巻号一覧

後続誌
34 巻, 9 号
選択された号の論文の10件中1~10を表示しています
- |<
- <
- 1
- >
- >|
巻頭言
-
猪飼 篤2013 年 34 巻 9 号 p. 475
発行日: 2013/09/10
公開日: 2013/09/21
ジャーナル フリーPDF形式でダウンロード (209K)
特集:生体材料,細胞表面のSPM評価
-
福間 剛士原稿種別: 研究紹介
2013 年 34 巻 9 号 p. 476-481
発行日: 2013/09/10
公開日: 2013/09/21
ジャーナル フリーRecent progress in frequency modulation atomic force microscopy (FM-AFM) has enabled its operation in liquid with true atomic resolution. However, the information contained in a two-dimensional height image obtained by FM-AFM is often insufficient for understanding the structures and phenomena at a solid/liquid interface. In this study, we have developed a method referred to as 3D scanning force microscopy (3D-SFM). Combined with FM-AFM, the method enables to visualize 3D distribution of water and fluctuating surface structures at solid/liquid interfaces. Here we present basic principle and applications of 3D-SFM. The 3D-SFM image obtained at a mica/water interface shows 3D distribution of the hydration layers and adsorbed water molecules with subnanometer-scale resolution. In addition, the 3D-SFM image obtained at a lipid/water interface shows 3D distribution of the fluctuating lipid headgroups. 抄録全体を表示PDF形式でダウンロード (1325K)
抄録全体を表示PDF形式でダウンロード (1325K) -
牛木 辰男, 中島 真人, 岩田 太原稿種別: 研究紹介
2013 年 34 巻 9 号 p. 482-487
発行日: 2013/09/10
公開日: 2013/09/21
ジャーナル フリーScanning ion conductance microscopy (SICM), first introduced by Hansma et al. (1989), can obtain topographic images of the sample surface and has been expected to be useful for biological studies. In this paper, we explained the principle of the SICM and showed some results of the application of SICM to biology. We showed SICM images of collagen fibrils, chromosomes and cultivated cells in liquid. We then applied SICM for imaging the surface of tissue blocks (e.g., trachea, etc.). Sequential time-lapsed SICM images of live cells were also shown for revealing the movement of cellular processes on the time scale of minutes. The advantages of SICM imaging in biology were discussed by comparison with atomic force microscopy (AFM) and/or scanning electron microscopy (SEM) images. 抄録全体を表示PDF形式でダウンロード (1748K)
抄録全体を表示PDF形式でダウンロード (1748K) -
高橋 康史, 珠玖 仁, 末永 智一原稿種別: 研究紹介
2013 年 34 巻 9 号 p. 488-493
発行日: 2013/09/10
公開日: 2013/09/21
ジャーナル フリーThe micro/nanoelectrode has unique characteristics, which are not expected for conventional electrodes, such as the capability to perform localized measurements, low double layer charging currents, low ohmic drops, and fast mass transport. Scanning electrochemical microscopy (SECM) with a micro/nanoelectrode for detecting electroactive chemical species and is an effective tool for the investigation of the localized chemical properties of sample surfaces and interfaces. Because SECM has high temporal resolution under physiological conditions, it is particularly well suited for quantitative measurements of chemicals like neurotransmitters, nitric oxide, reactive oxygen species, and oxygen, which are released/consumed by living cells. This article presents an overview of the recent progress of SECM.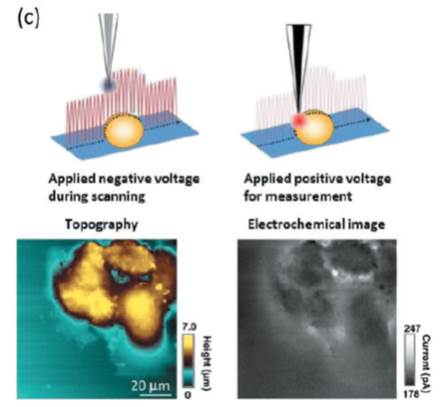 抄録全体を表示PDF形式でダウンロード (836K)
