDF (2596K) Full view HTML -
 Hirofumi Ueda2020Volume 68Issue 2 Pages 117-128
Hirofumi Ueda2020Volume 68Issue 2 Pages 117-128
Published: February 01, 2020
Released on J-STAGE: February 01, 2020
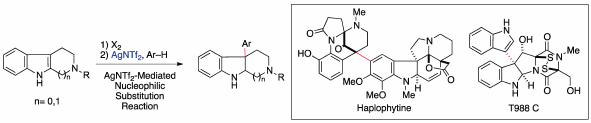
JOURNAL FREE ACCESS FULL-TEXT HTMLThe total syntheses of dimeric indole alkaloids, haplophytine, and T988s are described. These dimeric compounds comprising two structurally different indole units are ubiquitous in nature, and many possess pharmaceutically important activities. To realize an efficient chemical synthesis of these dimeric indole alkaloids, the establishment of convergent synthetic strategies and development of new coupling methods are indispensable. The linkage of two highly functionalized units at a late stage of the synthesis frequently induces synthetic problems such as chemoselectivity and steric repulsion. Moreover, although transition metal-catalyzed reactions are usually an effective method for the cross-coupling of two units, the application of these cross-coupling reactions to bond formation involving a sterically hindered C(sp3) is often difficult. Thus, even with precise modern synthetic methods, it is currently difficult to realize convergent syntheses of dimeric indole alkaloids possessing a quaternary carbon linking two units. To combat these synthetic problems, we developed a synthetic method to link two indole units using an Ag-mediated nucleophilic substitution reaction. In this review, we provide a detailed discussion of convergent synthetic strategies and coupling methods for dimeric indole alkaloids.
 Graphical Abstract Fullsize ImageView full abstractEditor's pick
Graphical Abstract Fullsize ImageView full abstractEditor's pickVarious dimeric compounds comprising two structurally different indole units are ubiquitous in nature. These compounds are a pharmaceutically important class of natural products because several compounds in this class exhibit display greater potency and unique biological activities compared with the corresponding monomeric compounds. In particular, these dimeric compounds, which possess molecular weights that deviate from Lipinski’s rule, are anticipated to be useful as new drug candidates in the middle molecule drug discovery. This review presents an overview of efficient convergent syntheses of dimeric indole alkaloids, haplophytine, and T988s with the development of synthetic methodologies for linking the two indole units.
Download PDF (2024K) Full view HTML
-
 Zhaoma Shu, Azumi Ota, Yukiya Takayama, Yuri Katsurada, Kosuke Kusamor ...2020Volume 68Issue 2 Pages 129-132
Zhaoma Shu, Azumi Ota, Yukiya Takayama, Yuri Katsurada, Kosuke Kusamor ...2020Volume 68Issue 2 Pages 129-132
Published: February 01, 2020
Released on J-STAGE: February 01, 2020
JOURNAL FREE ACCESS FULL-TEXT HTML
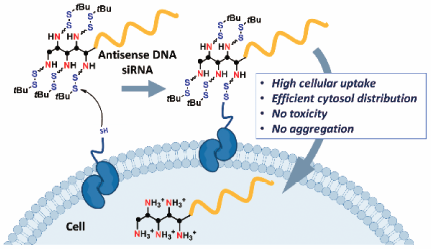
Supplementary materialEfficient methods for delivery of antisense DNA or small interfering RNA (siRNA) are highly needed. Cationic materials, which are conventionally used for anionic oligonucleotide delivery, have several drawbacks, including aggregate formation, cytotoxicity and a low endosome escape efficiency. In this report a bio-reactive mask (i.e., disulfide unit) for cationic amino groups was introduced, and the mask was designed such that it was removed at the target cell surface. Insolubility and severe cellular toxicity caused by exposed cationic groups are avoided when using the mask. Moreover, the disulfide unit used to mask the cationic group enabled direct delivery of oligonucleotides to the cell cytosol. The molecular design reported is a promising approach for therapeutic applications.
 Graphical Abstract Fullsize ImageView full abstractEditor's pick
Graphical Abstract Fullsize ImageView full abstractEditor's pickA new efficient delivery method of oligonucleotide (ON) therapeutics is developed. Here, antisense ON and small interfering RNA (siRNA) with disulfide-masked amino units were designed and synthesized for efficient intracellular delivery. The developed method actually enabled direct delivery of these ON into the cytosol, where these ON showed the targeted silencing effects, with minimal cytotoxicity. The molecular design and evaluation reported in this article would be very informative for further developing efficient cytosol-delivery methods of therapeutic ONs for medicinal application.
