
- |<
- <
- 1
- >
- >|
-
Hidetoshi SomekawaArticle type: Review
2019Volume 83Issue 3 Pages 65-75
Published: March 01, 2019
Released on J-STAGE: February 25, 2019
Advance online publication: February 18, 2019JOURNAL FREE ACCESS FULL-TEXT HTMLDevelopment of magnesium alloys, which exhibit high strength and high ductility (fracture toughness), is critical for ensuring safety and reliability in structural applications. It is well-known that grain refinement and/or alloying are impressive strategies to attain such properties in metallic materials. In the former case, grain boundaries of magnesium and its alloys have unique characteristics, e.g., sites for non-basal dislocation activity and occurrence of partial grain boundary sliding. As a result, strength as well as ductility (fracture toughness) tend to increase and improve with grain refinement. In the latter case, 29 types of solid solution elements, which have a maximum solubility of more than 0.1 at%, can dissolve in magnesium. Several elements are generally added to magnesium simultaneously to achieve good mechanical properties via a synergistic effect. In industrial fields, ternary magnesium alloys such as Mg-Al-Zn and Mg-Zn-Zr alloys, which have fine-grained structures, have been widely used; however, there is no still clear and systematic understanding of the impact of various alloying elements on properties for magnesium. In this paper, we review recent results on the effect of solid solution alloying elements on ductility (fracture toughness), with focusing on polycrystalline binary magnesium alloys. Regarding the toughness, crack-propagation behavior and/or fracture behavior are quite sensitive to the alloying element, regardless of the grain size. Twin boundaries in particular are recognized as harmful defects, because the act as crack-propagation site. Nevertheless, changing the electric bonding behavior through alloying has the potential to increase toughness. As for the ductility, alloying elements also dramatically affect the room-temperature plastic deformation; activation of not only non-basal dislocation slip but also grain boundary sliding plays a notable role in enhancing the elongation-to-failure in tension.
 Graphical Abstract Fullsize ImageView full abstractDownload PDF (1461K) Full view HTML
Graphical Abstract Fullsize ImageView full abstractDownload PDF (1461K) Full view HTML
-
Takayuki Kodera, Takashi Ogihara, Kenji Ohshita, Nobuyoshi Aoyagi, Yum ...Article type: Regular Article
2019Volume 83Issue 3 Pages 76-81
Published: March 01, 2019
Released on J-STAGE: February 25, 2019
Advance online publication: January 18, 2019JOURNAL FREE ACCESS FULL-TEXT HTMLMonodispersed silver particles were prepared through wet chemical reduction method. As-prepared silver particles had uniform polyhedron morphology with narrow size distribution. Powder X-ray diffraction analysis showed that the silver powders had high crystallinity. The particle size of silver powders was controlled by adjusting the concentration of starting reagents and temperature. In a wet chemical reduction reaction, monodispersed silver particles were formed when the molar ratio of reducing agent and silver nitrate was 0.5. It was possible to control the particle size from 120 nm to 800 nm by adjusting the concentration of silver nitrate. The particle size of silver powders decreased to 70 nm with increasing the concentration of gum Arabic. Thermal mechanical measurement showed that the shrinkage of silver paste was 25% at 900℃. Electrical measurements revealed that the specific resistivity of the silver paste sintered at 900℃ for 10 min was 1.81 × 10−8 Ω m. It was expected that the silver powders exhibited excellent electrical property as electrode materials for integrated circuits, multilayer ceramic capacitor and solar cells etc.
