巻号一覧

後続誌
36 巻, 7 号
選択された号の論文の13件中1~13を表示しています
- |<
- <
- 1
- >
- >|
巻頭言
-
本間 芳和2015 年 36 巻 7 号 p. 333
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリーPDF形式でダウンロード (172K)
第34回表面科学学術講演会特集号 [I]
-
薄井 洋行, 成田 匡邦, 藤田 佳宏, 坂口 裕樹原稿種別: 論文
2015 年 36 巻 7 号 p. 334-338
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリーWe prepared Ni-P-coated Si particles by an electroless deposition method as an anode material of a high-performance Li-ion battery, and optimized the interface between electrode and electrolyte by using an ionic liquid electrolyte. For the improvement of an adhesion between Ni-P and Si, we carried out a surface roughening of uncoated Si by a chemical etching, and then a thermal treatment of Ni-P-coated Si particles. Galvanostatic charge-discharge tests were performed for Ni-P-coated Si composite electrodes prepared by a gas-deposition method using the resulting particles. The results showed that an enhanced anode performance was obtained for the electrode of annealed Ni-P/(etched Si) in a conventional organic electrolyte due to the improved adhesion. In addition, the electrode exhibited a significantly stable cyclability in the ionic liquid electrolyte: a high discharge capacity of 1000 mAhg-1 was maintained with no capacity fading for 1100 cycles. 抄録全体を表示PDF形式でダウンロード (1062K)
抄録全体を表示PDF形式でダウンロード (1062K) -
久保田 純, 堂免 一成原稿種別: 研究紹介
2015 年 36 巻 7 号 p. 339-344
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリーDevelopment of non-platinum cathode catalysts for polymer electrolyte fuel cell (PEFC) is one of the indispensable subject for broad commercialization of PEFC. The requirements of cathode catalysts for PEFC are stability in strong acidic conditions, appropriate electroconductivity, and high surface area as well as activity of reaction sites. Oxide and nitride catalysts of group 4 and 5 element, such as Ti, Nb, Ta, and Zr, has obvious resistivity for strong acidic conditions as same as the precious metals, so that they are expected to be applied to cathode catalysts. With nano-sizing of catalyst particles, these materials show obvious activity. This article stands on the recent achievement of developments of non-platinum cathode catalysts of oxide and nitride catalysts of group 4 and 5 elements, such as Ti, Nb, Ta, and Zr reported by our group. 抄録全体を表示PDF形式でダウンロード (1531K)
抄録全体を表示PDF形式でダウンロード (1531K) -
池田 隆司, Zhufeng HOU, Guo-Liang CHAI, 寺倉 清之原稿種別: 研究紹介
2015 年 36 巻 7 号 p. 345-350
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリーCarbon alloy catalysts (CACs) are one of promising candidates for platinum-substitute cathode catalysts for polymer electrolyte fuel cells. We have investigated possible mechanisms of oxygen reduction reactions (ORRs) for CACs via first-principles-based molecular dynamics simulations. In this contribution, we review possible ORRs at likely catalytic sites of CACs suggested from our simulations. 抄録全体を表示PDF形式でダウンロード (1495K)