Download PDF (2238K) Full view HTML
-
Meng-Qiao Zhou, E Jin, Jing Wu, Fei Ren, Yu-Zhi Yang, Dong-Dong Duan2020Volume 68Issue 2 Pages 133-139
Published: February 01, 2020
Released on J-STAGE: February 01, 2020
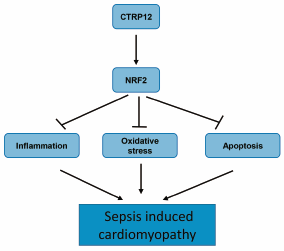
JOURNAL FREE ACCESS FULL-TEXT HTMLC1q/tumor necrosis factor (TNF)-related protein 12 (CTRP12) is a secretory protein that participates in the regulation of glucose and lipid metabolism in obesity and diabetes. Its role in cardiovascular disease, particularly sepsis-induced cardiac injury, is unclear. Here, we stimulated cardiomyocytes with lipopolysaccharide (LPS) to establish an in vitro cardiomyocyte injury model and CTRP12 was overexpressed with an adenovirus delivery system. Overexpression of CTRP12 reduced the transcription and release of pro-inflammatory cytokines from LPS-stimulated cardiomyocytes, including TNFα, interleukin-1 (IL-1), and IL-6. Reactive oxygen species (ROS) level increased and the oxidation/redox system was disturbed in LPS-stimulated cardiomyocytes, as evident from the decrease in superoxide dismutase activity and an increase in reduced nicotinamide adenine dinucleotide phosphate (NADPH) oxidase activity and malondialdehyde level. CTRP12 overexpression decreased the increasing level of ROS and ameliorated the unbalance in the oxidation/redox system in LPS-stimulated cardiomyocytes. The viability of cardiomyocytes decreased after LPS stimulation, and the cells underwent apoptosis. CTRP12-overexpressing cardiomyocytes showed a decrease in the number of terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling (TUNEL)-positive cells, and the ratio of B cell lymphoma (Bcl)-1/Bax in these cells was recovered. In comparison with the control group, LPS-stimulated cardiomyocytes showed reduced expression of nuclear factor E2-related factor 2 (NRF2), while CTRP12-overexpressing cardiomyocytes showed elevated NRF2 expression. Small-interfering RNA-mediated silencing of NRF2 expression in cardiomyocytes resulted in the inhibition of the protective effects of CTRP12. Thus, CTRP12 ameliorated injury in LPS-stimulated cardiomyocytes in an NRF2-dependent manner.
 Graphical Abstract Fullsize ImageView full abstractDownload PDF (1747K) Full view HTML
Graphical Abstract Fullsize ImageView full abstractDownload PDF (1747K) Full view HTML -
 Morio Yoshimura, Yoshiaki Amakura, Sumiko Hyuga, Masashi Hyuga, Shunsu ...2020Volume 68Issue 2 Pages 140-149
Morio Yoshimura, Yoshiaki Amakura, Sumiko Hyuga, Masashi Hyuga, Shunsu ...2020Volume 68Issue 2 Pages 140-149
Published: February 01, 2020
Released on J-STAGE: February 01, 2020
JOURNAL FREE ACCESS FULL-TEXT HTML
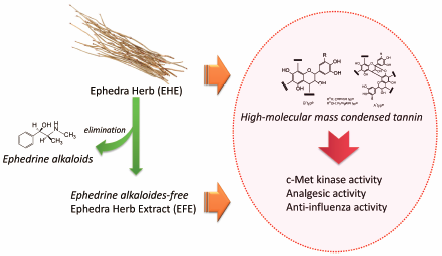
Supplementary materialPreviously, we reported that the c-Met inhibitory effect of Ephedra Herb extract (EHE) is derived from ingredients besides ephedrine alkaloids. Moreover, analgesic and anti-influenza activities of EHE and ephedrine alkaloids-free Ephedra Herb extract (EFE) have been reported recently. In this study, we examined the fractions containing c-Met kinase inhibitory activity from EHE and the fractions with analgesic and anti-influenza activities from EFE, and elucidated the structural characteristics of the active fractions. Significant c-Met kinase activity was observed in 30, 40, and 50% methanol (MeOH) eluate fractions obtained from water extract of EHE using Diaion HP-20 column chromatography. Similarly, 20 and 40% MeOH, and MeOH eluate fractions obtained from water extract of EFE were found to display analgesic and anti-influenza activities. Reversed phase-HPLC analysis of the active fractions commonly showed broad peaks characteristic of high-molecular mass condensed tannin. The active fractions were analyzed using 13C-NMR and decomposition reactions; the deduced structures of active components were high-molecular mass condensed tannins, which were mainly procyanidin B-type and partly procyanidin A-type, including pyrogallol- and catechol-type flavan 3-ols as extension and terminal units. HPLC and gel permeation chromatography (GPC) analyses estimated that the ratio of pyrogallol- and catechol-type was approximately 9 : 2, and the weight-average molecular weight based on the polystyrene standard was >45000. Furthermore, GPC-based analysis was proposed as the quality evaluation method for high-molecular mass condensed tannin in EHE and EFE.