抄録全体を表示PDF形式でダウンロード (836K) -
小出 裕基, 久保 光亮, 関根 泰斗, 水下 佳紀, Narangerel GANBAATAR, 林 智広原稿種別: 研究紹介
2013 年 34 巻 9 号 p. 494-499
発行日: 2013/09/10
公開日: 2013/09/21
ジャーナル フリーThe mechanism underlying the bioinertness of nonfouling self-assembled monolayers was investigated with protein adsorption experiments, platelet adhesion tests, and surface force measurements with an atomic force microscope. Our force measurements revealed strong repulsion operating between nonfouling SAMs in the range of 4 to 6 nm in PBS buffer solution. In addition, we found that the protein-adsorbing and cell-adhering SAMs did not exhibit such repulsion. We concluded that the repulsion originated from structured interfacial water molecules. Considering the correlation among the above results, we propose that the structured interfacial water with a thickness of 2 to 3 nm (half of the range of the repulsion observed in the surface force measurements) plays an important role in deterring proteins and cells from adsorption or adhesion.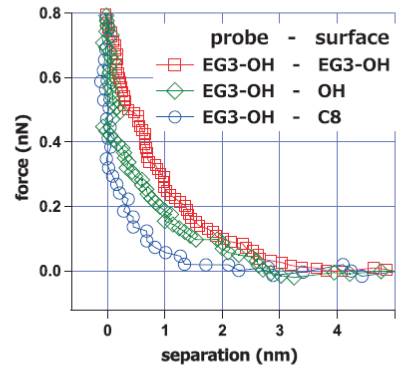 抄録全体を表示PDF形式でダウンロード (740K)
抄録全体を表示PDF形式でダウンロード (740K) -
水谷 武臣, 川端 和重原稿種別: 研究紹介
2013 年 34 巻 9 号 p. 500-504
発行日: 2013/09/10
公開日: 2013/09/21
ジャーナル フリーAtomic force microscope (AFM) has advantages in the measurement of mechanical properties of soft materials. Here, we introduced applications of AFM to elucidate a single cell mechanics and multi-cellular dynamics. Comparison of elasticity map of fibroblasts with distribution of intracellular cytoskeletal fibers showed correspondence of high elasticity regions andactin fiber distributions. A representative morphology of cellular membrane ruffling was clearly observed and elasticity of the ruffling regions was higher than that of the outside of the ruffling regions. Elasticity map of epithelial cells showed cell-cell contact point is high elasticity. Cellular elasticity measurements by AFM will be applied to medical usages and some modifications of AFM will open a new door to study dynamics of soft materials.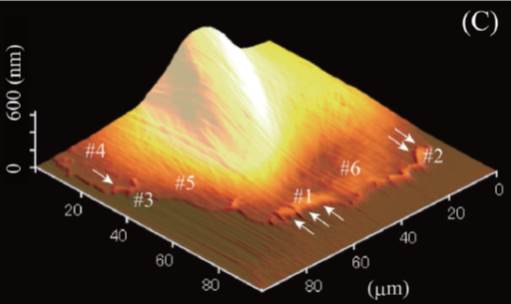 抄録全体を表示PDF形式でダウンロード (901K)
抄録全体を表示PDF形式でダウンロード (901K)
連載企画
エネルギー問題と表面科学
-
土屋 賢次2013 年 34 巻 9 号 p. 505-506
発行日: 2013/09/10
公開日: 2013/09/21
ジャーナル フリーPDF形式でダウンロード (593K)
談話室
海外研究体験記
-
鈴木 孝将2013 年 34 巻 9 号 p. 507-508
発行日: 2013/09/10
公開日: 2013/09/21
ジャーナル フリーPDF形式でダウンロード (443K)
先端追跡
-
2013 年 34 巻 9 号 p. 509
発行日: 2013/09/10
公開日: 2013/09/21
ジャーナル フリーPDF形式でダウンロード (182K)
表面科学技術者資格認定試験例題
-
2013 年 34 巻 9 号 p. 510-511
発行日: 2013/09/10
公開日: 2013/09/21
ジャーナル フリーPDF形式でダウンロード (2375K)
- |<
- <
- 1
- >
- >|