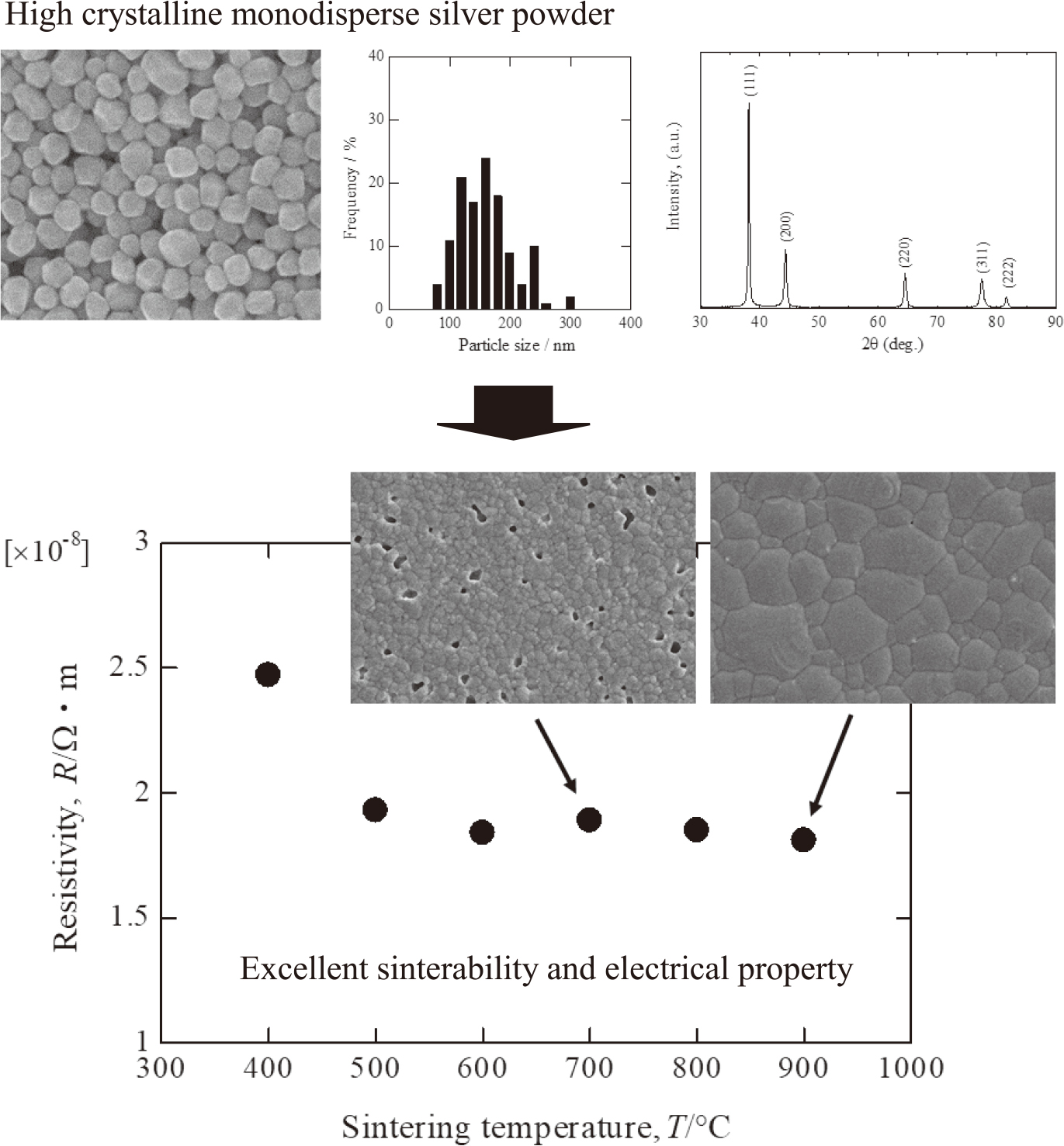 Graphical Abstract Fullsize ImageView full abstractDownload PDF (969K) Full view HTML
Graphical Abstract Fullsize ImageView full abstractDownload PDF (969K) Full view HTML -
Hidenobu Maruno, Akio NishimotoArticle type: Regular Article
2019Volume 83Issue 3 Pages 82-86
Published: March 01, 2019
Released on J-STAGE: February 25, 2019
Advance online publication: February 01, 2019JOURNAL FREE ACCESS FULL-TEXT HTMLAluminum alloys are light and have good workability; however, their low hardness and poor wear resistance are drawbacks limiting their wide application in the automotive industry. The deposition of diamond-like carbon (DLC) films, which exhibit high hardness and good wear resistance, onto the surface of Al alloy substrates, can overcome these drawbacks. As Al alloys and DLC films have low affinity for each other, adhesion between the two is poor; adhesion can be improved by using of an interlayer. For coatings with the same thickness, film properties are improved by forming multilayer structures. In this study, the sample was formed with a uniform film thickness of 2 µm, to change the thickness ratio of the Si-DLC/DLC layer to 1:1 or 1:3 and to change the number of multilayers [Si-DLC/DLC]N to 1, 2, and 4. The samples were then tested by nanoindentation, Rockwell indentation, scratch, and wear tests. The sample with a thickness ratio of 1:1 increased the hardness and hardness/Young's modulus (H/E) of the film by increasing the number of multilayers repetitions. However, for the sample with a thickness ratio of 1:3, 2 and 4 multilayer repetitions did not effect a significant difference in the hardness and H/E ratio. These samples did not improve the adhesion properties, because of no significant difference by the Rockwell indentation and scratch tests. By the wear test, samples with a 1:1 thickness ratio of the multilayer increased the delamination length and decreased the friction coefficient with increasing number of multilayers repetitions. Samples with a 1:3 thickness ratio of the multilayer increased the delamination length with increasing number of multilayers repetitions, whereas samples with 2 and 4 multilayers did not cause a significant difference in the delamination length. Moreover, the friction coefficient of the samples decreased with increasing number of multilayers repetitions. This study showed the delamination length of DLC films is proportional to increasing H/E ratio.