 Graphical Abstract Fullsize ImageView full abstractEditor's pick
Graphical Abstract Fullsize ImageView full abstractEditor's pickThe structural characteristics of the fractions from Ephedra Herb extract (EHE) having c-Met kinase inhibitory effects and the fractions from ephedrine alkaloids-free Ephedra Herb extract (EFE) having analgesic and anti-influenza activities were characterized. The fractions comprised high-molecular-mass condensed tannins, which were mainly of the procyanidin B and partly procyanidin A types, including pyrogallol- and catechol-type flavan 3-ols as the extension and terminal units. HPLC and gel permeation chromatography (GPC) analyses estimated that the ratio of pyrogallol- and catechol-type was approximately 9:2 and the weight-average molecular weight based on polystyrene standard was >45,000. The authors propose GPC-based analysis as the quality method for evaluating of high-molecular-mass condensed tannin in EHE and EFE.
Download PDF (3983K) Full view HTML -
Tokuko Takajo, Yoshinori Kurihara, Kodai Iwase, Daiki Miyake, Kazunori ...2020Volume 68Issue 2 Pages 150-154
Published: February 01, 2020
Released on J-STAGE: February 01, 2020
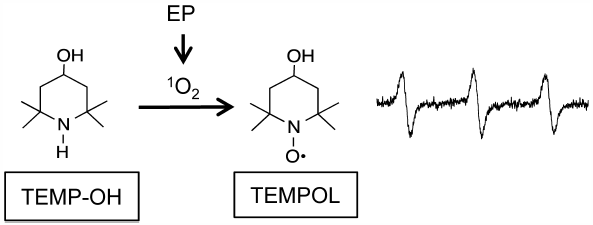
JOURNAL FREE ACCESS FULL-TEXT HTMLSinglet oxygen (1O2) is highly oxidative and exerts strong cytotoxic effects. We tried to establish the best combination of a singlet oxygen generation system and a detection method with ESR, for measurement of the quenching activities of various substances. The photosensitizing reaction of rose bengal or thermal decomposition of 4-methyl-1,4-etheno-2,3-benzodioxin-1(4H)-propanoic acid (endoperoxide, EP) was used for the generation of 1O2, and a sterically hindered secondary amine, 2,2,6,6-tetramethyl-4-piperidone (TEMPD) or 2,2,6,6-tetramethyl-4-piperidinol (TEMP-OH), was used as the 1O2 detection probe. These secondary amines were oxidized by 1O2 to form stable nitroxide radicals, which were detectable by ESR. TEMPD was found to be readily oxidized by air, causing large background signals in comparison with TEMP-OH. The ESR signal obtained by the irradiation of rose bengal with visible light in the presence of TEMP-OH consisted of two kinds of nitroxide radical overlapping. In contrast, only a single nitroxide signal was observed when TEMP-OH was reacted with 1O2 generated from EP. Therefore, the best combination should be EP as the 1O2 generator and TEMP-OH as the detection probe. When using this combination, we found that the concentrations of some organic solvents such as dimethyl sulfoxide and acetonitrile should be kept constant for reliable quantification, because the concentrations of organic solvents affect the ESR signal intensity.