抄録全体を表示PDF形式でダウンロード (1495K) -
松永 宗一郎, 片桐 創一原稿種別: 研究紹介
2015 年 36 巻 7 号 p. 351-356
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリーZr/O/W Schottky emitter surface was investigated by using surface analytical techniques. We improved RHEED equipment by installing a heating stage and a micro-imaging system, and surface periodicity on the Zr/O/W(100) and (110) of a Schottky emitter tip have been directly measured. Reversible structural change from c(2×4) + c(4×2) to p(1×1) has been observed only in (100) surface. We also measured initial velocity of the secondary ions by using specialized TOF-SIMS and investigated generation sites of Zr, W, and their oxides. A structural model of a Zr/O/W surface was proposed that Zr atoms are adsorbed on O atoms in W-O layers formed on the W single crystal. 抄録全体を表示PDF形式でダウンロード (1257K)
抄録全体を表示PDF形式でダウンロード (1257K) -
中尾 秀信, 床波 志保, 山本 陽二郎, 椎木 弘, 武田 良彦原稿種別: 研究紹介
2015 年 36 巻 7 号 p. 357-362
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリーWe report on a facile method for preparing fluorescent carbon nanowires (CNWs) with pyrolysis of highly aligned DNA nanofibers as carbon source. Silver nanoparticles (AgNPs)-doped CNWs were also produced using pyrolysis of DNA nanofibers with well-attached AgNPs, indicating emission enhancement assisted by localized plasmon resonances. Furthermore, surface enhanced Raman scattering (SERS) imaging of AgNPs-doped CNWs revealed the generation of carbonization involved in pyrolysis process of DNA molecules. 抄録全体を表示PDF形式でダウンロード (1480K)
抄録全体を表示PDF形式でダウンロード (1480K) -
石倉 真保, 鈴木 悠宜, 二又 政之原稿種別: 研究紹介
2015 年 36 巻 7 号 p. 363-368
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリーIn order to clarify the relevance of gap mode Raman spectroscopy in metal nanoparticles/gap/metal substrates, we investigated the effect of dielectric constants of substrates, and the size of gold nanoparticles (AuNPs). First, we obtained pronounced enhancement of 105-108 in Raman scattering of thiol molecules on various metal substrates with large damping, and even on silicon substrates. Also, significantly larger enhancement was obtained for larger AuNPs, fopr example by a factor of 103 for larger particles with a radius (r) of 50 nm compared with those with r = 15 nm. These experimental observations can be explained by a coupling of dipole in AuNPs formed at localized surface plasmon resonance, and image dipole induced in the substrates. The observed properties in gap mode Raman spectroscopy are consistent with those anticipated by theoretical calculations using finite difference time domain (FDTD) method. 抄録全体を表示PDF形式でダウンロード (1868K)