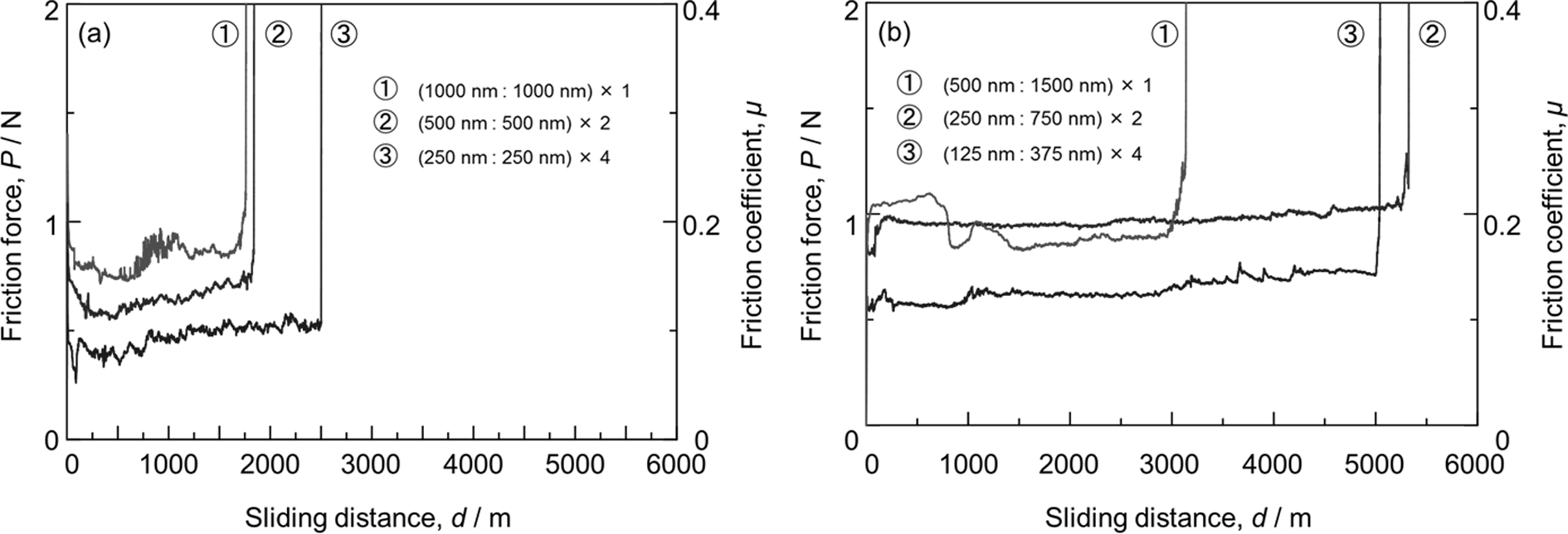 Fig. 3 Friction change curves of Si-DLC/DLC multilayer coating during the wear test: (a) thickness ratio of 1:1 and (b) thickness ratio of 1:3. Fullsize ImageView full abstractDownload PDF (1608K) Full view HTML
Fig. 3 Friction change curves of Si-DLC/DLC multilayer coating during the wear test: (a) thickness ratio of 1:1 and (b) thickness ratio of 1:3. Fullsize ImageView full abstractDownload PDF (1608K) Full view HTML -
Fumiyoshi Kirino, Naoshi Ohono, Satoko TaguchiArticle type: Regular Article
2019Volume 83Issue 3 Pages 87-96
Published: March 01, 2019
Released on J-STAGE: February 25, 2019
Advance online publication: February 08, 2019JOURNAL FREE ACCESS FULL-TEXT HTMLThe metal craft production techniques for an ancient Chinese coin named Ittou in the Shin period of ancient China were studied from material science viewpoints. This cultural artefact was manufactured in the Shin period of ancient China. An infrared image shows the surface state and the materials absorbed on the surface. According to infrared images of the cultural artefact, roughness on the surface of this coin was created on cooling during mintage. UV-fluorescence images show luminescence at corrosion products and yellow soil. In X-ray transmission images, some voids in this coin were observed, indicating the metal craft production technique used during this period. The main element of this cultural artefact is Cu and other elements are Sn and Pb. X-ray diffraction patterns indicate the presence of Cu-Sn base intermetallic compounds. Ittou coins are made from bronze. The characters on the coins were formed using an inlay technique called zougan. Yellow soil adhered to the surface of the examined coin from where it was excavated. The material used to form the characters on the coin is 10 mass% Cu-Au alloy, and flaws were observed on the surface of the characters. The compositions of ancient Kan coins produced between the east Kan and west Kan periods were also analyzed. The composition of these ancient coins was the same as that of the coin from the Shin period of old China, indicating that the same metal craft production technique was used in both periods. These techniques were improved in this period, enabling many coins to be manufactured.