 Graphical Abstract Fullsize ImageView full abstractDownload PDF (492K) Full view HTML
Graphical Abstract Fullsize ImageView full abstractDownload PDF (492K) Full view HTML -
Hiroshi Hisada, Akira Okayama, Takuya Hoshino, James Carriere, Tatsuo ...2020Volume 68Issue 2 Pages 155-160
Published: February 01, 2020
Released on J-STAGE: February 01, 2020
JOURNAL FREE ACCESS FULL-TEXT HTML
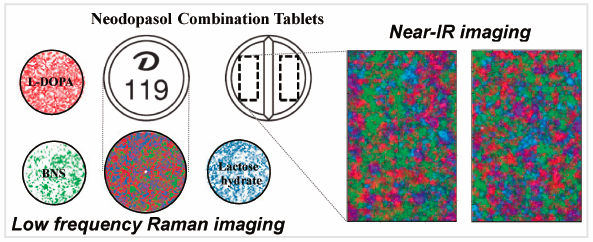
Supplementary materialCombination tablets containing multiple active pharmaceutical ingredients (APIs) are expected to improve patient convenience by decreasing the number of tablets to be taken; thus, numerous formulations containing multiple APIs have recently been developed. To allow for dose adjustments based on patient conditions, many tablets have a bisection line to allow equal division of tablets. However, there have been no investigations regarding content uniformity among divided combination tablets. Therefore, in this study, the content uniformity of combination tablets after division was investigated using near IR and low-frequency (LF) Raman spectroscopy imaging as well as the Japanese Pharmacopoeia (JP) content uniformity tests. As model drugs, five tablets of three combination drugs containing 3-(3,4-dihydroxyphenyl)-L-alanine (L-DOPA) and benserazide hydrochloride (BNS) as APIs for treating Parkinson’s disease were bisected; the resultant 10 samples were subjected to the JP content uniformity tests. We found that acceptance values of L-DOPA and BNS were 11.0–21.9% and 13.3–17.5%, respectively, with some non-conformity to the maximum allowed acceptance value (15.0%) as per the current JP. Image analyses by near IR showed that L-DOPA, BNS, lactose, and corn starch were uniformly distributed in each tablet; moreover, LF Raman spectroscopy imaging also supported the result that L-DOPA, BNS, and lactose were evenly distributed. Therefore, drug content in the tablets was uniform; thus, careful manipulation was recommended in the tablet bisection. However, the results of bisection line specifications and hardness tests revealed that the ease of division differed depending on the tablets, which warrants attention.
 Graphical Abstract Fullsize ImageView full abstractDownload PDF (4541K) Full view HTML
Graphical Abstract Fullsize ImageView full abstractDownload PDF (4541K) Full view HTML -
Guangpu Liu, Yang Jiao, Yongqiang Lin, Haifang Hao, Yanli Dou, Juan Ya ...2020Volume 68Issue 2 Pages 161-166
Published: February 01, 2020
Released on J-STAGE: February 01, 2020
Advance online publication: December 07, 2019JOURNAL FREE ACCESS FULL-TEXT HTML
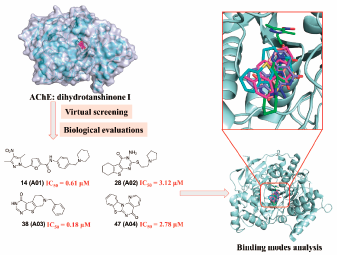
Supplementary materialDiscovery of novel multifunctional inhibitors targeting acetylcholinesterase (AChE) has becoming a hot spot in anti-Alzheimer’s disease (AD) drug development. In the present study, four potent small molecule inhibitors (A01, A02, A03 and A04) of AChE with new chemical scaffold were identified. Inhibitor A03 displayed the most potent inhibition activity on AChE at enzymatic level with IC50 value of 180 nM, and high selectivity towards AChE over butyrylcholinesterase (BChE) by more than 100-fold. The binding modes of compounds A01–A04 were carefully analyzed by molecular docking and molecular dynamics (MD) simulation to provide informative clues for further structure modification. Finally, the anti-amyloid beta (Aβ) aggregation and neuroprotective activity were also well investigated. Our findings highlighted the therapeutic promise of AChE inhibitors A01–A04 for AD treatment.
 Graphical Abstract Fullsize ImageView full abstractDownload PDF (2110K) Full view HTML
Graphical Abstract Fullsize ImageView full abstractDownload PDF (2110K) Full view HTML -
Kenji Matsumoto, Yoshinori Kobayashi, Tsukasa Hirokane, Masahiro Yoshi ...2020Volume 68Issue 2 Pages 167-172
Published: February 01, 2020
Released on J-STAGE: February 01, 2020
JOURNAL FREE ACCESS FULL-TEXT HTML
Supplementary materialA heterogeneous metal catalyst enabled the intramolecular oxidative coupling of diarylamines to form carbazoles with molecular oxygen as the sole oxidant. Rh/C had efficient catalytic activity and allowed the catalyst loading to be reduced to 0.1 mol% while maintaining excellent yields of carbazoles. This reaction is operationally simple in an open-to-air setup, and provides a green and atom-economical process for an efficient synthetic approach to N-substituted carbazoles.