抄録全体を表示PDF形式でダウンロード (1868K) -
有馬 健太, 川合 健太郎, 森田 瑞穂原稿種別: 研究紹介
2015 年 36 巻 7 号 p. 369-374
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリーWe demonstrate the metal-assisted chemical etching of Ge surfaces in water including dissolved oxygen molecules (O2). It is shown that Ge surfaces around loaded Ag or Pt particles are etched anisotropically in water. The origin of the formation of etch pits on Ge (100) is explained by the enhanced oxidation of Ge to be soluble GeO2 around the metallic particles by their catalytic activity, reducing dissolved O2 to H2O molecules. The reason for the appearance of (111) microfacets is also discussed. Secondly, we apply this electroless chemical etching in water for the nanoscale patterning of Ge surfaces using a cantilever probe in an atomic force microscopy setup. We investigate the effects of probematerials, dissolved-O2 concentration and pressing forces on the etched depth of the Ge surface. The deepest pattern is obtained when both a Pt-coated probe and saturated-dissolved-oxygen water are used. 抄録全体を表示PDF形式でダウンロード (1278K)
抄録全体を表示PDF形式でダウンロード (1278K) -
山田 豊和原稿種別: 研究紹介
2015 年 36 巻 7 号 p. 375-381
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリー1-nm-size single organic molecules have attracted as a key material to realize ultra-small spintronic devices such as single molecular giant magnetoresistance memory. The origin of such unique magnetic properties is spin-polarization vector in the single molecules induced by contacting magnetic electrodes. Atomically-resolved spin-polarized STM has been used to measure quantitatively the spin polarization; however, in this study we point out that the reported method includes crucial ambiguity. A use of normalized (dI/dV)/T shows true spin polarization of the single molecules on magnetic domains. 抄録全体を表示PDF形式でダウンロード (1129K)
抄録全体を表示PDF形式でダウンロード (1129K)
連載企画
環境問題と表面科学
-
竹内 義明2015 年 36 巻 7 号 p. 382-384
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリー日本に甚大な被害をもたらした台風としてよく知られているのは1959年の伊勢湾台風で,死者・行方不明者5098人を記録している。台風被害は,強風被害,高潮被害,大雨による洪水・浸水および土砂災害,台風通過に伴う竜巻被害,塩風害,うねりや風浪による船舶被害などが特徴的である。先進国においては人的被害こそ減っているが,都市域での大規模水害による交通・通信等インフラの広域被害がしばしば起こっている。台風は将来にわたって社会的に影響の大きな気象現象であるため,世界中で活発に研究が行われている。本稿では,台風の発生から消滅までを概説したのち,進展著しい観測技術と研究の動向(特に,「表面」に関わる部分)についてその一端を紹介する。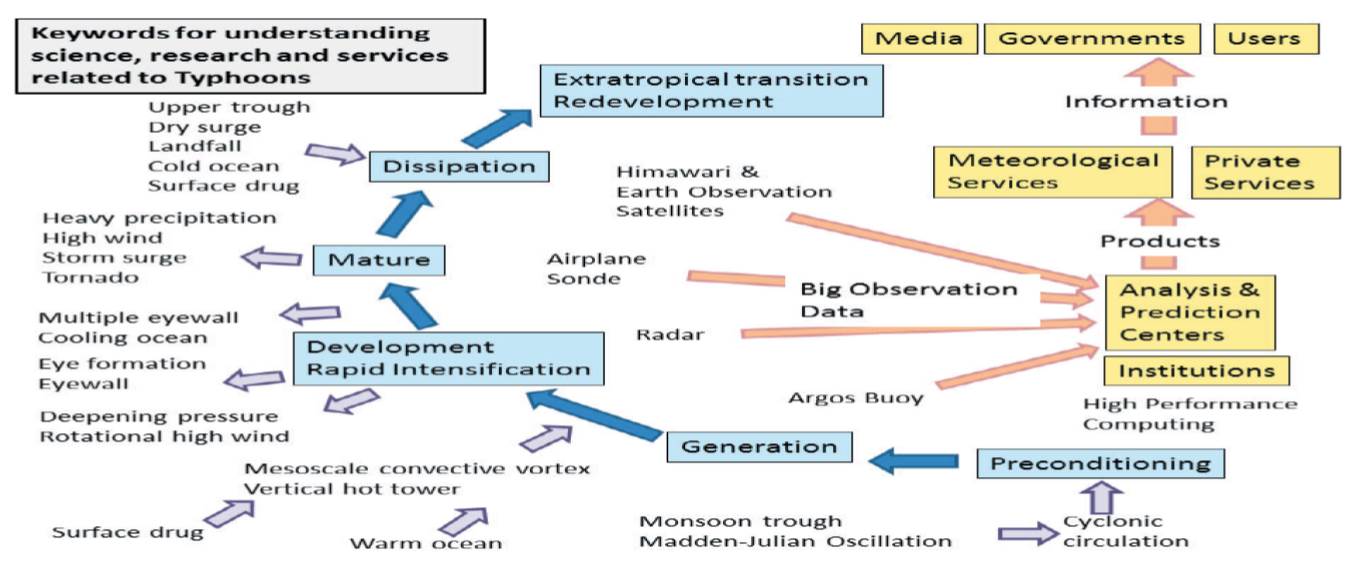 抄録全体を表示PDF形式でダウンロード (556K)
抄録全体を表示PDF形式でダウンロード (556K)
談話室
海外研究体験記
-
池田 亜矢子2015 年 36 巻 7 号 p. 385-386
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリーPDF形式でダウンロード (316K)
表面科学技術者資格認定試験例題
-
2015 年 36 巻 7 号 p. 387
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリーPDF形式でダウンロード (1361K)
先端追跡
-
2015 年 36 巻 7 号 p. 388
発行日: 2015/07/10
公開日: 2015/07/21
ジャーナル フリーPDF形式でダウンロード (133K)
- |<
- <
- 1
- >
- >|