View full abstractDownload PDF (20033K) Full view HTML -
Tadahiko Furuta, Katsuomi Shiina, Yoshihisa Ueda, Shinobu Shimazaki, K ...Article type: Regular Article
2019Volume 83Issue 3 Pages 97-106
Published: March 01, 2019
Released on J-STAGE: February 25, 2019
Advance online publication: February 08, 2019JOURNAL FREE ACCESS FULL-TEXT HTMLThe effects of the C and Al content on the resistivity and rigidity of newly developed ultra-high strength titanium-based metal matrix composites (Ti-MMCs) fabricated using a technique that we refer to as blended elemental reactive sintering (BERS), were investigated. The composition of the MMCs was TiC(1−X)/Ti-6Al-4V and Ti-8.6Al-5.7V, which were compared with those for TiB/, SiC/and AlN/Ti-MMCs. The blended TiC reacted with Ti powder and transformed to TiC(0.50~0.62) during sintering. The resulting TiC(0.50~0.62)/Ti-8.6Al-5.7V exhibited a specific resistance of 2.3 μΩm, a Young's modulus of 135 GPa, and a tensile strength of 1.25 GPa, with a substantial elongation of approximately 2.5%. On the other hand, TiB/Ti-6Al-4V had excellent mechanical properties, but an extremely low electrical resistance because the conducting TiB particles have a resistivity of only 0.07 μΩm. Blended SiC and AlN also reacted with Ti powder during sintering and formed a brittle phase at the interface between the particles and the Ti matrix. As a result, Ti-6Al-4V MMCs that are suitable for use as structural materials could not be fabricated using BERS with SiC or AlN.
The reasons for the high specific resistance of TiC(0.50~0.62)/Ti-8.6Al-5.7V are considered to be as follows. First, due to their C deficiency, the resistivity of individual TiC(0.50~0.62) particles is approximately 1.7 μΩm, which is about three times higher than the value of 0.52 μΩm for stoichiometric TiC particles. In addition, the solubility of the excess C and Al in the Ti matrix also increases the specific resistance.
 Graphical Abstract Fullsize ImageView full abstractDownload PDF (5271K) Full view HTML
Graphical Abstract Fullsize ImageView full abstractDownload PDF (5271K) Full view HTML
- |<
- <
- 1
- >
- >|