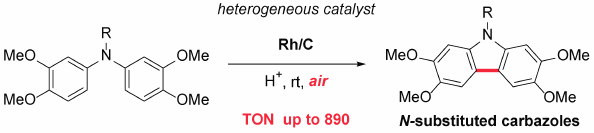 Graphical Abstract Fullsize ImageView full abstractDownload PDF (585K) Full view HTML
Graphical Abstract Fullsize ImageView full abstractDownload PDF (585K) Full view HTML -
Ji Mao, Hiroaki Itoh, Kaori Sakurai, Masayuki Inoue2020Volume 68Issue 2 Pages 173-178
Published: February 01, 2020
Released on J-STAGE: February 01, 2020
JOURNAL FREE ACCESS FULL-TEXT HTMLAn ion-channel-forming natural peptide, gramicidin A (1), exhibits potent antimicrobial activity against Gram-positive bacteria, although medical applications are limited to topical use due to its mammalian cytotoxicity. We recently reported that the artificial macrocyclic analogue 2 provides a promising starting point for developing new ion-channel-based systemic antibacterial agents because of its low mammalian cytotoxicity compared to that of the parent 1. To dissect the molecular factors involved in the species selectivity of 2, we evaluated the ion transport activities, phospholipid affinities, and conformational properties of 1 and 2 using various compositions of phospholipids. A combination of lipid dot blot assays and circular dichroism (CD) analysis with H+/Na+ exchange assays revealed that the higher H+/Na+ exchange activity of 2 than that of 1 in liposomes containing 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphatidylcholine (POPC) or 1-palmitoyl-2-oleoyl-sn-glycero-3-phospho-(1′-rac-glycerol) (POPG) is attributable to its higher affinity towards the phospholipids than that of 1. Notably, we also discovered that 2 showed weaker H+/Na+ exchange activity in liposomes containing 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphatidylethanolamine (POPE). CD analysis of 2 in liposomes indicated that the weak H+/Na+ exchange activity is induced by disturbance of the ion-conducting β6.3-helical conformation in the POPE-containing lipid bilayer. These results suggest that the POPE-induced attenuation of the ion-conducting activity of 2 contributes to the alleviation of undesirable mammalian cytotoxicity of 2 compared to that of 1.
 Graphical Abstract Fullsize ImageView full abstractDownload PDF (778K) Full view HTML
Graphical Abstract Fullsize ImageView full abstractDownload PDF (778K) Full view HTML
-
Ken-ichi Izutsu, Yasuhiro Abe, Chikako Yomota, Hiroyuki Yoshida2020Volume 68Issue 2 Pages 179-180
Published: February 01, 2020
Released on J-STAGE: February 01, 2020
JOURNAL FREE ACCESS FULL-TEXT HTMLThe purpose of this study was to clarify applicability of three-dimensional X-ray micro-computed tomography (3D X-ray micro-CT) to elucidate interior morphology of spherical adsorptive carbon fine granules. Scanning of small single spherical granule hold on the rotating sample stage provided the structural information without particular preparation (e.g., slicing) that can affect the definite morphology. The three model formulations with similar appearance showed different internal structure in the 3D images, including large hollow in one of them. Other formulations showed some small empty or higher density area in the filled granules, suggesting uneven distribution of carbon. The results indicated relevance of the X-ray micro-CT analysis on the physical characterization of the spherical adsorptive carbon granule formulations.
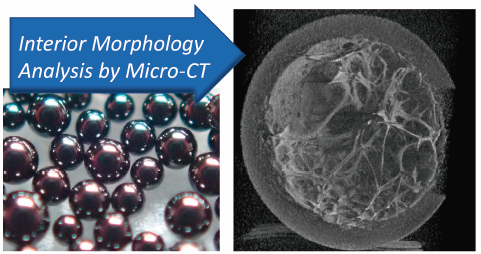 Graphical Abstract Fullsize ImageView full abstractDownload PDF (1576K) Full view HTML
Graphical Abstract Fullsize ImageView full abstractDownload PDF (1576K) Full view HTML
- |<
- <
- 1
- >
- >|